
LSWLifeScienceWorld lost one of its initial Advisory Board Members and a well-wisher on the 6th December, 2022. Dr. S G A Rao, was our guide and always gave valuable suggestions on the editorial front and contributed articles in the stem cell area which was his specialisation.
He conceived the entire program of our interactive meeting titled ‘Holistic Approach to Cancer-An Integrated Therapy” in Association with the Ministry of Ayush, that was held on February 6, 2016, in Mumbai.
Dr. S G A Rao was an internationally renowned scientist in stem cell biology, and was former Head of Division of Chemotherapy and Stem cell Biology, Cancer Research Institute, Tata Memorial Centre, Mumbai.
He was Founder Chairman, International Stem cell Services Ltd, Bengaluru, Founder-Society for Regenerative Medicine & Tissue Engineering and Managing Trustee, Stem Foundation.
Reaffirms our commitment to transforming care delivery in India, by improving access to high-quality healthcare for all
Conceptualized, designed, and manufactured in India for the world, Cios Fit is a mobile C-arm designed for demanding multidisciplinary environments in busy Operating Rooms

Siemens Healthineers achieved yet another milestone in its journey of transforming care delivery in India. The future-ready medical imaging manufacturing facility at Bengaluru produced 500 units of Cios Fit, a multidisciplinary mobile C-arm, conceptualized, designed, and manufactured in India and exported to over 47 countries across the globe.
Cios Fit, is designed for the tough environment in an OR, where multiple procedures are performed in a day, and keeping in mind the high patient load at the medical institutions. Since the start of manufacturing in 2019, Cios Fit has been helping healthcare providers deliver high-value care for their patients. Despite the challenges imposed by the pandemic over the past two years, agile processes and dedicated efforts of the team ensured reaching this benchmark.

Speaking on this milestone, Vivek Kanade, Managing Director, Siemens Healthcare Private Limited said, “This is indeed an encouraging moment for our entire team. Their dedication ensured we met our commitments despite turbulent times. Entrusted to transforming care delivery in India, Cios Fit truly resonates with our purpose. We pioneer breakthroughs in healthcare. For Everyone. Everywhere.”
Siemens Healthineers recently inaugurated the new production line at the Bengaluru facility to manufacture Computed Tomography Scanners under the PLI scheme and launched MAGNETOM Free.Star, a disruptively simple approach to MRI that transforms access to high-value care.
Pharmaceuticals & Medical Devices Bureau of India (PMBI), under the aegis of Department of Pharmaceuticals is going to celebrate their 4th Jan Aushadhi Diwas by organizing weeklong celebrations at different locations across the country covering all States/UTs. This will generate awareness about the usages of generic medicines and benefits of Jan Aushadhi Pariyojana.
Various events during the week long celebrations will be conducted with closed coordination of PMBJKs owners, Beneficiaries, State/UT officials, Public Representatives, Doctors, Health Workers, Nurses, Pharmacists, Jan Aushadhi Mitra’s and other stakeholders to discuss about the Scheme, its salient features and achievements. All the activities will be organized within the spirit of “Azadi Ka Amrit Mahotsav” and several events have been planned at 75 locations.The events have been planned in different cities starting from 1st of March 2022 to 7th March 2022, which will focus on awareness about the Jan Aushadhi Scheme, Seminars, participation of children, women and NGO’s, Heritage walks and Health walks and many other activities.Day wise activities planned to be held across the country are as under:
