The Ministry of Health and Family Welfare, Government of India, convened a National Sensitization Meeting on Strengthening the Pre-Conception and Pre-Natal Diagnostic Techniques (Prohibition of Sex Selection) Act, 1994 at Vigyan Bhawan, New Delhi.
The deliberations focused on reinforcing the effective implementation of the PC&PNDT Act in view of emerging challenges, with an emphasis on coordinated efforts to address gaps, ensure compliance, and uphold the objectives of the legislation.
Delivering the keynote address, Ms. Aradhana Patnaik, Additional Secretary & Mission Director (NHM), Ministry of Health and Family Welfare, emphasized that the PC&PNDT Act is not just a legal instrument but a moral and social safeguard against Gender-Biased Sex Selection. She also stated that, “Women by birth are more resilient and have stronger immune System, hence the survival of female child is more possible naturally than a male child.”

Furthermore, she added that, “Instead of taking against action Gender-Biased Sex Selection, we should focus on the Prevention Part of the PC&PNDT Act.” She also emphasised that “The focus of the society or an Individual should be having a healthy Child rather than Gender of the child.”
Highlighting the impact of the Pre-conception and Pre-natal Diagnostic Techniques (Prohibition of Sex Selection) Act, 1994, she said that, ‘the country has recorded a positive improvement in the Sex Ratio at Birth (SRB). As per the Sample Registration System (SRS) Report 2023, the SRB has increased by 18 points—from 819 females per 1,000 males during 2016-18 to 917 females per 1,000 males in 2021-23. The National Sex Ratio at Birth for the period 2021-23 thus stands at 917 females per 1,000 males, reflecting the progress made through strengthened implementation of the PC&PNDT Act and related interventions.’

The inaugural session witnessed the release of IEC material, including TVC Video, radio jingle and informative posters developed by the Ministry as part of a 360-degree communication campaign, with a theme “जब लड़का लड़की है बराबर, तो पूछना क्यों?”. The National Sensitization Meeting, focused upon the directions of the Hon’ble Supreme Court of India in the case of WPC No. 341 (2008), the role of online intermediaries and digital platforms, and the monitoring compliance in the digital ecosystem, including the Section 22 of the PC&PNDT Act (which explicitly prohibits advertisements and promotions related to pre-conception and pre-natal sex determination), highlighting the pressing issue of online violations and misuse of new technologies, reinforcing the urgent need for proactive engagement with digital intermediaries and stronger compliance mechanisms to uphold the spirit of the Act.

Ms Meera Srivastava, Joint Secretary, Reproductive and Child Health (RCH), MoHFW, Dr. Indu Grewal, Additional Commissioner- PC&PNDT, MoHFW, and senior officials from Central Ministries, Representative from 36 State Governments/UTs, enforcement agencies, digital intermediaries, were also present.

Representatives from the state governments including Telangana, Haryana, Tamil Nadu, Himachal Pradesh, Rajasthan, Chhattisgarh, Madhya Pradesh, and Gujarat, shared their good practices and challenges in enforcement, while representatives from online platforms engaged in an open discussion on strengthening compliance with Section 22.
The “Swasth Nari Sashakt Parivar” Abhiyaan, launched from 17th September 2025 to 2nd October 2025, concluded with widespread participation across India, benefiting lakhs of women, children, and families through comprehensive health services.

By the end of 02nd October 2025, over 18 lakh (18,08,071) health camps (comprising both screening and speciality camps) were held under the Abhiyaan, registering a footfall of nearly 10 crore citizens (9,85,63,619) nationwide and benefitting more than 6.5 crore women.
Key achievements of the Abhiyaan include:
· Hypertension & Diabetes Screening: Over 1.78 crore citizens screenedfor hypertension and1.72 crore for diabetes.
· Cancer Screening: More than 37 lakh women screened for breast cancer, and over 19 lakhs for cervical cancer. Oral cancer screening has covered more than 69 lakh people.
· Maternal and Child Health: More than 62.60 lakh antenatal check-ups conducted, while more than 1.43 crore children received life-saving vaccines.
· Anaemia & Nutrition: More than 1.51 crore screened for anaemia. Nutrition counselling sessions reached more than 1.16 crore people.
· TB & Sickle Cell Screening: Over 85 lakh citizens screened for TB and10.23 lakh for Sickle Cell Disease. 10.69 lakh Ni-kshay Mitras registered.
· Blood Donation & PM-JAY: More than 4.30 lakh blood donors registered, alongside more than 10.69 lakhs Ayushman/PM-JAY cards issued.


In addition to the extensive network of NHM health camps, AIIMS, other Institutes of National Importance (INIs), tertiary care hospitals, Ayushman Arogya Mandirs, medical colleges, and private institutions have also been at the forefront of this national drive. These facilities have hosted thousands of specialty camps, providing advanced screening, diagnostics, counselling and treatment services to beneficiaries, thereby complementing the efforts of state governments and community-level health workers.

