11 November 2025 — In a drug development landscape defined by shifting policies, geopolitical uncertainty, and increasing competitive pressure, a new collaborative strategy platform, Nmblr, has launched to empower teams to reach peak sales faster and with greater confidence. Built on decades of healthcare expertise, Nmblr helps mid-sized biopharma and med-tech organisations move from breakthrough science to real-world impact - where payors see value, physicians want to prescribe, and patients have access to innovation.
Designed to foster agility and alignment, Nmblr connects every function and team – global, regional, and local – so they move as one and create effective commercialisation strategies that ensure assets show up in the right shape and to a receptive market — maximising both commercial success and patient impact.
This news follows research commissioned by Nmblr that confirmed nearly three out of five biopharma professionals (58%) believe their company’s potential is restricted by rigidity in daily strategic decision-making. The findings reveal that strategic misalignment is endemic across the industry, threatening both speed to market and the ability to adapt in real-time. The survey found that poor cross-functional alignment (47%), inadequate up-front data planning (57%), and the recycling of outdated strategies (85%) all contribute to significant inefficiency, resulting in almost one in five (18.9%) pharmaceutical product launches being delayed.
In response, the launch of Nmblr signals a bold paradigm shift for the industry, moving beyond siloed approaches to empower purpose-driven teams to act in unison, guided by real-time insights and shared objectives. Intuitive and feature-rich, Nmblr supports teams from concept to launch and beyond to move as one — addressing long-standing barriers, such as siloed teams with misaligned incentives, varying degrees of understanding, and an abundance of data but too little distilled insight.
“The pace and complexity of today’s biopharma landscape means that great science alone is no longer enough,” said Janice MacLennan, Founder of Nmblr. “Commercial success depends on how effectively organisations can connect their thinking — aligning diverse teams, insights, and objectives around one clear and agile strategy. Nmblr was built to make that possible. It gives teams the structure, visibility, and shared purpose they need to turn breakthrough science into real-world impact.”
Industry experts have welcomed the launch. “Most companies still improvise their commercial planning - and many struggle if not stumble as a result,” said Peter Vanovertveld, senior leader in healthcare access and business strategy. “Even with good people, if there isn’t structure and discipline, it’s very hard to build an effective commercialisation strategy. There’s a science and an art to doing it well and Nmblr strengthens both. It’s like an exoskeleton for the commercial strategy process. It doesn’t replace human leadership, but it makes teams stronger, more coordinated, and more resilient. It puts years of proven marketing thinking into a digital, practical framework for healthcare - helping organisations move faster and with far greater confidence.”
Department of Pharmaceuticals intensified monitoring of availability, production and supply of drugs used for Covid-19 Treatment in early April, 2021 as number of cases of Covid-19 started increasing steeply. Remdesivir is a patented drug, manufactured in India under voluntary licenses granted by Gilead Life Sciences USA, the patent holder, to 7 Indian pharmaceutical companies (Cipla, Dr Reddy’s Lab., Hetero, Jubilant Pharma, Mylan, Syngene and Zydus Cadila).
To augment the domestic production capacity, all the seven domestic licensed manufacturers of Remdesivir were asked to ramp up production quickly. With combined efforts of the central government and the manufacturing companies, the production capacity of the licensed manufacturers has increased unprecedentedly from 38 lakh vials per month to nearly 119 lakh vials per month. With accelerated approval of 38 additional manufacturing sites, the number of approved manufacturing sites of Remdesivir in country has increased from 22 sites to 60 sites. With the help of Ministry of External Affairs, the manufacturers of Remdesivir are being facilitated for getting supplies of requisite raw materials and equipments from foreign countries.
All efforts are being made to enhance the availability of the drug both through imports as well as augmented domestic production. Exports of Remdesiver has been prohibited since 11th April, 2021. Customs duty has been exempted on Remdesivir injection, Remdesivir API and Beta Cyclodextrin (SBEBCD) used in the manufacture of Remdesivir with effect from 20th April 2021.