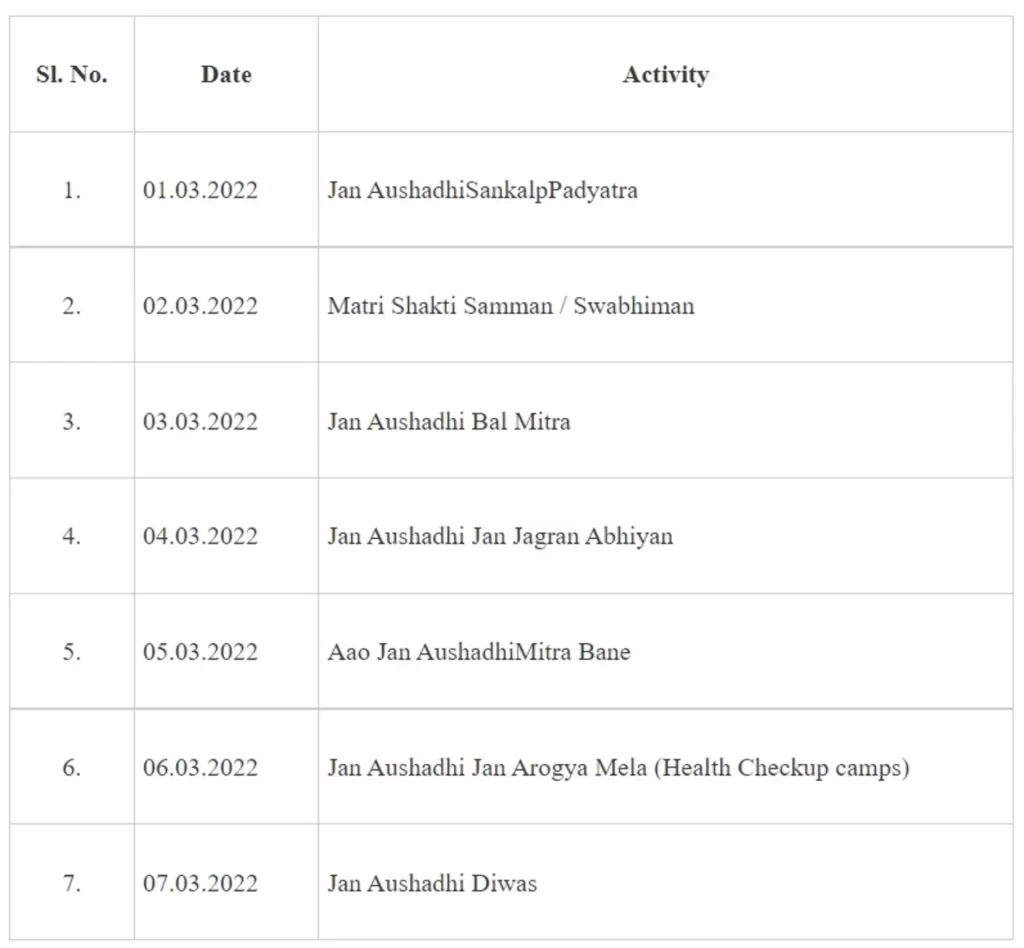
The main event of “Jan Aushadhi Diwas” will be held on 7th March 2022 (Monday) at Vigyan Bhawan, New Delhi. Union Minister of Health & Family Welfare and Chemicals & Fertilizers, Dr. Mansukh Mandaviya and Minister of State for Chemicals & Fertilizers, Shri Bhagwant Khuba will grace the event.
As on 31st January, 2022, the number of stores has increased to 8,675. Under the PMBJP, all 739 districts of the nation have been covered. The scheme ensures easy reach of affordable medicine to the people in every nook and corner of the country. The Government has set a target to increase the number of Pradhan Mantri Bhartiya Janaushadhi Kendras (PMBJKs) to 10,500 by the end of March 2025. Product basket of PMBJP comprises of 1451 drugs and 240 surgical instruments. Further, new medicines and nutraceuticals products like protein powder, malt-based food supplements, protein bar, immunity bar, sanitizer, masks, glucometer, oximeter, etc. have been launched. At present three IT enabled warehouses of PMBJP are functional at Gurugram, Chennai & Guwahati and fourth one is ready to start operations at Surat. Further, 39 distributors have been appointed across the country to support the supply of medicines to remote and rural areas.
Prime Minister, Shri Narendra Modi inaugurated the post Union Budget webinar of Ministry of Health and Family Welfare. He highlighted India’s strength in universal primary healthcare, harnessing technology for providing equitable healthcare, need for vibrant Public Private partnerships for innovative healthcare solutions and integrating healthcare providers and consumers through smart platforms for providing easily accessible, affordable quality healthcare to all. The valedictory address was delivered by Dr Mansukh Mandaviya, Union Minister of Health & Family Welfare in presence of Shri Sarbananda Sonowal, AYUSH minister and Dr Bharati Pravin Pawar, MOS (HFW), Dr V.K. Paul (NITI Aayog), and Dr MahendraMunjapara, MOS (AYUSH).
This is fifth webinar in a series of post budget webinars addressed by Prime Minister. Union Ministers, healthcare professionals from public and private sectors, and professionals from para-medics, nursing, health management, technology and research participated in the webinar.The objective of the webinar was to involve the stakeholders in taking forward the various initiatives of the Government in the health sector.
In his valedictory address, Union Health Minister noted that vision of the Prime Minister for a holistic healthcare has guided the integration of various healthcare platforms. “Today’s brainstorming sessions with all stakeholders will help to create a well defined blueprint for timely action for citizen-centric policies and output-based programmes”, he stated.The consultation with private sector, non-government organizations including experts and health sector stakeholders through deep-discussions is a step in enriching the policies, initiatives and actions of the Ministry, he stated. “India is strongly poised to implement the framework of “Total Health and One Health” through its focused, integrative and holistic policies”, he added. Ayurveda and Mental Health form a vital part of Total Health and should be extended through telemedicine services.
With the new approach to early Union Budget presentation, the funds allocation and programme implementation can now be started in a much timely manner leading to timely spending of allocated funds don't lapse due to untimely planning, he stressed.Reforms in timely manner have ensured that India is not left behind on the global platform. “Our strategic policies of vaccine research and manufacturing are in sync with the emerging developments globally”, he noted.
