The 44th edition of the India International Trade Fair (IITF), organised by the India Trade Promotion Organisation (ITPO), concluded today with an Award Presentation Ceremony held at the Musical Fountain Stage, Bharat Mandapam, New Delhi. The IITF 2025 Awards for Excellence in Display were presented by Managing Director, ITPO, Dr. Neeraj Kharwal.
In the Partner State category, Rajasthan received the Gold Medal, Bihar received the Silver Medal and Uttar Pradesh received the Bronze Medal. Maharashtra received a Special Commendation. In the Focus State category, Jharkhand was awarded the Gold Medal.
In the States and Union Territories category, Odisha won the Gold Medal, Madhya Pradesh received the Silver Medal and Puducherry received the Bronze Medal. Commendation Certificates were awarded to Delhi, Goa and Karnataka.
In the Thematic Presentation by States category, Meghalaya received the Gold Medal, Kerala the Silver Medal and Andhra Pradesh the Bronze Medal. Commendation Certificates were presented to Chhattisgarh, Arunachal Pradesh and Tripura.
As part of the Swachh Bharat Abhiyan activities observed during IITF, awards for Swachh Pavilion were presented to Haryana (Gold), Punjab (Silver) and Assam (Bronze). Commendations were awarded to Andaman & Nicobar Islands, Uttarakhand and Tamil Nadu.
In the Foreign Pavilions category, Thailand (Thai SMEs Exporter Association) received the Gold Medal, Iran (Dorna Seyd Makran) received the Silver Medal and Dubai (Al Rawdha General Trading LLC) received the Bronze Medal. Special Commendations were given to Republic of Korea (Es Korea Co. Ltd.), Turkey (Tillo Hediyelik Esya Sanayi) and the Tibetan Chamber of Commerce.
In the Ministries and Government Departments category, the Ministry of Defence received the Gold Medal, the Ministry of Mines received the Silver Medal and the Ministry of Railways received the Bronze Medal. Special Commendations were awarded to the Ministry of Ayush, Ministry of Power and the Ministry of Rural Development (SARAS Ajeevika Mela).
In the Ministries, PSUs and PSBs category, the Airports Authority of India received the Gold Medal, the National e-Governance Division (NeGD) received the Silver Medal and the Food Corporation of India received the Bronze Medal. Special Commendations were awarded to the Ministry of Textiles, State Bank of India and Life Insurance Corporation of India.
In the Commodity Boards category, the Tea Board India received the Gold Medal, the Spice Board India received the Silver Medal and the Coir Board received the Bronze Medal. Special Commendations were presented to the National Turmeric Board, Coconut Development Board and National Jute Board.
In the Public Communication & Outreach category, the Directorate General of Taxpayer Services received the Gold Medal, the Ministry of Health and Family Welfare received the Silver Medal and the Central Board of Direct Taxes received the Bronze Medal. Special Commendations were awarded to the Registrar General & Census Commissioner of India, the Securities and Exchange Board of India and the Ministry of Environment, Forest and Climate Change.
In the Empowering India (Ministries & Government Departments) category, the Ministry of Agriculture and Farmers Welfare received the Gold Medal, the Ministry of MSME received the Silver Medal and NBCC (India) Ltd. received the Bronze Medal. Special Commendations were presented to the Ministry of Statistics and Programme Implementation, Coal India Ltd. and NAFED.
In the Private Sector category, RDM Care (AYUR) received the Gold Medal, Dairy India Pvt. Ltd. (Ananda) received the Silver Medal, and United Ekta Engineering Udyog Pvt. Ltd. and Mittal Electronics (Sujata) received the Bronze Medal. Special Commendations were given to Juhi Arts (Jyoti Verma), Kuber Essentials, Pansari Industries, Romana Herbalcare Pvt. Ltd. (Rosa) and Shree Shyam Til Patti (Raj Kumari).
In the Food Stalls category, Bansal Food and Beverages received the Gold Medal, Rohilya Food Pvt. Ltd. received the Silver Medal and Daana Pani received the Bronze Medal. Special Commendations were awarded to Vinayak Enterprises – Saada Punjab and Mother Dairy Fruit and Vegetables Pvt. Ltd.
In the Vending Points category, Daana Pani received the Gold Medal, Gath Bandhan Farms received the Silver Medal and Bansal Foods and Beverages received the Bronze Medal.
Managing Director, ITPO, Dr. Neeraj Kharwal, stated that the 14-day fair witnessed broad participation from diverse sectors and recorded over 18 lakh visitors. He noted that the theme “Ek Bharat, Shreshtha Bharat” was reflected throughout the event, with displays ranging from cutting-edge technologies and state-of-the-art pavilions to agricultural innovations and eco-friendly initiatives, showcasing India’s resilience, creativity and vast potential. He further invited suggestions to help enhance future editions of the fair.
In his welcome address, Lt. Col. Harsh Kondilya, noted that participants reported encouraging business outcomes during this year’s fair. He congratulated the award winners across categories and expressed appreciation to participants and visitors for their continued confidence in the IITF platform.
Present on the occasion were Chairman, Airports Authority of India, Shri Vinay Kumar; Executive Director, ITPO, Shri Premjit Lal; OSD, ITPO, Lt. Col. Harsh Kondilya; and General Manager, ITPO, Shri S.N. Bharti.
India and the UAE successfully convened the third meeting of the Joint Committee under the India–UAE CEPA in New Delhi. The meeting was co-chaired by Additional Secretary, Department of Commerce, Shri Ajay Bhadoo and Assistant Undersecretary for International Trade Affairs, UAE, H.E Juma Al Kait. Both sides welcomed the strong growth in bilateral trade, which crossed USD 100.06 billion in FY 2024–25, reflecting a robust increase of 19.6% and reaffirming the UAE’s position as one of India’s key trading partners. The India-UAE Joint Commission serves as the primary institutional mechanism to review progress, address challenges, and implement the CEPA.
The two sides conducted a comprehensive review of progress under CEPA and detailed discussions were held on market access issues, data sharing, allocation of Gold TRQ, anti-dumping matters, services, Rules of Origin, BIS licensing etc. The India side also briefed the UAE on its recent decision to allocate Gold TRQ through a transparent competitive bidding process.
Both sides reviewed recent high-level engagements, including meetings between Hon’ble Commerce & Industry Minister Shri Piyush Goyal and H.E. Dr. Thani in Mumbai and Dubai. They reaffirmed their shared commitment to expanding non-oil/ non precious metal trade towards the USD 100 billion target by 2030. Discussions also covered regulatory cooperation in pharmaceuticals, resolution of issues related to Certificates of Origin, BIS coordination, and early signing of the MoU on Food Safety and Technical Requirements between Agricultural and Processed Food Products Export Development Authority (APEDA), India and Ministry of Climate Change & Environment (MoCCAE), UAE.