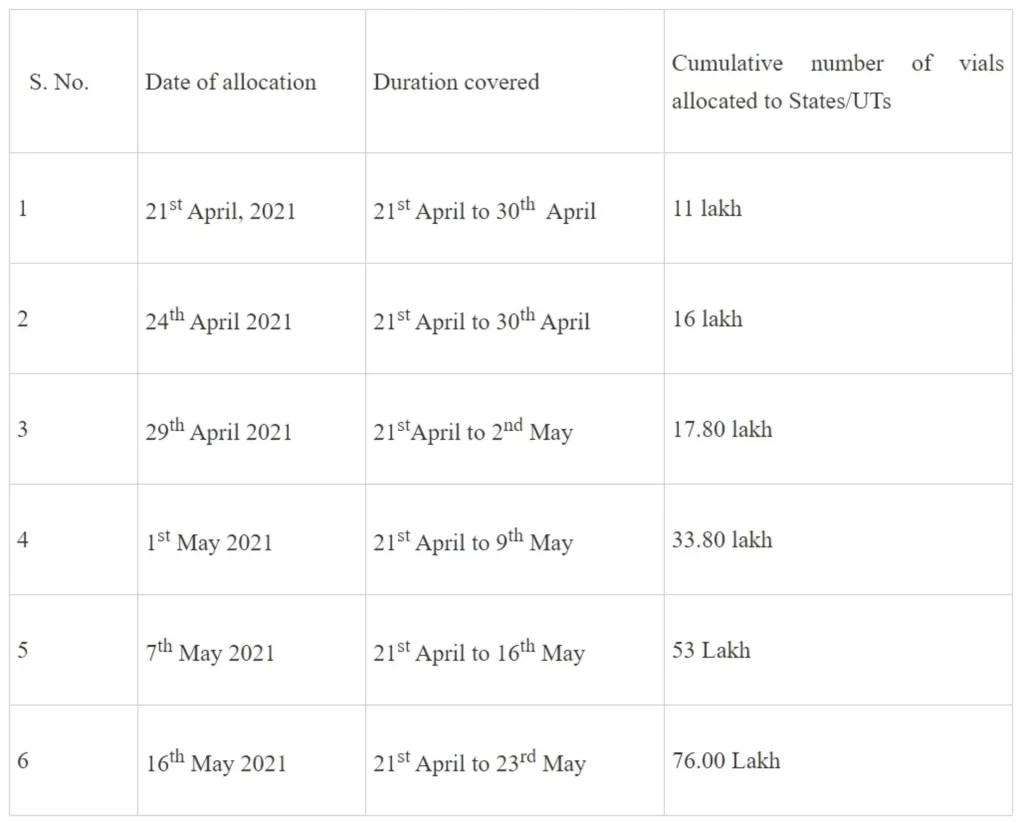
Looking to the suddenly increased demand in the country, in order to ensure equitable distribution of Remdesivir among different regions of the country, central government has been making allocations to States and UTs since third week of April. Interim allocation of 11 lakh vials was made for 19 states and UTs with high demand on 21stApril 2021 for period up to 30th April. This allocation was increased to 16 lakh vials on 24th April as more supplies became available and all States and UTs were allocated the vials. In a series of allocations issued subsequently, with latest allocation issued on 16th May, 76 lakh vials in total have been allocated among States for period up to 23rd May, 2021.
Summary of Allocation
The State Governments and UTs have been asked to monitor proper distribution within their jurisdiction covering both government as well as private hospitals and in line with judicious use as advised in the “National Clinical Guidance for management of adult Covid-19 patients” issued jointly by AIIMS/ICMR Covid-19 National Task Force and Joint Monitoring Group of MoHFW. State Governments / UTs have been advised to place adequate purchase orders with the marketing companies immediately for the quantity that they want to purchase out of allocation for the State/ UT as per supply plan in close coordination with liaison officers of the companies. The coordination with private distribution channel in the state is also to be made by the States. The State governments have also been advised to put in place a mechanism for issuance of this drug to the needy patients and well advertise it among the public in the State.
All the seven Indian manufacturers have been making supplies to States as per the allocation both against government purchase orders and also through their private distribution channels in States. Total 54.15 lakh vials of Remdesivir have been supplied across the country by the Drug companies during 21st April-15th May, 2021 period. Supply of Remdesivir to Various State Governments and central Institutions is continuously being monitored and whenever any complaint regarding supply is received, it is taken up immediately with the concerned manufacturer. Department of Pharmaceuticals, through National Pharmaceuticals Pricing Authority (NPPA), is in constant touch with all the States through their Nodal officers and manufacturing companies through their Liaison Offficers.
In addition to allocation mentioned above, as on 16.05.2021, a total of 5.26 lakh vials of Remdesivir, which have been received through donation from other countries/organizations and 40000 vials which have been commercially imported, have also been allocated to States and UTs.
Minister of Defence, Shri Rajnath Singh and Minister of Health and Family Welfare, Dr. Harsh Vardhan today jointly launched the new COVID drug 2-DG at DRDO Bhawan. The anti-COVID-19 therapeutic application of the drug 2-deoxy-D-glucose (2-DG) has been developed by Institute of Nuclear Medicine and Allied Sciences (INMAS), a lab of Defence Research and Development Organisation (DRDO), along with Dr Reddy’s Laboratories (DRL), Hyderabad.

One box each of the sachets of the drug was handed over to Dr Randeep Guleria. Director, All India Institute of Medical Sciences (AIIMS), New Delhi and Lt Gen Shri Sunil Kant of Armed Forces Medical Services (AFMS).