Highlighting the role of technology and Research and Development, Dr. Mansukh Madaviya said that today India is at par with global leaders in vaccine research due to focus on indigenization.He added that telemedicine and Ayushman Bharat Digital Mission will rightly propel India towards a revolution in the health care sector.Teleconsultation is a revolution and it is providing expert health care services even in the remotest of the areas.Today, India is the leading country in the world to have created more than 17 Crore Health IDs in India, highest globally, Dr Mandaviya stated.
The webinar had breakout sessions on the following three themes:
Shri Rajesh Bhushan, Union Health Secretary underlined the fact that ABDM should not be seen as a standalone digital platform but an enabler that will create an ecosystem which will link multifarious existing digital platforms, with the aim to provide quality healthcare services.
The participants highlighted the importance of integrated, quality, easily accessible healthcare solutions through digital healthcare. ABDM was found important for empowering health consumers by giving them control over healthcare choices and making healthcare more equitable. Some speakers termed ABDM as a "game-changer". Its importance in setting minimum standards for a largely fragmented healthcare landscape in the country was found paramount as it will reduce the volatility of service providers and services.
The eminent speakers also highlighted that ABDM has the potential to reshape India's healthcare in the post-pandemic era. It can expand the provision of universal healthcare through purposive and strategic linked partnerships between diagnostic labs, pharmacists, hospitals, etc., across the public-private divide. It was noted that ABDM can also encourage innovation and research through smart start-ups and healthcare incubators. With digital platforms connecting multifarious partners, our reach in global healthcare providers and healthcare consumers will substantially increase, giving impetus to medical value tourism.Some speakers also highlighted the importance of instant gratification for the consumer to see all medical and healthcare records through automated document aggregation on one platform.
Dr R S Sharma, CEO NHA allayed fears and apprehensions regardingprivacy and safety of healthcare data stating that consumer data and identity are protected through clearly defined protocols. Terming ABDM as a "win-win" platform for both healthcare providers and consumers, he highlighted the feature of Unified Health Interface for linking healthcare solutions of various kinds of systems through well-defined APIs which accommodate various languages.
A presentation on leveraging the technology of eSanjeevani towards ensuring the goal of “accessible, affordable and equitable healthcare for all” was made by Shri Lav Agarwal, JS, Health Ministry, wherein stakeholders were apprised of the ways and means of augmenting eSanjeevani as the National Telemedicine Platform. He briefed the participants that 2.68 crore consultations have been recorded so far in the eSanjeevani platform and it is a global achievement when compared with the other countries.
The participants highlighted the importance of telemedicine and eSanjeevani initiative of the government in providing affordable, equitable and accessible health care to all. It was noted that e-Sanjeevani has brought in a revolution in the health sector and is rapidly bridging the digital divide in the health sector as envisioned by the Prime Minister. The speakers appreciated how eSanjeevani has led to massive improvement in access to specialised health services, particularly in the rural and remote areas. This service has been a boon for the patients in urban areas as well, especially during the second surge of the ongoing Covid-19 pandemic that burdened the healthcare services delivery system in the country. Some speakers highlighted that Covid-19 has given a huge impetus to the adoption of telemedicine in the country, thereby increasing the footprint of digital health services. A consensus was built that innovative use of technology in the health sector has a huge potential to transform the health care paradigm and eSanjeevani will propel the growth of the sector.
Some speakers noted that eSanjeevani can be integrated with other associated specialties to increase access and availability in the community. Its services can be extended to vulnerable sections of the population like inmates in jail, old age homes. Innovative implementation solutions for e-ICU and remote healthcare monitoring are also envisaged through the platform. The platform can also act as a tele-mentoring platform for doctors for CMEs and for paramedics and other staff through a content management system (CMS). Experts suggested that eSanjeevani can be the gateway for all digital platforms, with integration of cutting-edge technology, AI-enabled chatbots. It can support tele-follow-ups and referrals along with support for veterinary services. Experts also discussed that solutions need to be explored for connectivity issues in remote areas though innovative Public Private Partnership (PPP) models to ensure quality service delivery to our citizens. Taking a holistic approach, various platforms of other ministries like Labour and Railways can also be integrated. It was emphasized that the strategies must be explored on sharing our solutions with other countries.
The eminent speakers also spoke about the various individual initiatives taken by them to promote teleconsultation through their networks and how they look forward to collaborating and working in coordination with the government on the eSanjeevani platform to provide universal health care. To expand the reach and facilities available on eSanjeevani platform introducing tele-ICU, Tele-emergencies, Ayurveda and Tele-Operations on the eSanjeevani app were discussed. The session participants agreed that if the vision of One India, One Health is to be realised then Telemedicine is definitely the way forward.
Shri Vikas Sheel, AS&MD (NHM) made the opening presentation. The speakers welcomed the initiative and opined that the proposed intervention is a timely step especially in view of recent pandemic experience.This prompt and timely initiative indicates proactive governance.