The meeting concluded with both sides agreeing to strengthen trade facilitation, regulatory collaboration, data sharing and convening of Services Subcommittee meeting. The UAE delegation also had a meeting with Commerce Secretary, Shri Rajesh Agrawal where issues related to optimum CEPA utilisation by both sides were discussed. The visit of the UAE delegation underscores the commitment of both countries to deepen the balance of trade, expand market opportunities, and further reinforce the strategic partnership under CEPA.
From 1 January to 2 November 2025, a total of 20 412 suspected diphtheria cases, including 1 252 deaths (an average case fatality ratio [CFR] - 6 %) have been reported across eight Member States in the WHO African Region (Algeria, Chad, Guinea, Mali, Mauritania, Niger, Nigeria, and South Africa). Several of these countries have been experiencing ongoing outbreaks since 2023. Children and young adults represent the majority of the cases, with females accounting for a slightly greater proportion. Case confirmation and management remain constrained. Laboratory confirmation remains low due to shortages of diagnostic supplies and limited testing capacity. At the same time, the global shortage of diphtheria antitoxin (DAT), and uneven clinical capacity to provide this essential treatment, pose significant challenges for effective case management. Diphtheria is a vaccine-preventable bacterial disease which can lead to severe systemic complications, including myocarditis, kidney failure, and neurological damage. Many of the affected countries are fragile, conflict-affected or have system vulnerabilities where health systems are overstretched, routine services are disrupted and access to essential services is limited. These settings are characterized by high population mobility, and crowded living conditions, especially among displaced populations. WHO continues to work across all levels of the organization to identify and implement the most appropriate mechanisms to support affected countries and mitigate the impact of the outbreaks. Given this context, the regional public health risk is assessed as high due to the potential for further geographic expansion of outbreaks, high case fatality rates, insufficient resources for outbreak control, and limited surveillance and laboratory systems. At the global level, the public health risk is considered low, as most countries outside the African Region have established immunization programs and adequate surveillance systems.
Read the full DON here: https://www.who.int/emergencies/disease-outbreak-news/item/DON588

Media contacts:
mediainquiries@who.int
An industry interface organised by Export Promotion Council for EOUs & SEZs (EPCES) brought together over 150 stakeholders for an interaction with Commerce Secretary Shri Rajesh Agrawal, IAS in Chennai, today. Representatives from Special Economic Zone (SEZ) units and developers highlighted issues related to Special Economic Zone(SEZ)– Domestic Tariff Area (DTA) transactions, duty foregone, ICEGATE connectivity constraints, import monitoring systems, and reverse job work challenges faced by MSMEs. IT/ITES stakeholders raised concerns over rules on vacant built-up area classification, timelines for renewal of Letters of Approval (LoA), and procurement attestation requirements that differ from GST norms.
Participants also drew attention to the lack of parity for SEZ units under concessional import duty and duty drawback schemes available to DTA units, operational inefficiencies introduced by the new Procurement Certificate process, countervailing duties imposed by the US, and the need to review QCO applicability for SEZ-to-DTA sales, particularly in cases such as KASEZ.
Responding to the issues, the Commerce Secretary thanked EPCES for enabling direct industry engagement and assured that the Ministry would examine each challenge in detail with a view to strengthening ease of doing business. He noted that shifts in global value chains, increased demand for DTA market access, and the evolving impact of FTAs necessitate SEZ policies that align with current economic realities.
He emphasised the importance of long-term vision and data-driven policymaking, urging industry to prepare structured analyses to support reform considerations. He assured stakeholders that connectivity concerns with BSNL and issues related to import monitoring systems would be reviewed, reiterating the Government’s commitment to enhancing the SEZ ecosystem to make it more dynamic, responsive and globally competitive.
He concluded by reaffirming that the Ministry will examine all issues raised and work toward pragmatic solutions to build a more flexible, efficient and globally aligned SEZ framework.
The two-day National One Health Mission Assembly 2025 concluded successfully at Bharat Mandapam today. The second day featured focused technical deliberations that further strengthened the collective national effort to build an integrated and resilient One Health ecosystem. The Assembly convened senior officials from key ministries, scientific bodies, development partners and implementation agencies, underscoring the importance of coordinated action across human, animal and environmental health.
Following a strong start on Day 1 during which senior leadership across government reaffirmed their commitment to joint stewardship and whole-of-government collaboration, today’s sessions sustained that momentum through deeper scientific, operational and programmatic discussions. These dialogues advanced shared priorities and laid the groundwork for enhanced preparedness, rapid response capabilities, and long-term One Health integration across sectors.
The day’s proceedings were led by senior national and international experts. Dr V K Paul, Member (Health), NITI Aayog, set the tone with a call for sustained collaboration, system preparedness and stronger national capacities. He was joined by Dr Rajiv Bahl, Secretary, Department of Health research and Director General, ICMR and Dr Rajesh S Gokhale, Secretary of the Department of Biotechnology, who highlighted the importance of innovation, translational science and integrated surveillance. Contributions from Mr. Scott Newman of Food and Agriculture Organization (FAO) and Smt. Vandana Jain, Joint Secretary, Ministry of Health and Family Welfare reinforced the value of multisectoral engagement and global cooperation. Experts from institutions such as DRDO, ICAR, THSTI, CEPI, FIND, AYUSH and the International Vaccine Institute also participated, bringing diverse scientific and implementation perspectives to the discussions.
Speaking at the Assembly, Dr Vinod Kumar Paul, Member (Health), NITI Aayog, said, “India’s progress in One Health relies on a strong whole-of-government approach that advances a healthier and more resilient future. Community participation remains central to this effort. The media plays a crucial role in shaping public understanding and addressing misinformation, while our law and order systems serve as vital force multipliers during emergencies. Strengthening these partnerships will ensure timely, trusted and coordinated action when it matters most.”

Dr Paul underscored that community engagement is the foundation of early disease detection, surveillance and rapid response, and highlighted that community-led mobilisation was one of India’s greatest strengths during the COVID-19 pandemic. He stressed that One Health preparedness must extend to the grassroots, where frontline workers, local governments and communities form the first line of defence. He also noted that India’s One Health journey has now reached an important inflection point, with the Assembly successfully bringing together diverse stakeholders to advance integrated action across sectors.
Dr Rajiv Bahl, Secretary, Department of Health Research and Director General, ICMR, stated, “We need science, technology and development to work in unison. The goal is not only to build diagnostics, therapeutics and vaccines for future outbreaks, but to do so with speed. By bringing together experts from across sectors, the National one Health Mission platform is helping shape a more agile, prepared and responsive ecosystem for managing current and future public health threats.”