Speaking at the launch Dr Harsh Vardhan remarked that today is a historic day in our fight against COVID and congratulated the scientific community and DRDO for their patience and perseverance for developing first indigenous medicine. He said that the drug has the potential to become a game changer in our response against COVID pandemic as it reduces the dependence of patients on oxygen administration and has the potential of getting absorbed differentially and in a selected manner. In the COVID infected cells, it inhibits virus synthesis and energy production for the process.
Dr Harsh Vardhan further stated that around thirty hospitals were involved in the clinical trials of this drug. He said that the drug will go to the selective cells and prevent synthesis of virus and reduce recovery time.
Expressing his gratitude towards the Defence Minister, Dr. Harsh Vardhan said that due to the efforts of Defence Ministry and DRDO, several PSA plants have been successfully installed in Delhi and across the country.
Dr Harsh Vardhan also cautioned against complacency and said that we will win over the war against COVID by collective efforts and by following COVID Appropriate Behaviour.

Secretary Department of Defence R&D and Chairman DRDO Dr G Satheesh Reddy expressed confidence that the anti-COVID drug will help the patients recover from the deadly virus, hoping that DRL, Hyderabad will take it forward and soon make the drug available for the patients.
Joining the event virtually, Chairman, DRL Shri Kallam Satish Reddy said, “Dr. Reddy’s is glad to have partnered with DRDO and INMAS in the development of 2-DG. This is a re-affirmation of our company’s efforts to address COVID through a host of therapeutics and vaccine.”
Shri Rajesh Bhushan, Union Secretary (Health & Family Welfare), Dr Sunil Kumar, DG (Health Services), Dr Randeep Guleria, Director, AIIMS New Delhi were present at the launch event. Besides Chairman, DRL, Director, Centre for Cellular and Molecular Biology Dr Rakesh Mishra and many doctors, hospitals & labs across the country joined the event virtually.
Minister of State Shri Mansukh Mandaviya today assured that the Government is monitoring supply of each COVID-19 Essential Drug. All drugs used in the management of COVID-19 are now available in India by ramping up production and increasing the imports. The availability of these drugs is being monitored by implementing three-pronged strategy of Supply Chain Management, Demand Side Management and Affordability.
Protocol Drugs:
Remdesivir
Enoxaparin
MethylPrednisolone
Dexamethasone
Tocilizumab
Ivermectin
Non-protocol Drugs:
Favipiravir
Amphotericin
Apixaban
CDSCO and NPPA are coordinating with manufacturers to enhance production and getting data about current stock, current capacities, Projected production for the May, 2021.
Remdesivir:
Number of plants producing Remdesivir increased from 20 to 60 resulting in 3 times more availability in just 25 days.
Production ramped up 10 times, from 10 lakhs vials/month in April’21 to ~1 crore/ month in May’21.
Toscilizumab injection:
It is made available in the country by importing 20 times more than what was being done in normal times.
Dexamethasone 0.5 mg tablets:
Production enhanced from 6-8 times within a month
Dexamethasone Injection production ramped up almost 2 times.
Enoxaparin Injection production increased 4 times in mere one month.
Methyl Prednisolone Injection:
Production increased almost 3 times in a months time.
Ivermectin 12 mg tab production enhanced in the country by ~5 times within a month from 150 lakhs in April to 770 lakhs in May,2021.
Favirpiravir:
A non-protocol drug but it is used to lower the virus load .
The production increased 4 times within a month
From 326.5 lakh in April,21 to 1644 lakh in May,21.
Amphoterecin B Injection:
Production ramped up ~ 3 times in a month.
3.80 lakh vials are in production and
3 lakh vials are being imported
Total 6.80 lakh vials will be available in the country.
The Liberalised Pricing & Accelerated National COVID-19 Vaccination Strategy has been implemented from 1st May 2021. As part of the Strategy, in every month 50% of the total Central Drugs Laboratory (CDL) cleared vaccine doses would be procured by Govt. of India, and GOI would continue to make it available to the State Govts totally free of cost, as was being done earlier. In addition to this, every month balance 50% of the CDL cleared vaccine doses would be available for direct procurement by the State Govts & Private Hospitals.
Union Health Ministry has been providing advance information on availability of COVID vaccine doses to be supplied to the States and UTs during the two fortnights of the month, and also the amount available for direct procurement by the States from the manufacturers by the State and the private hospitals. The Prime Minister has highlighted this in his interaction with the State and District Officials on the COVID-19 situation yesterday.