It was pointed out that the proposed Tele-Mental Health Programme needs to cover all aspects of mental health care across the full continuum of care (primary, secondary & tertiary), bringing all the stakeholders and service providers on the same platform, including private sector and AYUSH providers. The speakers also highlighted that the model for Tele-Mental health ought to bebe citizen centric which could enable the citizens with specific and special needs to access appropriate services.Ownership and participation of state governments would be essential for implementation of the initiative.
It was also discussed that e-learning and training initiatives should be an integral part of the Programme, given the severe shortage of psychiatric professionals in the country.
Development of capacities of the Front-Line Health Workers such as Medical Officer, Community Health Officers, ANMs and ASHAs for early identification of symptoms of mental health issues and once identified, for providing mental health care services at the Primary Health Care level, would be critical. This would also address the barriers to access to digital consultations through facilitated consultations.
The speakers opined that all efforts need be made to integrate both the AYUSH service providers and the AYUSH practices in the range of mental healthcare services envisaged to be delivered. Suitable provisions should be made in the National Mental Health Programme. In addition, existing Telemedicine Guidelines should be aligned with the provisions of the Mental Health Care Act 2017. Suitable amendments may also be carried out in the Rules made under the Act to remove any inconsistencies and legal challenges. It was pointed out that the strengths of Ayushman Bharat Digital Mission (ABDM) and eSanjeevani platform should be leveraged.
Shri Rajesh Bhushan, Secretary, MoHFW, Shri Vaidya Rajesh Kotecha, Secretary, AYUSH, Dr R S Sharma, CEO,Shri Vikas Sheel, AS & MD, NHM, MoHFW, Shri Lav Agarwal, Joint Secretary, MoHFW, NHA, Dr Praveen Gedam, Additional CEO, NHA, Dr Pratima Murthy, Director, NIMHANS, Prof BN Gangadhar, Padmashree awardee and former Director of NIMHANS, Professor K. Srinath Reddy, President, PHFI, Dr Mohan Isaac, Clinical Prof. of Psychiatry, The University of Western Australia, Dr Anant Bhan, Researcher in Global Health, Sangath-USA/Goa, Dr Preeti Kumar, Vice President, PHFI and other senior officials of Ministry of Health and Family Welfare, AYUSH Ministry, NIMHANS, NHM and public-private organisations were present in the meeting.
Dr. Mansukh Mandavia,Union Minister for Health and Family Welfare launched the National Polio Immunization Drive for 2022 by administering polio drops to children below five years of age in the Ministry of Health and Family Welfare today.
Addressing the Occasion, Dr. Mansukh Mandaviya said, “India’s strategic fight against polio is a success story of India’s public health policy against vaccine preventable diseases. We need to continue to be vigilant and ensure that every child under 5 years of age must get polio drops.”

“Under the leadership of our Hon’ble Prime Minister, Universal Immunization Programme is focusing to protect children from more diseases than ever before and has introduced several new vaccines like Pneumococcal Conjugate Vaccine (PCV), Rotavirus vaccine, and Measles-Rubella vaccine (MR) in the recent past. Further, to provide additional protection to our children, the Government of India has also introduced the injectable Inactivated Polio Vaccine into its routine immunization program. While we are making efforts to protect our children from more and more diseases, it is important that all vaccines under the programme reach every child of our country.”, Dr. Mandaviya further added.
Stressing on the importance of observing National Immunisation Day, the Union Health Minister HFM said, “The aim of Swastha Bharath can only be achieved if our children are healthy. The objective of Mission Indradhanush or Polio Vaccination Drive is to protect our children against such deadly diseases. Since our neighbouring countries are still not polio free, we should remain vigil and continue the vaccination program. Over 15 crore children of age less than 5 years will be vaccinated in the coming months. Door-to-door Vaccination campaigns through robust micro-planning will be done to ensure that no one is left. I congratulate all the Health Care Workers, Stakeholders like WHO, UNICEF, Rotary club and NGOs for making this immunization programme a Lok Bhagidari Andolan as envisioned by our Hon’ble Prime Minister. I request all families to come forward to get their children Vaccinated.”
The Union Health Secretary highlighted the achievements and the way forward to implement the National Immunisation Programme. He said, “Though India is polio free, it is still our responsibility to remain vigil. Transit teams deployed at all the states to ensure immunization. And, continuous pulse polio implementation at borders are happening as our neighbouring countries are still reporting Polio Cases.”
The Polio National Immunization Day 2022 (NID) will be organized across the country on Sunday, 27th February 2022. India conducts one nationwide NID and two Sub-National Immunization Day (SNIDs) for polio every year to maintain population immunity against wild poliovirus and to sustain its polio free status. During the Polio NID, Over 15 crore children will be covered across all 36 States and UTs in 735 districts. During the drive, polio drops will be provided to children through 7 lakh booths across the country. Approximately 23.6 crore houses will be visited by nearly 24 lakh volunteers and 1.5 lakh supervisors. The State of Meghalaya has already conducted the drive in the state on 24th January 2022 while Mizoram plans to conduct the drive on 1st March 2022 due to local reasons. The election bound states of Uttar Pradesh and Manipur plan to organize the Polio NID on 20th March and 24th March 2022 respectively.