Dr Rajesh S Gokhale, Secretary, Department of Biotechnology, noted, “COVID-19 was a watershed moment that exposed both the fragility and the global interdependence of our technological future. What is now clear is that the triad of intelligence — biological, artificial and natural — will redefine all future technologies. Their convergence will drive a scale and speed of innovation that is difficult to imagine today. Harnessing this potential will be central to strengthening India’s One Health and bioscience capabilities.”

In the discussions on medical countermeasures, experts from leading national and international research institutions underscored the need to accelerate the development of vaccines, diagnostics and therapeutics that can rapidly respond to emerging threats. They emphasized building agile research platforms, strengthening regulatory systems for emergency use, and expanding collaborations between scientific agencies, industry and global partners. State governments added practical insights, sharing experiences on implementing surveillance systems, inter-departmental coordination and operational readiness at the field level.
Conversations on capacity building and community engagement further highlighted that a strong One Health system depends on skilled human resources, trusted institutions and empowered communities. Experts from wildlife health, public health training, community-based research and health systems development stressed the need for multi-tiered training architectures, integration of One Health into professional education, and sustained community partnerships. State governments shared diverse implementation experiences that demonstrated the importance of tailored community engagement strategies and locally adapted solutions.
The day also featured an exhibition showcasing India’s expanding One Health capabilities. Institutions demonstrated innovations in surveillance, biosafety, digital platforms, laboratory networks and collaborative research efforts, reflecting the country’s growing technological and scientific capacity. A felicitation ceremony was held for the National One Health Hackathon, which invited tech-driven and community-focused solutions to strengthen India’s One Health Mission, bringing together multidisciplinary student and early-career teams to develop practical prototypes addressing real-world One Health challenges.

The deliberations concluded with a shared recognition that One Health is essential to achieving the national vision of Viksit Bharat. By advancing scientific excellence, enabling cross-sectoral cooperation and strengthening preparedness across all levels of the system, India continues to move towards a safer and more resilient future.
The Governor of the Japan Bank for International Cooperation (JBIC), Mr. Nobumitsu Hayashi, led a delegation to the National Industrial Corridor Development Corporation (NICDC) flagship industrial park, Dholera Special Investment Region (Dholera SIR), Gujarat, today to review the progress of India’s first and emerging Semicon City and assess the fast-advancing greenfield and ‘Plug-and-Play’ industrial infrastructure ecosystem. The visit comes at a time when India is witnessing unprecedented momentum in semiconductor manufacturing, with Dholera positioned at the forefront as the country’s most ambitious greenfield smart industrial city.
During the visit, JBIC Governor Mr. Hayashi appreciated the world-class planning, infrastructure readiness and strong governance framework of Dholera SIR. He expressed confidence in India’s semiconductor growth story and noted the rapid progress of the Tata Electronics Semiconductor Fabrication Facility. He stated that Dholera’s integrated systems, scalable utilities and future-ready infrastructure make it a globally competitive destination for advanced electronics and semiconductor investments.
The visit began at the ABCD Building, Dholera’s administrative and command centre, where the delegation reviewed the integrated planning framework, digital governance architecture and centralised utility management systems. The delegation then visited major utility installations, including the Integrated Command and Control Centre (ICCC), the Experience Centre and the Torrent Power 400 kV Substation, highlighting Dholera’s strong utility backbone and readiness for advanced manufacturing.
At the Tata Electronics Semiconductor Fabrication Facility in Dholera SIR, the delegation held discussions with Tata Group leadership and reviewed the 163-acre semiconductor fabrication plant being developed with an investment of ₹91,000 crore in partnership with Powerchip Semiconductor Manufacturing Corporation (PSMC), Taiwan. The facility, India’s first commercial semiconductor fabrication plant, will commence production in 2027, manufacturing 50,000 wafers per month across 110 nm to 28 nm nodes and generating more than 20,000 high-skilled jobs. JBIC Governor Hayashi expressed optimism that the project would strengthen India–Japan technological collaboration and contribute to global semiconductor value chains.
The Chief Executive Officer and Managing Director of the National Industrial Corridor Development Corporation (NICDC), Shri Rajat Kumar Saini, briefed the delegation on the key features of Dholera SIR. The Managing Director of the Dholera Industrial City Development Limited (DICDL), Shri Kuldeep Arya, along with officials from NICDC and DICDL, was also present.
Japanese companies continue to expand their presence across NICDC industrial cities, reflecting strong confidence in India’s manufacturing ecosystem. The Toyota Group remains a major contributor with investments across multiple NICDC nodes covering more than 116 acres and exceeding ₹1,700 crore. The latest addition to the pipeline is NX Logistics India Private Limited, which has expanded operations from its 12.86-acre facility in Shendra (AURIC) to a new facility in Dholera, raising its total commitment to ₹86 crore and creating 400 jobs. Other Japanese companies, including Nippon Express, Nagata, Sango and Fuji Silvertech, are also scaling operations across logistics, component manufacturing and industrial infrastructure projects.
Dholera Industrial City has emerged as a key destination for strategic international engagements exploring opportunities in advanced manufacturing, semiconductors and smart industrial ecosystems. Recently, the Ambassador of Japan to India and Bhutan, Mr. Ono Keiichi, led a 70-member Japanese business delegation representing sectors such as semiconductors, electronics, machinery, infrastructure, logistics and financial services. The visit strengthened Japan’s engagement and reinforced Dholera’s position as a preferred destination for Japanese investors. Earlier, the Vice Governor of Iwate Prefecture, Japan, Mr. Jun Sasaki, along with representatives from the Japan International Cooperation Agency (JICA), visited Dholera on 04 September 2025 to explore collaboration opportunities in industrial development and high-technology manufacturing.
Over the past year, Dholera has hosted delegations from JBIC, the Japan Chamber of Commerce, the Japan External Trade Organization (JETRO), JICA, leading Japanese corporations and global investors. These engagements, along with continuous discussions with Japanese companies evaluating semiconductor, electronics system design and manufacturing (ESDM) and advanced manufacturing opportunities, highlight Dholera’s growing role in India–Japan industrial cooperation under the National Industrial Corridor Development Programme (NICDP).
Dholera is also developing into a complete smart industrial city supported by robust social infrastructure. Key facilities under development include a 200-bed multispeciality hospital, an integrated school, a guest house, a corporate hotel, an international tent city, a multi-cuisine food court and a fire station. Private developers are progressing with residential and commercial projects, including a mixed-use complex in the Activation Area with apartments, retail and business spaces.
With rapid infrastructure development, a strong pipeline of global investors and India’s most comprehensive semiconductor manufacturing ecosystem, Dholera is emerging as a major driver of India’s industrial transformation and a preferred hub for next-generation industries. The visit of JBIC Governor Hayashi reflects international confidence in Dholera’s capabilities and its strengthening role in global and India–Japan semiconductor value chains.