Union Health Ministry has again written to the States and UTs on allocation of COVID vaccine doses (for both Covishield and Covaxin) during May 2021 and during first fortnight of June 2021 from the Govt of India channel (which is available free of cost), and availability of Vaccine doses (for both Covishield and Covaxin) that can be procured directly by States & Private Hospitals during months of May and June 2021. This advance visibility will enable better and more effective planning by the States.
According to the advance visibility provided by Govt of India to States/UTs, a total of 5 crore 86 lakh and 29 thousand doses will be provided free of cost by Govt of India to States from 1st May 2021 to 15th June 2021.
In addition, as per information received from vaccine manufacturers, a total of 4 crore, 87 lakh and 55 thousand doses will also be available till end of June 2021 for direct procurement by States/UTs.
In view of the above visibility of vaccines with clear supply timelines till June 2021, and in order to ensure efficient and judicious utilization of available doses for successful implementation of COVID-19 vaccination drive, the States/UTs have been advised the following:
To prepare a district-wise, COVID Vaccination Centre (CVC)-wise plan for administration of COVID-19 vaccine.
To use multiple media platforms for dissemination of such a plan to enhance awareness among the masses.
Both the States Govts & Private CVCs to publish their vaccination calendar on CoWIN digital platform in advance.
States & Private CVCs to desist from publishing single day vaccination calendars.
To ensure that there is no overcrowding at the CVCs.
To ensure that the process of booking appointments on CoWIN is hassle-free.
The States and UTs have been advised to direct the concerned officials to prepare advance plan for administration of COVID-19 vaccine till 15th of June 2021.
The vaccination exercise as a tool to protect the most vulnerable population groups in the country from COVID-19 continues to be regularly reviewed and monitored at the highest level.
Dr. Harsh Vardhan, Union Minister of Health and Family Welfare today interacted with State Health Ministers and Principal Secretaries/ Additional Chief Secretaries of West Bengal and the 8 States of the North-eastern Region in the presence of Shri Ashwini Kumar Choubey, Union Minister of State for Health and Family Welfare. These states are depicting a higher growth rate in the number of daily cases, high mortality and increasing positivity rate.
Shri Biplab Kumar Deb, Chief Minister & Health Minister of Tripura, Shri Keshab Mahanta, Health Minister, Assam, Shri A.K Hek, Health Minister, Meghalaya, Shri LalpoklakpamJayantakumar Singh, Health Minister, Manipur, Dr R Lalthangliana, Health Minister (Mizoram), Shri AloLibang, Health Minister (Arunachal Pradesh), Shri PangnyuPhom, Health Minister (Nagaland) and Dr Mani Kumar Sharma, Health Minister (Sikkim) were present virtually in this meeting.
He applauded the dedication and patience shown by state administrations in their fight against the pandemic and in simultaneously ensuring the welfare of the people. He said that “we fought together in 2020 and will fight together in 2021 under the leadership of Hon’ble Prime Minister”.
Speaking of the government efforts that have resulted in this achievement, Dr. Harsh Vardhan outlined, “From having just 1 laboratory at the start of the pandemic, today there are over 2000 laboratories for testing COVID across the country. We have increased our capacity to test 25 lakh people daily. Yesterday, India achieved a historical first by testing the highest number of over 20 lakh people in a single day. This is a global record too”. He stressed on the continued need to follow COVID Appropriate Behaviour along with renewed and stringent focus on containment measures for addressing the present surge.
Underscoring the critical importance of vaccination drive, the Minister highlighted that India has achieved another landmark by crossing 18 cr doses recently. He thanked all the states and UTs for their contribution in achieving this feat. He also pointed out that, more than 1 crore doses are still in stock with the state governments. He added that “Between August to December 2021, India will have procured 216 crore vaccine doses while, by July this year, 51 crore doses will be procured”. Dr Harsh Vardhan also urged states to ensure that the healthcare workers and frontline workers are fully vaccinated, as they form the vulnerable categories. The Union Health Minister said that by the end of the year, the country will be in a position to vaccinate at least all of its adult population.
Dr Harsh Vardhan highlighted the critical challenges faced by the NE states and West Bengal: In Mizoram, all districts are showing a rise in new cases. Nagaland has shown a sharp increase in daily cases (15-20 per day to 300 per day) and weekly positivity rate (1% to 34%); there is a need to strengthen testing facilities in peri-urban and rural areas. In Assam, Kamrup (Metropolitan) is contributing almost 45% of the daily new cases, while in Meghalaya, East Khasi Hills and Righboi are reporting sharp increases in daily cases. Manipur’s recovery rate of 78% and CFR of more than 1% was highlighted as a matter of concern. Sikkim was advised to strengthen community surveillance and ensure strict monitoring of home quarantine to address its high CFR. In Arunachal Pradesh, ICU occupancy is almost 22.5% while Capital complex and Changlang districts are reporting maximum cases. All districts of West Bengal are showing a steep increase in positivity rate. Kolkata, North 24 Parganas, South 24 Parganas, Howrah and Nadia were flagged as districts of concern. In Tripura, there has been a steep rise in positivity from 1.3% in April to around 8.7% now. West Tripura, Unakoti, South Tripura were flagged as districts of concern.