The booth activities will be followed by house-to-house surveillance (mop-up rounds) over the next two to five days to identify and vaccinate children who missed getting vaccinated at the booths. Vaccination teams have also been deployed at bus terminals, railway stations, airports and ferry crossings to vaccinate children in transit to ensure no child misses the life-saving dose.
India has been free of polio for more than a decade, with the last case of wild poliovirus reported on 13th January 2011. However, India continues to remain vigilant to prevent re-entry of the poliovirus into the country from neighboring countries of Afghanistan and Pakistan, where wild poliovirus continues to cause the disease.
It is also ensured that all measures for safety during pandemic are observed by following all COVID Appropriate Behaviors (CAB) such as preventing overcrowding at booths, maintaining physical distance, wearing masks, washing hands and administering polio drops in well ventilated-settings.
The meeting was attended by Shri Vikas Sheel, AS & MD, Ministry of Health and Family Welfare, Shri Ashok Babu, Joint Secretary, Ministry of Health and Family Welfare, Dr Roderico H. Ofrin, WHO Representative to India and other senior officials of the Ministry of Health and Family Welfare.
FOR IMMEDIATE RELEASE (NORTH BETHESDA, MARYLAND USA, 27 January 2022) -- The International Society for Pharmaceutical Engineering (ISPE) announced Matthew Hepburn, MD, Senior Advisor to the Director, Pandemic Prevention, U.S. Office of Science and Technology Policy (OSTP), Executive Office of the President, as a confirmed keynote for the 2022 ISPE Facilities of the Future Conference, taking place on 1–2 February in North Bethesda, Maryland and virtually.
The opening keynote session COVID's Impact on Pharma Facilities of the Future will take a high-level look ahead at global pharmaceutical manufacturing and related regulatory systems. Hepburn will discuss Lessons Learned from Operation Warp Speed for Rapid Vaccine Development at the opening plenary session and the executive forum dinner.
The mission of OSTP is “to maximize the benefits of science and technology to advance health, prosperity, security, environmental quality, and justice for all Americans.” At the OSTP, Hepburn works on preparing the country for future pandemics, with a spotlight on the acceleration of vaccines, therapies, and test development.
Previously, Hepburn was the Director of Vaccine Development at Countermeasures Acceleration Group, formerly known as Operation Warp Speed, which is an effort between the Defense Department and the Department of Health and Human Services. During the Obama administration, he was the Director of Medical Preparedness for the White House National Security Council. Additionally, he served as an infectious disease doctor for the U.S. Army for over two decades.
Representatives from Bristol-Myers Squibb, CRB, and Merck will round out a compelling line-up of expert speakers at the opening plenary session. Featuring technical presentations from regulatory authorities and industry leaders already planning and building “facilities of the future,” the 2022 ISPE Facilities of the Future Conference will explore topics such as patient-specific cell and gene therapy facilities, new developments in the use of artificial intelligence, innovations in treatment and the transforming technologies that produce them, and moving forward from the COVID-19 era.
To ensure this experience is accessible to all, this conference will be in North Bethesda, Maryland, with virtual components—delivering thought-provoking learning and global networking opportunities whether attendees choose to join us in-person or virtually. Explore the agenda and register at ISPE.org/FOF22.
The International Society for Pharmaceutical Engineering (ISPE) is a not-for-profit association serving its Members through leading scientific, technical, and regulatory advancement across the entire pharmaceutical lifecycle. The 18,000 Members of ISPE are building solutions in the development and manufacture of safe, effective pharmaceutical and biologic medicines, and medical delivery devices in more than 90 countries around the world. Founded in 1980, ISPE has its worldwide headquarters and training center in North Bethesda, Maryland, USA, and its operations center in Tampa, Florida, USA. Visit ISPE.org for more information.
Tecentriq is the first and only cancer immunotherapy approved for NSCLC in the adjuvant setting
Approval is based on the Phase III IMpower010 study showing adjuvant Tecentriq improved disease-free survival by more than one-third in PD-L1-positive Stage II-IIIA lung cancer, compared with best supportive care
Basel, 15 October 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the US Food and Drug Administration (FDA) has approved Tecentriq® (atezolizumab) as adjuvant treatment, following surgery and platinum-based chemotherapy, for adults with Stage II-IIIA non-small cell lung cancer (NSCLC) whose tumours express PD-L1≥1%, as determined by an FDA-approved test.
“Tecentriq is now the first and only cancer immunotherapy available for adjuvant treatment of NSCLC, introducing a new era where people diagnosed with early lung cancer may have the opportunity to receive immunotherapy to increase their chances for cure,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Today’s landmark approval gives physicians and patients a new way to treat early lung cancer that has the potential to significantly reduce risk of cancer recurrence, after more than a decade with limited treatment advances in this setting.”