Basel, 18 November 2025 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today positive phase III results from the lidERA Breast Cancer study evaluating investigational giredestrant as an adjuvant endocrine treatment for people with oestrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, early-stage breast cancer. The study met its primary endpoint at a pre-planned interim analysis, showing a statistically significant and clinically meaningful improvement in invasive disease-free survival with giredestrant versus standard-of-care endocrine therapy. lidERA is the first phase III trial of a selective oestrogen receptor degrader (SERD) to demonstrate a significant benefit in the adjuvant setting. The majority of breast cancer cases are diagnosed at an early stage.2
“Today’s results underscore the potential of giredestrant as a new endocrine therapy of choice for people with early-stage breast cancer, where there is a chance for cure,” said Levi Garraway, MD, PhD, Roche’s Chief Medical Officer and Head of Global Product Development. “Given that ER-positive breast cancer accounts for approximately 70% of cases diagnosed, these findings – together with recent data in the advanced ER-positive setting – suggest that giredestrant has the potential to improve outcomes for many people with this disease.”
Overall survival data were immature at the time of interim analysis, but a clear positive trend was observed. Giredestrant was well tolerated and adverse events were consistent with its known safety profile, with no unexpected safety findings observed. Data from lidERA will be presented at an upcoming medical meeting and shared with health authorities with the aim of bringing this potential treatment option to patients around the world.
ER-positive breast cancer accounts for approximately 70% of breast cancer cases.3 Currently, up to a third of people eventually experience recurrence on or after adjuvant endocrine therapy treatment for early-stage breast cancer.4-6 Additionally, many have to interrupt or stop treatment early due to safety or tolerability issues, thereby increasing the risk of death.7,8 These limitations underscore the need for more effective and better-tolerated options that can enhance adherence and prevent or delay disease recurrence.
lidERA is the second positive phase III readout for giredestrant following evERA Breast Cancer, which was presented at the European Society for Medical Oncology Congress 2025.1 The scientific rationale for lidERA was supported by prior results in the neoadjuvant setting, including the coopERA trial showing that giredestrant was superior to an aromatase inhibitor in reducing malignant cell division (Ki67 levels).9 This growing body of evidence supports the potential of giredestrant to meaningfully improve outcomes compared with standard-of-care endocrine therapy across ER-positive early-stage and advanced breast cancer.1
Roche’s extensive giredestrant clinical development programme spans multiple treatment settings and lines of therapy, reflecting our commitment to deliver innovative medicines to as many people with ER-positive breast cancer as possible.
About the lidERA Breast Cancer study
lidERA Breast Cancer [NCT04961996] is a phase III, randomised, open-label, multicentre study evaluating the efficacy and safety of adjuvant giredestrant versus standard-of-care endocrine therapy in people with medium- or high-risk stage I-III oestrogen receptor-positive, human epidermal growth factor receptor 2-negative breast cancer. Over 4,100 patients were enrolled in the study.10
The primary endpoint is invasive disease-free survival (iDFS) excluding unrelated cancers in other organs (second primary non-breast cancers).10 Key secondary endpoints include overall survival, iDFS including second primary non-breast cancers, disease-free survival and safety.10
About giredestrant
Giredestrant is an investigational, oral, potent next-generation selective oestrogen receptor degrader and full antagonist.11
Giredestrant is designed to block oestrogen from binding to the oestrogen receptor, triggering its breakdown (known as degradation) and stopping or slowing down the growth of cancer cells.12
Giredestrant has an extensive clinical development programme and is being investigated in five company-sponsored phase III clinical trials that span multiple treatment settings and lines of therapy to benefit as many people as possible:
About oestrogen receptor (ER)-positive breast cancer
Globally, the burden of breast cancer continues to grow, with 2.3 million women diagnosed and 670,000 dying from the disease every year.17 Breast cancer remains the number one cause of cancer-related deaths amongst women, and the second most common cancer type.18
ER-positive breast cancer accounts for approximately 70% of breast cancer cases.4 A defining feature of ER-positive breast cancer is that its tumour cells have receptors that attach to oestrogen, which can contribute to tumour growth.19
Despite treatment advances, ER-positive breast cancer remains particularly challenging to treat due to its biological complexity.20 Patients often face the risk of disease progression, treatment side effects and resistance to endocrine therapy.20,21 There is an urgent need for more effective treatments that can delay clinical progression and reduce the burden of treatment on people’s lives.20,21
About Roche in breast cancer
Roche has been advancing breast cancer research for more than 30 years, and it continues to be a major focus of research and development. Our legacy began with the development of the first targeted therapy for human epidermal growth factor receptor 2-positive breast cancer, and we continue to push the boundaries of science to address the complexities of all breast cancer subtypes.
By leveraging our dual expertise in pharmaceuticals and diagnostics, we are dedicated to providing tailored treatment approaches and improving outcomes for every patient, from early to advanced stages of the disease. Together with our partners, we are relentlessly pursuing a cure, as we strive for a future where no one dies from breast cancer.
About Roche
Founded in 1896 in Basel, Switzerland, as one of the first industrial manufacturers of branded medicines, Roche has grown into the world’s largest biotechnology company and the global leader in in-vitro diagnostics. The company pursues scientific excellence to discover and develop medicines and diagnostics for improving and saving the lives of people around the world. We are a pioneer in personalised healthcare and want to further transform how healthcare is delivered to have an even greater impact. To provide the best care for each person we partner with many stakeholders and combine our strengths in Diagnostics and Pharma with data insights from the clinical practice.
For over 125 years, sustainability has been an integral part of Roche’s business. As a science-driven company, our greatest contribution to society is developing innovative medicines and diagnostics that help people live healthier lives. Roche is committed to the Science Based Targets initiative and the Sustainable Markets Initiative to achieve net zero by 2045.
Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan.
For more information, please visit www.roche.com.
All trademarks used or mentioned in this release are protected by law.
References
[1] Mayer E, et al. Giredestrant (GIRE), an oral selective oestrogen receptor (ER) antagonist and degrader, + everolimus (E) in patients (pts) with ER-positive, HER2-negative advanced breast cancer (ER+, HER2– aBC) previously treated with a CDK4/6 inhibitor (i): Primary results of the Phase III evERA BC trial. Presented at: ESMO Congress; 2025 October 17-21; Berlin, Germany. LBA #16.
[2] Benitez Fuentes JD, et al. Global Stage Distribution of Breast Cancer at Diagnosis. A Systematic Review and Meta-Analysis. JAMA Oncol. 2024;10(1):71-78.
[3] Kinslow C, et al. Prevalence of Estrogen Receptor Alpha (ESR1) Somatic Mutations in Breast Cancer. JNCI Cancer Spectrum; 2022 Oct;6(5):pkac060.