Dr Harsh Vardhan pointed towards the new emerging trend in the country. He highlighted that now the smaller states are showing an upward trend and there is a need to be cautious about this trend. He asked the states to focus on increasing testing in a timely manner along with upgrading the health infrastructure. The Union Health Minister said that with changes in guidelines, more and more of antigen tests can be done now, so that mortality rates can be decreased.He reiterated the need to focus on peri urban and rural areas significantly.
Shri Ashwini Kumar Choubey, MOS (HFW) reiterated that testing should be increased, particularly RT-PCR. He stressed on the need for extensive use of tele-consultation especially in the hilly states to reach to the underserved areas. Centre is also assisting states on medicines for Black Fungus and on Oxygen. “We have to maintain Covid protocol and we will win this war against Covid”, he stated.
Dr Sujeet K. Singh, Director NCDC presented an overview of the COVID trajectory in the States. He suggested the ramp up of RAT testing and vaccination in rural areas as the spread is now going to pockets in the North East which are difficult to reach in terms of geography. The rise of positivity to more than 33% in some pockets was noted as a point of particular concern as it indicates high transmission in belts that are underserved by the country’s health network. He espoused the need to properly monitor home quarantine cases and enhanced surveillance for ILI/SARI cases to curb high CFR in certain Hill States.
Shri Vikash Sheel, Additional Secretary (Health) presented on the optimal and judicious use of vaccine doses. All the States, barring Tripura, has had a lower coverage of HCW/FLW workers than the National average (90% and 82% respectively). In the 45+ age-group category, Meghalaya (28%), Manipur (26%), West Bengal (25%), Assam (23%), Nagaland (22%) had a lower coverage than the National average (32%). States were advised to ensure complete utilization of available vaccination slots for 45+ age-group/HCW/FLW with awareness campaigns for conveying the importance of completing vaccination.
State administrators were advised to reduce vaccine wastage; Assam, Manipur, Meghalaya and Nagaland had clocked substantial wastage and were advised to retrain their staff to achieve minimum wastage. A 2/3-member team to be constituted at State level to regularly coordinate with manufacturers for timely supplies of vaccine through ‘Other than Govt. of India channel’ on the lines of the team operating at Union Government of India level. This 2/3 member team would also coordinate with Pvt. Hospitals. The list of Pvt. Hospitals in these States along with doses contracted and supplied have been already shared with States. Dr Harsh Vardhan assured the states of all support from the Centre in augmenting their vaccine drive.

Taking note of advice to augment their preparedness to contain COVID-19, the State Health administrators expressed their thanks for the help they have been receiving from the Union Government.
Shri Rajesh Bhushan, Union Health Secretary, Shri Lav Agarwal, Joint Secretary (Health), Dr. Sujeet K. Singh, Director, NCDC and other senior Health officials were present in the meeting.
Union Minister for Chemicals and Fertilizers Shri Sadananda Gowda held a meeting today with senior officials Department of Pharmaceuticals to review the availability of medicines being used in the management of COVID19.

In a tweet shared by the Minister he said that Department of Pharmaceuticals is working closely with the States to ensure uninterrupted availability of medicines across the country.
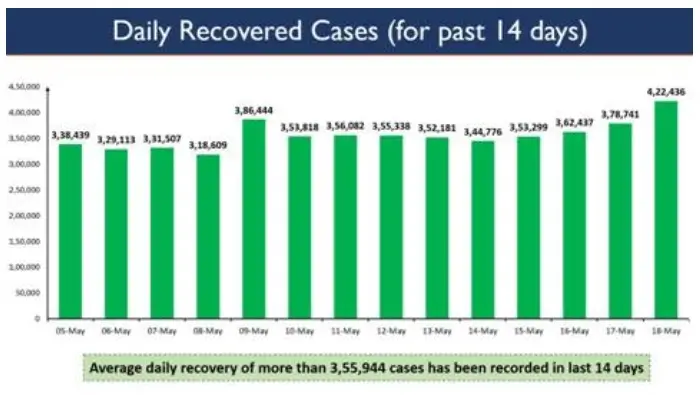
In a landmark achievement, with the dedication and tireless effort of all the healthcare and frontline workers, India has recorded daily recoveries of more than 4 lakh COVID patients in a single day for the first time.
4,22,436 recoveries were registered in the last 24 hours.