“Too many patients with early-stage lung cancer experience disease recurrence following surgery. Now, the availability of immunotherapy following surgery and chemotherapy offers many patients new hope and a powerful new tool to reduce their risk of cancer relapse,” said Bonnie Addario, Co-founder and Chair, GO2 Foundation for Lung Cancer. “With this approval, it is more important than ever to screen for lung cancer early and test for PD-L1 at diagnosis to help bring this advance to the people who can benefit.”
The approval is based on results from an interim analysis of the Phase III IMpower010 study. The results showed treatment with Tecentriq, following surgery and platinum-based chemotherapy, reduced the risk of disease recurrence or death by 34% (hazard ratio [HR]=0.66, 95% CI: 0.50-0.88) in people with Stage II-IIIA NSCLC (UICC/AJCC 7th edition) whose tumours express PD-L1≥1%, compared with best supportive care (BSC). Safety data for Tecentriq were consistent with its known safety profile and no new safety signals were identified. Fatal and serious adverse reactions occurred in 1.8% and 18%, respectively, of patients receiving Tecentriq. The most frequent serious adverse reactions (>1%) were pneumonia (1.8%), pneumonitis (1.6%), and pyrexia (1.2%).
The review of this application was conducted under the FDA’s Project Orbis initiative, which provides a framework for concurrent submission and review of oncology medicines among international partners. According to the FDA, collaboration among international regulators may allow people with cancer to receive earlier access to products in other countries where there may be significant delays in regulatory submissions. Simultaneous applications were submitted to regulators in Switzerland, the UK, Canada, Brazil and Australia under Project Orbis. Additionally, the FDA reviewed and approved the application under its Real-Time Oncology Review pilot programme, which aims to explore a more efficient review process to ensure safe and effective treatments are available to patients as early as possible. The IMpower010 data have also been submitted as the basis of marketing applications to the European Medicines Agency (EMA) and other global health authorities.
Tecentriq has previously shown clinically meaningful benefit in various types of lung cancer, with six currently approved indications in the US. In addition to becoming the first approved cancer immunotherapy for adjuvant NSCLC, Tecentriq was also the first approved cancer immunotherapy for front-line treatment of adults with extensive-stage small cell lung cancer (SCLC) in combination with carboplatin and etoposide (chemotherapy). Tecentriq also has four approved indications in advanced NSCLC as either a single agent or in combination with targeted therapies and/or chemotherapies. Tecentriq is available in three dosing options, providing the flexibility to choose administration every two, three or four weeks.
Roche has an extensive development programme for Tecentriq, including multiple ongoing and planned Phase III studies across different settings in lung, genitourinary, skin, breast, gastrointestinal, gynaecological, and head and neck cancers. This includes studies evaluating Tecentriq both alone and in combination with other medicines, as well as studies in metastatic, adjuvant and neoadjuvant settings across various tumour types.
IMpower010 is a Phase III, global, multicentre, open-label, randomised study evaluating the efficacy and safety of Tecentriq compared with BSC, in participants with Stage IB-IIIA NSCLC (UICC/AJCC 7th edition), following surgical resection and up to 4 cycles of adjuvant cisplatin-based chemotherapy. The study randomised 1,005 people with a ratio of 1:1 to receive either Tecentriq (up to 16 cycles) or BSC. The primary endpoint is investigator-determined DFS in the PD-L1-positive Stage II-IIIA, all randomised Stage II-IIIA and intention-to-treat (ITT) Stage IB-IIIA populations. Key secondary endpoints include overall survival (OS) in the overall study population, ITT Stage IB-IIIA NSCLC.
Lung cancer is one of the leading causes of cancer death globally.1 Each year 1.8 million people die as a result of the disease; this translates into more than 4,900 deaths worldwide every day.1 Lung cancer can be broadly divided into two major types: NSCLC and SCLC. NSCLC is the most prevalent type, accounting for around 85% of all cases.2 Approximately 50% of patients with NSCLC are diagnosed with early-stage (Stages I and II) or locally advanced (Stage III) disease.3 Today, about half of all people with early lung cancer still experience a cancer recurrence following surgery.4 Treating lung cancer early, before it has spread, may help prevent the disease from returning and provide people with the best opportunity for a cure.
Tecentriq is a monoclonal antibody designed to bind with a protein called Programmed Death Ligand-1 (PD-L1), which is expressed on tumour cells and tumour-infiltrating immune cells, blocking its interactions with both PD-1 and B7.1 receptors. By inhibiting PD-L1, Tecentriq may enable the activation of T-cells. Tecentriq is a cancer immunotherapy that has the potential to be used as a foundational combination partner with other immunotherapies, targeted medicines and various chemotherapies across a broad range of cancers. The development of Tecentriq and its clinical programme is based on our greater understanding of how the immune system interacts with tumours and how harnessing a person’s immune system combats cancer more effectively.