[4] O’Shaughnessy J, et al. Real-world risk of recurrence and treatment outcomes with adjuvant endocrine therapy in patients with stage II-III HR+/HER2- early breast cancer. Breast. 2025; 81:104437.
[5] Pan H, et al. 20-Year Risks of Breast-Cancer Recurrence after Stopping Endocrine Therapy at 5 Years. NEJM. 2017;377:1836–1846.
[6] Khatpe AS, et al. Nexus between PI3K/AKT and Estrogen Receptor Signaling in Breast Cancer. Cancers (Basel). 2021;13(3):369.
[7] Hershman DL, et al. Early discontinuation and non-adherence to adjuvant hormonal therapy are associated with increased mortality in women with breast cancer. Breast Cancer Res Treat. 2011;126:529–537.
[8] Rosso R, et al. Adherence to Adjuvant Endocrine Therapy in Breast Cancer Patients. Curr Oncol. 2023 Jan 21;30(2):1461-1472.
[9] Hurvitz SA, et al. Neoadjuvant palbociclib plus either giredestrant or anastrozole in oestrogen receptor-positive, HER2-negative, early breast cancer (coopERA Breast Cancer): an open-label, randomised, controlled, phase 2 study. Lancet Oncol. 2023;24:1029–1041.
[10] ClinicalTrials.gov. A Study Evaluating the Efficacy and Safety of Adjuvant Giredestrant Compared With Physician's Choice of Adjuvant Endocrine Monotherapy in Participants With Estrogen Receptor-Positive, HER2-Negative Early Breast Cancer (lidERA Breast Cancer) [Internet; cited 2025 November]. Available from: https://clinicaltrials.gov/study/NCT04961996.
[11] Martin M, et al. Giredestrant (GDC-9545) vs physician choice of endocrine monotherapy (PCET) in patients (pts) with ER+, HER2– locally advanced/metastatic breast cancer (LA/mBC): Primary analysis of the phase 2, randomised, open-label acelERA BC study. Presented at: The European Society for Medical Oncology Annual Meeting; 2022 September 9-13; Paris, France. Abstract #211MO.
[12] Metcalfe C, et al. GDC-9545: A novel ER antagonist and clinical candidate that combines desirable mechanistic and pre-clinical DMPK attributes. Presented at: San Antonio Breast Cancer Symposium; 2018 December 4-8; San Antonio, Texas, USA. Abstract #P5-04-07.
[13] ClinicalTrials.gov. A Study Evaluating the Efficacy and Safety of Giredestrant Plus Everolimus Compared With the Physician's Choice of Endocrine Therapy Plus Everolimus in Participants With Estrogen Receptor-Positive, HER2-Negative, Locally Advanced or Metastatic Breast Cancer (evERA Breast Cancer) [Internet; cited 2025 November]. Available from: https://clinicaltrials.gov/study/NCT05306340.
[14] ClinicalTrials.gov. A Study Evaluating the Efficacy and Safety of Giredestrant Combined With Palbociclib Compared With Letrozole Combined With Palbociclib in Participants With Estrogen Receptor-Positive, HER2-Negative Locally Advanced or Metastatic Breast Cancer (persevERA Breast Cancer) [Internet; cited 2025 November]. Available from: https://clinicaltrials.gov/study/NCT04546009.
[15] ClinicalTrials.gov. A Study to Evaluate Efficacy and Safety of Giredestrant Compared With Fulvestrant (Plus a CDK4/ 6 Inhibitor), in Participants With ER-Positive, HER2-Negative Advanced Breast Cancer Resistant to Adjuvant Endocrine Therapy (pionERA Breast Cancer) [Internet; cited 2025 November]. Available from: https://clinicaltrials.gov/study/NCT06065748.
[16] ClinicalTrials.gov. A Study to Evaluate the Efficacy and Safety of Giredestrant in Combination With Phesgo (Pertuzumab, Trastuzumab, and Hyaluronidase-zzxf) Versus Phesgo in Participants With Locally Advanced or Metastatic Breast Cancer (heredERA Breast Cancer) [Internet; cited 2025 November]. Available from: https://clinicaltrials.gov/study/NCT05296798.
[17] World Health Organisation. Breast Cancer [Internet; cited 2025 November]. Available from: https://www.who.int/news-room/fact-sheets/detail/breast-cancer.
[18] World Health Organization. Cancer Today [Internet; cited 2025 November]. Available from: https://gco.iarc.fr/today/en/dataviz/bars?mode=cancer&types=1&group_populations=1&sexes=2&key=asr&age_end=14.
[19] National Cancer Institute. Hormone Therapy for Breast Cancer [Internet; cited 2025 November]. Available from: https://www.cancer.gov/types/breast/breast-hormone-therapy-fact-sheet.
[20] Hanker A, et al. Overcoming Endocrine Resistance in Breast Cancer. Canc Cell. 2020 Apr 13;37(4):496–513.
[21] Başaran G, et al. Ongoing unmet needs in treating estrogen receptor-positive/HER2-negative metastatic breast cancer. Cancer Treat Rev. 2018 Feb;63:144-55.
Roche Global Media Relations
Phone: +41 61 688 8888 / e-mail: media.relations@roche.com
| Hans Trees, PhD Phone: +41 79 407 72 58 | Sileia Urech Phone: +41 79 935 81 48 |
| Nathalie Altermatt Phone: +41 79 771 05 25 | Lorena Corfas Phone: +41 79 568 24 95 |
| Simon Goldsborough Phone: +44 797 32 72 915 | Karsten Kleine Phone: +41 79 461 86 83 |
| Kirti Pandey Phone: +49 172 6367262 | Yvette Petillon Phone: +41 79 961 92 50 |
| Dr Rebekka Schnell Phone: +41 79 205 27 03 |
Roche Investor Relations
| Dr Bruno Eschli Phone: +41 61 68-75284 e-mail: bruno.eschli@roche.com | Dr Sabine Borngräber Phone: +41 61 68-88027 e-mail: sabine.borngraeber@roche.com |
| Dr Birgit Masjost Phone: +41 61 68-84814 e-mail: birgit.masjost@roche.com |
Investor Relations North America
Loren Kalm
Phone: +1 650 225 3217
e-mail: kalm.loren@gene.com
Union Ministry of Health and Family Welfare, Government of India, in collaboration with Edith Cowan University (ECU), Australia, and Jhpiego, successfully inaugurated Day 1 of the two-day Roundtable on ‘Strengthening the Nursing Workforce in India and Australia: Building Collaborative Pathways for a Resilient and Skilled Nursing Workforce Aligned with the SDGs’, here today.
The dialogue aimed to foster deeper cooperation, share best practices, and develop joint pathways to build a resilient, future-ready nursing workforce aligned with the Sustainable Development Goals (SDGs).