Average Daily Recovery of more than 3,55,944 cases has been recorded in the last 14 days. The graph below depicts the Daily Recoveries in the last 14 days.
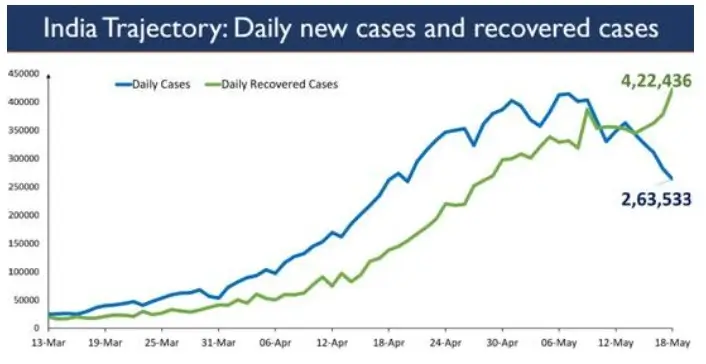
2,63,533 new cases were registered in the last 24 hours.
The daily new COVID cases being registered in India are now less than 3 Lakhs consecutively for the second day. A net decline of 1,63,232 cases is recorded in the active caseload in the last 24 hours.
India’s daily new cases trajectory and recovered cases is depicted below since 13th March 2021.
India’s cumulative recoveries have reached 2,15,96,512 today. The National Recovery Rate has grown further to touch 85.60%.
Ten states account for 75.77% of the new recoveries.
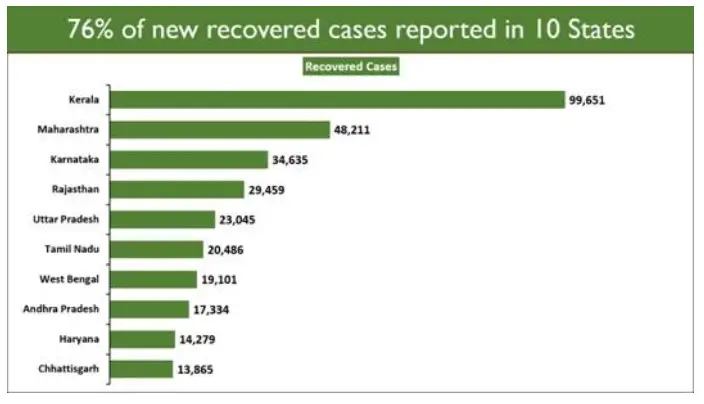
On the other front, India’s total Active Caseload has decreased to 33,53,765 today. It now comprises 13.29% of the country's total Positive Cases.
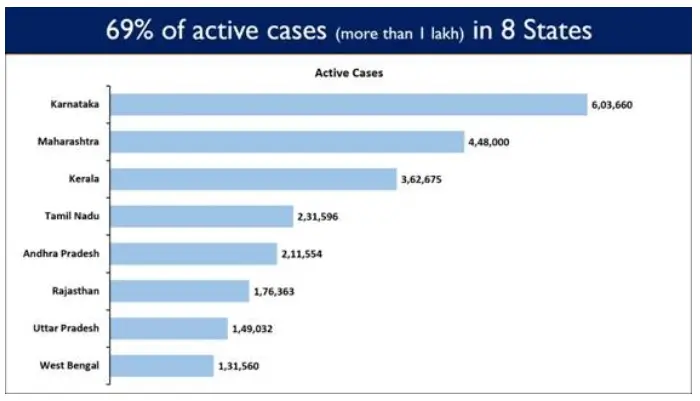
8 States cumulatively account for 69.01% of India’s total Active Cases.
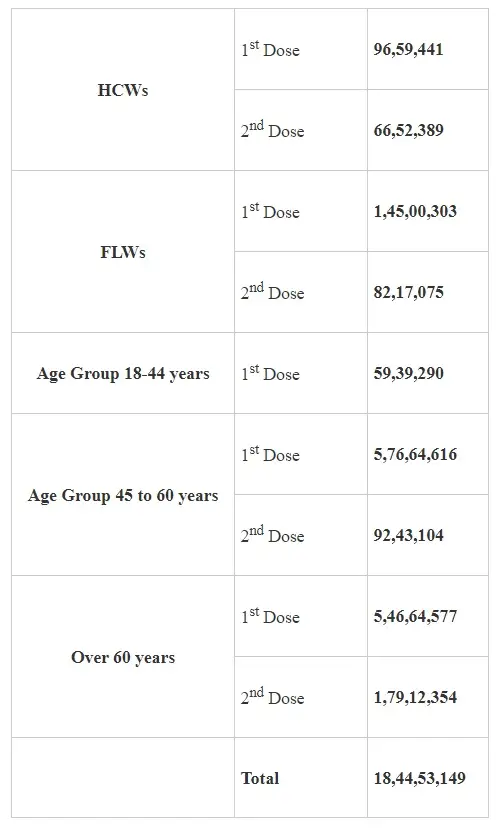
The cumulative number of COVID-19 vaccine doses administered in the country has reached nearly 18.44 Cr today under the Phase-3 of the nationwide Vaccination Drive.