Tecentriq has shown clinically meaningful benefit in advanced NSCLC and SCLC, with five currently approved indications in the EU. Tecentriq is approved in the US, EU and countries around the world, either alone or in combination with targeted therapies and/or chemotherapies in various forms of NSCLC, SCLC, certain types of metastatic urothelial cancer, in PD-L1-positive metastatic triple-negative breast cancer and for hepatocellular carcinoma. In the US, Tecentriq is also approved in combination with Cotellic® (cobimetinib) and Zelboraf® (vemurafenib) for the treatment of people with BRAF V600 mutation-positive advanced melanoma.
Roche’s rigorous pursuit of groundbreaking science has contributed to major therapeutic and diagnostic advances in oncology over the last 50 years, and today, realising the full potential of cancer immunotherapy is a major area of focus. With over 20 molecules in development, Roche is investigating the potential benefits of immunotherapy alone, and in combination with chemotherapy, targeted therapies or other immunotherapies with the goal of providing each person with a treatment tailored to harness their own unique immune system to attack their cancer. Our scientific expertise, coupled with innovative pipeline and extensive partnerships, gives us the confidence to continue pursuing the vision of finding a cure for cancer by ensuring the right treatment for the right patient at the right time.
In addition to Roche’s approved PD-L1 checkpoint inhibitor, Tecentriq® (atezolizumab), Roche’s broad cancer immunotherapy pipeline includes other checkpoint inhibitors, such as tiragolumab, a novel cancer immunotherapy designed to bind to TIGIT, individualised neoantigen therapies and T-cell bispecific antibodies.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Transforming global food systems to become more inclusive, fair and sustainable may seem an insurmountable challenge, yet there are concrete actions policymakers can take, according to a new report released by the UN’s International Fund for Agricultural Development (IFAD) today.
“We are living in a world of huge and unfair contradictions. There are 800 million hungry people and yet high obesity rates. Nutritious diets are expensive yet many small-scale farmers are poor. Current food growing practices are not good for our environment. It is clear that we need a revolution. A revolution so dramatic that previous versions of food systems are unrecognizable,” said Dr. Jyotsna Puri, Associate Vice President of IFAD’s Strategy and Knowledge Department that leads the production of the Rural Development Report, IFAD’s flagship publication.
Puri sees this week’s UN Food Systems Summit as a watershed moment to commit to real change, with the Rural Development Report offering governments recommendations for concrete actions that can be taken.
The report, Transforming food systems for rural prosperity, stresses the importance of focusing investments and policy changes on rural food value chains so that all people can access adequate nutritious food in a manner that does not harm the environment, and so that food producers can earn decent incomes.
The majority of people in rural areas earn an income from working in small-scale agriculture, which is a vital source of national and global food. In fact, farms of up to 2 hectares produce 31 percent of the world’s food on less than 11 percent of the farmland.
The key recommendations of the report include:
Invest more in rural farms and local small and medium-sized enterprises (SMEs) that support activities after the farm gate, such as storing, processing, marketing and food distribution. A focus on local ownership and employment will increase job opportunities, particularly for women and young people, while giving small-scale farmers access to new and diverse markets.
Make available innovations (such as nature based solutions and agro-ecology) and affordable digital technologies to boost rural small-scale famers’ production so that farmers can be climate-resilient, using low carbon and sustainable techniques.
Develop and focus on pricing systems that reflect the full and true cost of production, including rewarding farmers for ecosystem services, such as maintaining healthy soil and regulating pests.
Promote accessible and affordable nutritious food. At least 3 billion people cannot currently afford healthy diets. Changing this requires focusing on nutrition education, empowering women to make nutrition decisions, and stronger government policies to regulate and steer market choices. Governments can use market-based instruments, income support and public procurement to focus on nutrition-rich foods.
Engage to rebalance global trade and governance to correct power imbalances. The present concentration of power within food systems calls for rethinking regulations and trade arrangements so that rural people in developing countries can benefit. Food markets need to be accessible to rural people, and on fair terms. Incentives need to be in place to reward nature-based practices and local, healthy diets.
“We know what needs to change to make the production, marketing and consumption of food fair and sustainable, which results in nutritious, affordable food for all. This report gives strong evidence and recommendations for specific actions. Now we need the investments and political will to take action,” said Puri.
Over the past 70 years, a focus on industrial farming and producing more calories at low cost has been accompanied by growing malnutrition, increased food waste, and a high environmental cost. Food systems are responsible for 37 percent of greenhouse gas emissions, and are also highly vulnerable to a changing climate.
The Food Systems Summit on 23 September, under the leadership of Secretary-General António Guterres, is intended to result in actionable commitments from heads of state and other leaders to transform global food systems. It is a culmination of 18 months of engagement with governments, food producers, civil society and companies on how to transform the way we produce, process and consume food.