Delivering the keynote address, Ms. Akanksha Ranjan, Deputy Secretary (Nursing & Dental), Ministry of Health and Family Welfare, noted that the roundtable is taking place at an opportune moment, coming just after the three-day National Strategic Meeting that discussed the future direction of nursing policy in India. She reiterated that “nurses are the backbone of healthcare” and emphasized the need to build a more resilient and competency-based nursing workforce.
Ms. Ranjan also highlighted that while 2.9 million nurses serve globally, the shortage stands at 4.5 million, creating significant global demand and opening pathways for ethical and well-governed nurse migration. Furthermore, she highlighted that India–Australia cooperation offers a valuable platform to jointly advance nursing education standards, expand workforce pathways, and promote ethical mobility, noting that bilateral collaboration can help both nations address emerging health system challenges more effectively.

Addressing the gathering, Dr. Deepika Khakha, Nursing Advisor, Directorate General of Health Services (DGHS), Ministry of Health and Family Welfare, stated that “Nurses remain the heartbeat of the healthcare system globally.” She emphasized that cross-learning is at the core of this roundtable, allowing India and Australia to jointly anticipate and address future Healthcare challenges.
Highlighting the Progress in Nursing Healthcare System, she stated that, India’s 3.5 million-strong nursing workforce continues to serve a rapidly evolving healthcare landscape, supported by a robust ecosystem of more than 5,000 nursing institutes offering multiple nursing programs. She informed participants that the Government of India is significantly investing in the health workforce, including the establishment of 157 new nursing institutions in future.

She also emphasized that, investing in faculty development creates a powerful cascade effect across the entire nursing ecosystem. She noted that when faculty are strengthened, the benefits naturally percolate down to students—who will ultimately form India’s future-ready and job-ready nursing workforce.
Dr. Khakha highlighted that the modernization of the nursing curriculum under the National Nursing and Midwifery Commission Act, 2023 is a transformative step toward improving the quality, competency, and equitable distribution of nursing professionals across the country. She underscored that competency-based education, digital learning platforms, enhanced clinical exposure, continuous professional development, modern regulatory frameworks, and strong leadership training will remain key pillars in advancing the quality of India’s nursing workforce.
Dr. Khakha also stated that this roundtable exemplifies the shared commitment and collaborative spirit between India and Australia. Such partnerships, she remarked, are vital for exchanging innovations, strengthening workforce planning, and aligning collective efforts with the Sustainable Development Goals. She concluded, reaffirming that collaboration and learning across borders are indispensable for shaping a resilient and future-ready nursing workforce.
Speaking at the occasion, Prof. Karen Strickland, Executive Dean, Edith Cowan University, Australia, commended the joint efforts of both countries in advancing nursing education and practice. She also emphasized that global collaboration is critical in preparing nurses who can navigate evolving healthcare demands and adopt emerging technologies.

Prof. Strickland noted that Australia and India share a long-standing partnership in nursing education, and this roundtable provides a valuable platform for sharing innovations, research insights, and best practices that can strengthen workforce capacity in both nations.
Dr. Kamlesh Lalchandani, Deputy Country Director, Jhpiego, also addressed the participants, highlighting Jhpiego’s continued partnership with the Government of India in strengthening nursing and midwifery systems. He emphasized the importance of evidence-based practice, innovation, and capacity building in shaping a responsive and resilient nursing workforce and strides made by India in the last decade in healthcare Delivery System.

The discussions identified priority areas for bilateral collaboration, including faculty development, joint research, exchange programmes, and digital learning innovations.

The workshop brought together senior officials from central and state governments, nursing leaders, academic experts and development partners, and served as a platform to advance the national agenda on nursing and midwifery reforms.
Union Minister of Commerce and Industry, Shri Piyush Goyal launched the Trade Intelligence & Analytics (TIA) Portal today in New Delhi. The Minister said that business needs to be more transparent with trade data to help stakeholders take the best decisions.
Shri Goyal stated that the Trade Intelligence Portal will open up new insights for importers, exporters, startups, and MSMEs. He added that smaller businesses, even in the remotest regions, will now have access to data that was previously available only to larger enterprises.
The Minister highlighted that there are huge opportunities in the global services sector that will now be accessible to all. He also noted that the portal will help exporters better utilise the Free Trade Agreements.
The Minister said that unless exports contribute significantly to foreign exchange reserves, it will be difficult to bring stability to forex rates, and without such stability, India will remain vulnerable to foreign forces. He described the current world tariff situation as a wake-up call and urged that government and the private sector must pool resources at this critical moment to avoid regretting a lost opportunity.
Shri Goyal invited stakeholders to bring their demands and assured that the Ministry will make every effort to address them. If an issue concerns the Department itself, it will be resolved quickly; if it concerns other departments, Commerce will actively coordinate and work towards a solution. He urged stakeholders to be more demanding so that necessary improvements can be accelerated.
Shri Goyal said the Ministry of Commerce has put in significant effort to develop this platform. He said the portal has the potential to support trade diversification, expand India’s trade basket to newer areas and newer products, and help recognise lost opportunities.
Recognizing the need for a robust intelligence and analytics framework to support evidence-based policymaking and enable targeted sectoral interventions, the Department of Commerce initiated the development of the Trade Intelligence & Analytics (TIA) Portal in March 2024. The Department felt it necessary to expand these capabilities across all commodity and territorial divisions, Export Promotion Councils (EPCs), and missions abroad in target countries.
In this context, the Department has developed a one-stop analytics Trade Intelligence & Analytics (TIA) Portal, designed to enhance trade analytics and foster data-driven evidence-based policymaking through a comprehensive and integrated platform. This one-stop solution caters to various perspectives, including India, global, and bilateral trade, with multiple databases covering trade and macro-economic indicators, providing trade insights crucial for informed decision-making. TIA Portal serves as a centralized digital hub that consolidates diverse trade databases—both global (UN Comtrade) and bilateral—into a single integrated system. Multiple existing analytics requirements were designed and developed into automated analytical capabilities, which now offer depth and breadth in understanding India’s bilateral trade dynamics as well as global trade flows between more than 220 nations. Trade Watch Tower provides country and commodity-level intelligence through specialized tools and visualisations that allow users to identify global as well as bilateral trends with enhanced analytical capabilities for identifying market diversification opportunities.
The portal offers more than 270 interactive visualisations across over 28 dashboards. With the phasing out of legacy trade information dissemination portals such as the Department of Commerce Monitoring Dashboard, Niryat Portal and Tradestat Portal, the new and more exhaustive capabilities of the TIA Portal significantly improve accessibility and usability of trade data at one place. Database collation has been automated through APIs for convenient user access, and seamless data extraction capability has been developed to extract and download commodity and country data. Automation has also been undertaken for workflows such as the Monthly Trade Report (more than 300 pages) and surge reports prepared in two versions based on trade alert data and final data (each more than 30 pages).