A total of 18,44,53,149 vaccine doses have been administered through 26,87,638sessions, as per the provisional report till 7 am today. These include 96,59,441 HCWs who have taken the 1st dose and 66,52,389 HCWs who have taken the 2nddose, 1,45,00,303 FLWs (1stdose), 82,17,075 FLWs (2nddose), 59,39,290 beneficiaries under 18-44 age group (1stdose), 5,76,64,616 (1stdose) and 92,43,104 (2nd dose) beneficiaries aged 45 to 60 years. 5,46,64,577 1stdose beneficiaries and 1,79,12,354 2nd dose beneficiaries of more than 60 years old.
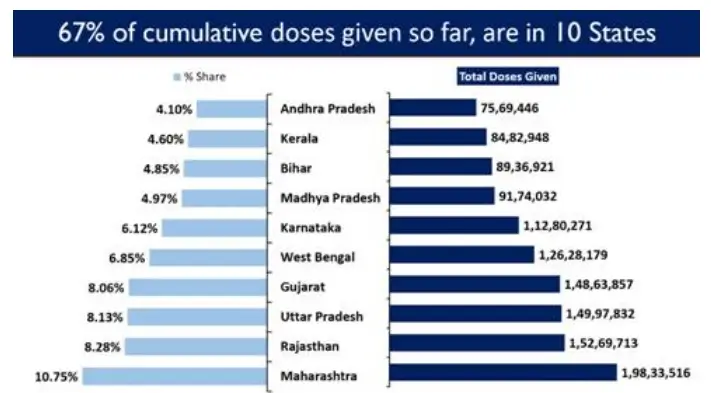
Ten states account for 66.70% of the cumulative doses given so far in the country.
6,69,884 beneficiaries of the age group 18-44 years received their first dose of COVID vaccine in the last 24 hours and cumulatively 59,39,290 across 36 States/UTs since the start of phase-3 of vaccination drive.
As on Day-122 of the vaccination drive (17th May, 2021), 15,10,418 vaccine doses were given. Across 14,447sessions, 12,67,201 beneficiaries were vaccinated for 1st dose and 2,43,217 beneficiaries received their 2nd dose of vaccine.
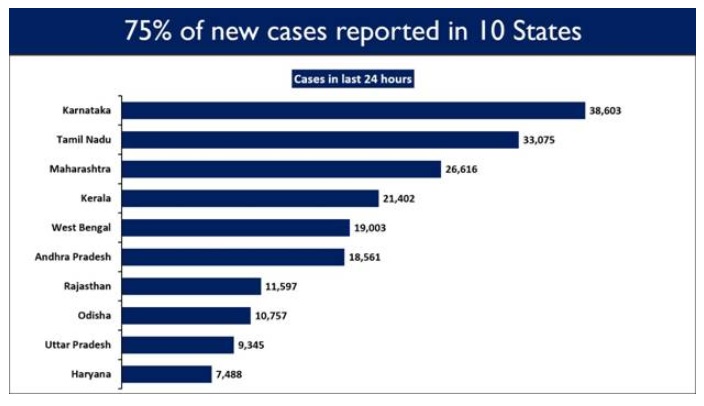
Ten States reported 74.54% of the new cases in last 24 hours. Karnataka has reported the highest daily new cases at 38,603, followed by Tamil Nadu with 33,075 new cases.
The National Mortality Rate currently stands at 1.10%.
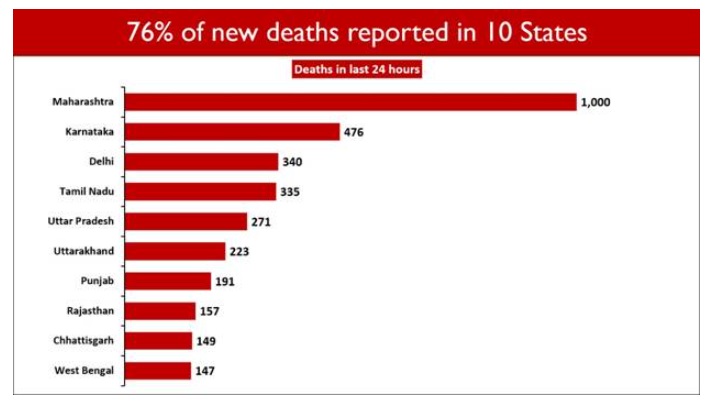
4,329 deaths were reported in the last 24 hours.