India with the proactive leadership of Honourable Prime Minister Ji and dedicated altruistic services of Modern Medical fraternity has literally just walking out of the disastrous second wave of Covid pandemic. With the global evidence available and the history of any pandemics the third wave is inevitable and imminent. But the past experience of last one and half years of war with the virus and based on the emerging evidences it is obvious with making the universal vaccination reach maximum possible population and strictly adopting to Covid appropriate behaviours we can face the third wave with confidence and mitigate its impact. However, it is painful to note in this crucial time every one need to work for the mitigation of third wave, in many parts of the country both government and public are complacent and engaged in mass gatherings without following Covid protocols. Tourist bonanza, pilgrimage travel, religious fervour all are needed, but can wait for few more months. Opening up these rituals and enabling people without vaccination to go scot free in these mass gatherings are potential super spreaders for the Covid third wave. The consequences of treating a patient with covid in hospital and its impacts on the economy will be much better than the economic loss we suffer with avoiding such mass gathering .It is the duty and responsibility of every one at this moment to strictly enforce the covid appropriate behaviours for minimum three more months and ensure every one nearer to our house are getting vaccinated .IMA appeal to all state Governments to translate the vision of Honourable Prime Minister Ji and control any mass gatherings in their state.
In the recent days, some States and UTs have reported an increasing number of patients suffering from Mucormycosis, popularly known as Black Fungus. Taking a note of the secondary and opportunistic fungal infections which are getting amplified due to the present surge in COVID cases and Mucormycosis becoming a cause of concern, Union Ministry of Health has advised all States/UTs to review their preparedness for infection prevention and control, as well as hygiene and sanitation in hospitals.
Union Health Secretary in his letter to Chief Secretaries and Administrators of all States and UTs has urged them to undertake the following activities/practices to ensure that there are robust infection prevention and control practices in COVID hospitals and other healthcare facilities:
Establish/activate the Hospital Infection Control Committee with the head of the institution or an administrator as the chairperson.
Designate an infection prevention and control nodal officer – preferably a microbiologist or senior infection control nurse.
Prepare and implement the Infection Prevention Control (IPC) Programme in the hospital/health facilities, as per the guidance given in National Guidelines for Infection and Control in Healthcare Facilities (available at https://www.mohfw.gov.in/pdf/National%20Guidelines%20for%20IPC%20in%20HCF%20-%20final(1).pdf). This involves the following key components of:
Infection prevention and control manual
Guidelines on antimicrobial use and management
Educational programmes and strategies
Risk assessment and risk management
Planning, monitoring, audit and feedback
Implementation strategies
Emphasise and strengthen procedures and practices for IPC. In the context of COVID-19,
Standard precautions are to be applied all across the hospital/health facility
Transmission-based precautions need heightened focus on droplet, airborne and contact precautions from the perspective of protecting healthcare workers and ensuring patient safety.
Improve the environment and facilitate:
Ventilation with focus on fresh air and natural ventilation wherever control systems with requisite air changes are not available.
Cleaning, disinfection and sanitation of the hospital environment and frequently touched surfaces, with recommended disinfectants like 1% sodium hypochlorite or 70% alcohol.
Safe water and food to prevent water or food borne diseases in hospital settings
Biomedical waste needs to be managed as per the CPCB guidelines available at https://cpcb.nic.in/uploads/Projects/Bio-Medical-Waste/BMW-GUIDELINES-COVID_1.pdf
Infection Prevention and Control practices needs to be enhanced in Intensive Care Units (ICUs) using a bundle-approach to prevent device associated infections such as ventilator associated pneumonia or catheter-associated blood stream, urinary infections etc.
Infection Prevention and control practise in the clinical laboratories and those attached to hospitals are very crucial for the safety of laboratory/hospital staff and health security of the community.
Meticulous adherence to Infection Prevention and Control while managing immunocompromised patients such as COVID-19 patients on steroid treatment, with co-morbidities (such as diabetes where good glycemic control needs to be established; the guidelines for which are available at https://www.mohfw.gov.in/pdf/ClinicalGuidanceonDiabetesManagementatCOVID19PatientManagementFacility.pdf).
In due course, establish surveillance of healthcare associated infections with focus on ventilator associated pneumonia, catheter-associated blood stream infection, catheter-associated urinary tract infection, surgical site infections, gastro-intestinal outbreaks. Further guidance can be taken from AIIMS HAI network; details available at https://www.haisindia.com Train all hospital staff to develop their skills in IPC, irrespective of their individual routine duties, in implementing procedures and protocols described in the Hospital Infection Control Manual.
A State Nodal Officer needs to be identified to monitor the implementation of infection prevention and control to provide evaluation and feedback of the IPC programme in the state.
States and UTs have been assured that Ministry of Health and Family Welfare will provide all necessary assistance to implement the National Guidelines for Infection Prevention and Control in Healthcare Facilities.