The Trade Data Analytics Portal is a cost-effective, open-source solution that prioritizes accessibility, scalability, and user-centric design. With no software licensing costs and only minimal server infrastructure expenses, it offers a sustainable model for long-term deployment. The portal integrates intuitive visualizations, advanced drill-down tools and automated workflows that reduce manual data handling and duplication of effort. By replacing fragmented and ad-hoc analytics with a unified and automated system, the portal enhances operational efficiency and empowers stakeholders to base strategies and policy decisions on empirical evidence. This improves responsiveness to global trade developments and strengthens the Department’s capacity for proactive, data-driven interventions.
The TIA Portal offers a comprehensive suite of tools and dashboards supporting global trade analysis and market intelligence. It provides extensive visual analytics with more than 270 interactive visualizations across more than 28 dashboards offering insights into bilateral and India-level trade patterns. Country and commodity intelligence tools help identify focus products for each market. The Trade Watch Tower identifies Champion Products by scanning global demand and mapping India’s supply capabilities. Dedicated dashboards track the performance of Production Linked Incentive (PLI) commodities, helping assess policy impact and market competitiveness. Surge monitoring tools track import and export surges across commodities and countries, including both FTA and non-FTA partners, enabling early detection of anomalies and emerging risks. A Critical Minerals Dashboard covers more than 30 critical minerals and maps India’s trade flows and global sourcing destinations at the HS code level. A Tariff Analysis Dashboard and a Target Monitoring Dashboard are under development for tariff insights and performance tracking.
The portal also provides tools to compare and contrast macroeconomic, trade and investment indicators across countries, enabling benchmarking, competitive assessments and strategic evaluations. To support advanced trade analytics, the portal incorporates trade indices such as the Trade Complementarity Index, which assesses alignment between India’s export profile and partner countries’ import needs; the Revealed Comparative Advantage index, which highlights products where India holds a competitive edge; and the Trade Intensity Index, which measures the strength of bilateral trade relationships relative to global flows. These indices enable deeper analytical capabilities and support evidence-based planning for trade expansion and policy formulation.
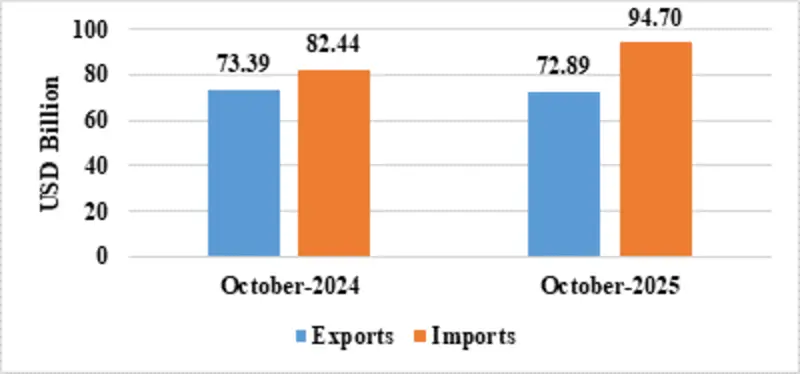
India’s total exports (Merchandise and Services combined) for October 2025* is estimated at US$ 72.89 Billion, registering a negative growth of (-) 0.68 percent vis-à-vis October 2024. Total imports (Merchandise and Services combined) for October 2025* is estimated at US$ 94.70 Billion, registering a positive growth of 14.87 percent vis-à-vis October 2024.
Table 1: Trade during October 2025*
| October 2025(US$ Billion) | October 2024(US$ Billion) | ||
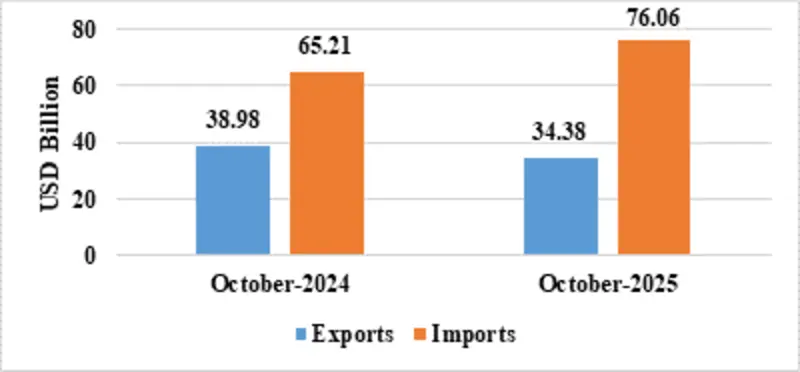
| Merchandise | Exports | 34.38 | 38.98 |
| Imports | 76.06 | 65.21 | |
| Services* | Exports | 38.52 | 34.41 |
| Imports | 18.64 | 17.23 | |
| Total Trade(Merchandise +Services) * | Exports | 72.89 | 73.39 |
| Imports | 94.70 | 82.44 | |
| Trade Balance | -21.80 | -9.05 |
* Note: The latest data for services sector released by RBI is for September 2025. The data for October 2025 is an estimation. (ii) Data for April-October 2024 and April-June 2025 has been revised on pro-rata basis using quarterly balance of payments data.
Fig 1: Total Trade during October 2025*
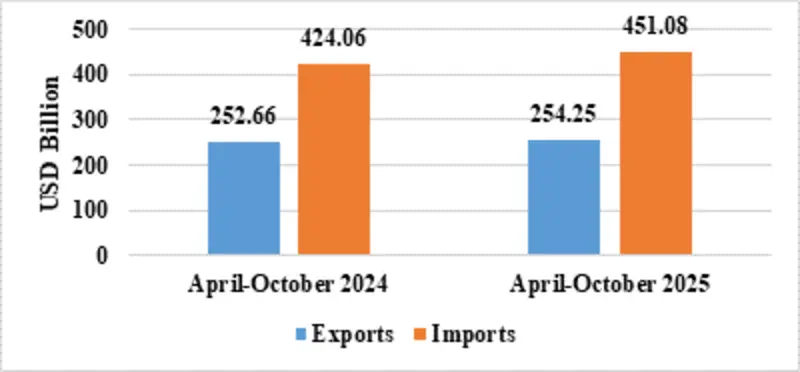
India’s total exports during April-October 2025* is estimated at US$ 491.80 Billion registering a positive growth of 4.84 percent. Total imports during April-October 2025* is estimated at US$ 569.95 Billion registering a growth of 5.74 percent.