Ten States account for 75.98% of the new deaths. Maharashtra saw the maximum casualties (1000). Karnataka follows with 476 daily deaths.
In addition, the foreign aid of relief materials continues to be swiftly cleared, apportioned and sent to States/UTs to combat COVID-19. Cumulatively, 11,321 Oxygen Concentrators; 15,801 Oxygen Cylinders; 19 Oxygen Generation Plants; 7,470 ventilators/Bi PAP; ~5.5 L Remdesivir vials delivered/ dispatched through road and air, so far.
The National Regulator of the country, the Drugs Controller General of India (DCGI), after careful examination, has accepted the recommendation of Subject Expert Committee (SEC) and accorded permission to conduct the Phase II/III clinical trial of Covaxin (COVID vaccine) in the age group 2 to 18 years, to its manufacturer Bharat Biotech Ltd on 12.05.2021.
M/s Bharat Biotech International Ltd., Hyderabad (BBIL) had proposed to carry out a Phase- II/III clinical trial of Covaxin in the age group of 2 to 18 years. The trial will be conducted in 525 healthy volunteers.
In the trial, the vaccine will be given by intramuscular route in two doses at day 0 and day 28.
As rapid regulatory response, the proposal was deliberated in Subject Expert Committee (SEC) (COVID-19) on 11.05.2021. The Committee after detailed deliberation recommended for grant of permission to conduct proposed Phase II/III clinical trial to certain conditions.
There have been reports in a section of the media followed by some uninformed tweets alleging delay in licensing for Covaxin and delayed approval for technology transfer for manufacturing COVAXIN vaccine in the country.
These news reports and the content mentioned in the tweet are completely baseless and factually incorrect.
The Union Government is consistently and proactively working to secure and enhance the availability of vaccines. Government of India, in its new liberalized strategy, has made specific provisions that vaccines for COVID-19, which have been developed & are being manufactured in foreign countries and which have been granted emergency use authorization by National Regulators of United States, European Medicine Agency (EU), United kingdom, Japan or which are listed in WHO (Emergency Use Listing), will be granted emergency use approval in India. It also provides for post-approval parallel bridging clinical trial in place of conduct of prior local clinical trial as per the provisions prescribed under Second Schedule of the New Drugs & Clinical Trials Rules 2019. This is a radical departure from past allowing rapid and simplified authorization of foreign vaccines by Drugs Controller General of India (DCGI).
This will ease and facilitate the import of Covid-19 vaccines and ensure increased availability of Covid-19 vaccines in India.
The New “Liberalized Pricing and Accelerated National Covid-19 Vaccination Strategy” aims at liberalized vaccine pricing and scaling up of vaccine coverage to incentivize vaccine manufacturers to rapidly augment their production and to attract new vaccine manufacturers. It would make pricing, procurement and administration of vaccines more flexible and ensure increased vaccine production as well as wider availability of vaccines in the country.
Government of India, as part of its policy to augment the domestic production of Covid-19 vaccine, has proactively encouraged Public Sector Undertakings (PSUs) as well as private companies to enter into technology transfer agreements with Indian vaccine manufacturers. Two Central Government PSUs, namely, Indian Immunologicals Ltd (IIL) and BIBCOL have entered into a technology transfer agreement with Bharat Biotech. In addition, one State Government Undertaking, namely, Haffkine Institute, has also entered into a technology transfer agreement with Bharat Biotech. All these technology transfer agreements have been actively promoted and assisted by Government of India. The Union Government has also extended substantial financial assistance to all the above 3 Undertakings. As a result of this proactive intervention of the Central Government, Indian Immunologicals Limited will be in a position to start production of Covaxin from September 2021, while Haffkine Institute and BIBCOL will start production of Covaxin from November 2021.
Government of India presently is also engaged in proactive dialogue with Bharat Biotech and some other PSUs, as well as private companies to execute technology transfer agreements. This would further enhance and augment the production of Covaxin in the country.
Under the new policy, 100 percent doses of imported and ready to use foreign vaccine will be available for other than Government of India channel comprising State Governments, private hospitals and hospitals of industrial establishments. The new Liberalized Pricing and Accelerated National Covid-19 Vaccination Strategy also Incentivizes in terms of prices to attract private manufacturers including offshore vaccine manufacturers to enter the country.
Government of India is proactively engaging with foreign vaccine manufacturers like Moderna, Pfizer etc., to apply for Emergency Use Authorization in India so that these vaccines can be easily imported and made available in India.
Simultaneously, Government of India along with other like-minded countries is also stressing on IPR waiver for the Covid-19 vaccines. These two interventions, taken in conjunction, will ensure easy availability of Covid -19 vaccines not only in India but globally too.