Table 2: Trade during April-October 2025*
| April-October 2025(US$ Billion) | April-October 2024(US$ Billion) | ||
| Merchandise | Exports | 254.25 | 252.66 |
| Imports | 451.08 | 424.06 | |
| Services* | Exports | 237.55 | 216.45 |
| Imports | 118.87 | 114.96 | |
| Total Trade(Merchandise +Services) * | Exports | 491.80 | 469.11 |
| Imports | 569.95 | 539.02 | |
| Trade Balance | -78.14 | -69.92 |
Fig 2: Total Trade during April-October 2025*
MERCHANDISE TRADE
Fig 3: Merchandise Trade during October 2025
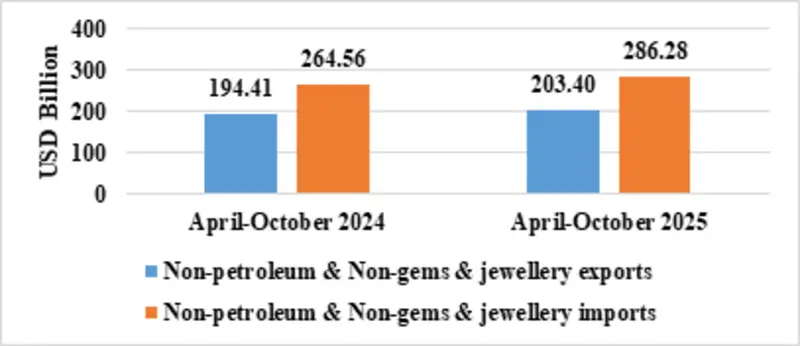
Fig 4: Merchandise Trade during April-October 2025
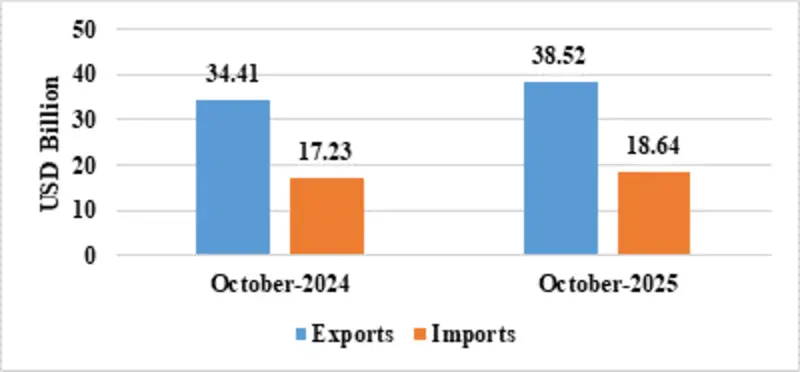
Table 3: Trade excluding Petroleum and Gems & Jewellery during October 2025
| October 2025(US$ Billion) | October 2024(US$ Billion) | |
| Non- petroleum exports | 30.43 | 34.57 |
| Non- petroleum imports | 61.27 | 46.33 |
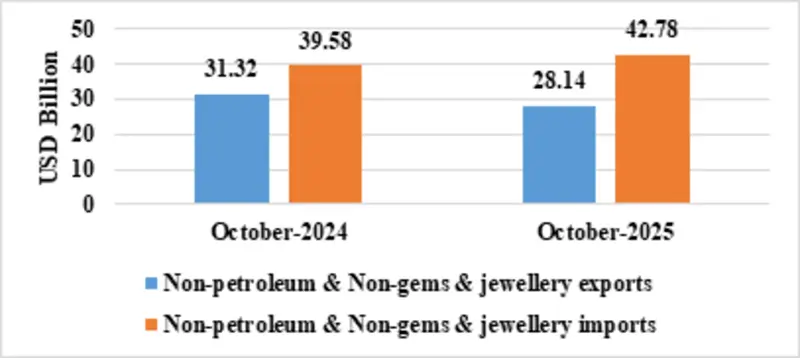
| Non-petroleum & Non-Gems & Jewellery exports | 28.14 | 31.32 |
| Non-petroleum & Non-Gems & Jewellery imports | 42.78 | 39.58 |
Note: Gems & Jewellery Imports include Gold, Silver & Pearls, precious & Semi-precious stones
Fig 5: Trade excluding Petroleum and Gems & Jewellery during October 2025
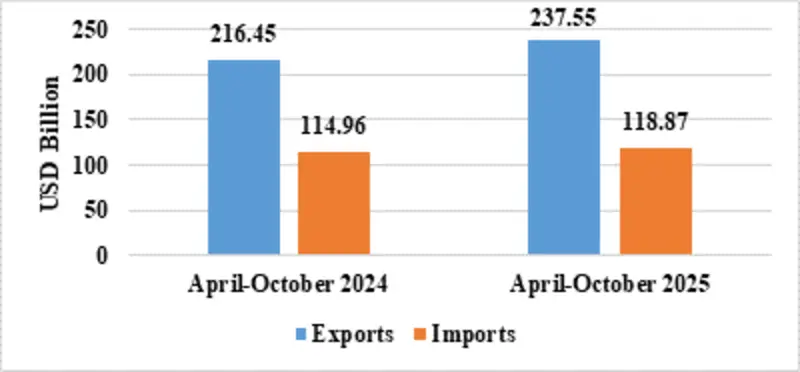
Table 4: Trade excluding Petroleum and Gems & Jewellery during April-October 2025
| April-October 2025(US$ Billion) | April-October 2024(US$ Billion) | |
| Non- petroleum exports | 219.90 | 211.60 |
| Non- petroleum imports | 344.17 | 312.13 |
| Non-petroleum & Non Gems & Jewellery exports | 203.40 | 194.41 |
| Non-petroleum & Non Gems & Jewellery imports | 286.28 | 264.56 |
Note: Gems & Jewellery Imports include Gold, Silver & Pearls, precious & Semi-precious stones
Fig 6: Trade excluding Petroleum and Gems & Jewellery during April-October 2025
SERVICES TRADE
Fig 7: Services Trade during October 2025*
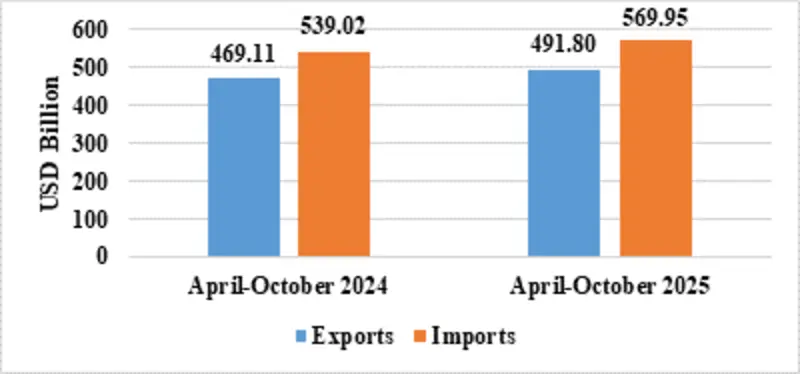
Fig 8: Services Trade during April-October 2025*