The Swasth Nari Sashakt Parivar Abhiyaan marks the largest ever health outreach for women and children in India. The initiative, jointly led by the Ministry of Health & Family Welfare (MoHFW) and the Ministry of Women & Child Development (MoWCD), involved organising lakhs of health camps from 17th September to 2nd October at Ayushman Arogya Mandirs, Community Health Centres (CHCs), District Hospitals and other government health facilities across the country to provide women-centric preventive, promotive, and curative health services at the community level.
The Swasth Nari Sashakt Parivar Abhiyaan strengthened screening, early detection, and treatment linkages for non-communicable diseases, anaemia, tuberculosis, and sickle cell disease, while also promoting maternal, child, and adolescent health through antenatal care, immunisation, nutrition, menstrual hygiene, lifestyle and mental health awareness activities. At the same time, the campaign mobilised communities towards healthy lifestyle practices with a special emphasis on obesity prevention, improved nutrition, and voluntary blood donation.
Union Minister of Health and Family Welfare, Shri Jagat Prakash Nadda presided as the Chief Guest at the 3rd Foundation Day event of AIIMS Bilaspur, in Himachal Pradesh today. He was joined by Shri Anurag Thakur, Member of Parliament, Lok Sabha; Shri Harsh Mahajan, Member of Parliament, Rajya Sabha; Shri Jai Ram Thakur, Ex-Chief Minister & Leader of Opposition, Himachal Pradesh and Col. Dr. Dhani Ram Shandil, Health Minister, Himachal Pradesh.


Addressing the gathering, Shri Nadda stated, “With state-of-the-art facilities such as advanced MRI, CT scanner, PET-CT, endoscopy suites, and bronchoscopy suites now in place, AIIMS Bilaspur has made remarkable progress within just three years of its establishment.” He added, “The fact that people from nearby states are also seeking treatment at AIIMS Bilaspur highlights the growing trust and confidence in this institute.”
Shri Nadda reiterated the government’s commitment to providing quality medical services. He noted significant achievements in nuclear medicine, cancer care, and renal transplants. “A major milestone for Himachal Pradesh has been the reduction in out-of-pocket expenditure. Whereas earlier people had to travel to PGI Chandigarh, today AIIMS Bilaspur meets the high-end tertiary medical needs of the region,” he stated.
On medical education, the Union Health Minister informed that 127 posts have recently been sanctioned for AIIMS Bilaspur, including 29 faculty and 98 non-faculty positions. He added that the Selection Committee will remain active for one year and is mandated to conduct at least four interview rounds to ensure these posts are filled promptly.
Shri Nadda announced plans for a 500-bed Vishram Sadan at AIIMS Bilaspur to accommodate patients traveling from distant areas. Additionally, Rs 165 crore has been sanctioned for the construction of Type III, IV, and V quarters, as well as UG student hostels, and Rs 5 crore for an indoor stadium.

Emphasizing the importance of upholding the AIIMS brand’s reputation, Shri Nadda called for replicating the successes of other AIIMS institutions by maintaining quality care and fostering innovation. He highlighted the recent initiative to organize short-term exchange programs between AIIMS centers across the country.
He also mentioned the formation of a team, in collaboration with NITI Aayog and the Indian Institute of Public Administration, to visit all AIIMS institutes and develop a future roadmap for departmental growth and sustainability.
Shri Nadda further highlighted the substantial increase in medical education over the past 11 years. “There are now 808 medical colleges in the country. Annual UG medical seats have grown from around 35,000 to 1.25 lakh. In the last Cabinet, the Hon’ble Prime Minister sanctioned 5,000 new UG and PG seats in government medical colleges,” he stated. He added that in the next five years, an additional 75,000 medical seats will be introduced in government medical institutions.

During the event, the institute’s Kulgeet was officially released, symbolizing the spirit and ethos of AIIMS Bilaspur. The proceedings also included a special award distribution ceremony, where meritorious students in recognition of their outstanding academic and extracurricular achievements were felicitated by the Union Health Minister.
Background:
AIIMS Bilaspur, Himachal Pradesh, established under the Pradhan Mantri Swasthya Suraksha Yojana (PMSSY),is a premier tertiary healthcare institute serving not only Himachal Pradesh but also the neighboring states. The foundation stone of AIIMS Bilaspur was laid by Hon'ble Prime Minister Shri Narendra Modi On October 3, 2017, and the institute was dedicated to the nation by the Hon’ble Prime Minister on October 5, 2022. Today, the institute is functioning with more than 734 operational beds, providing comprehensive healthcare, advanced diagnostics, cutting-edge cancer care, and life-saving transplant services.
Guided by the motto “Sarve Santu Niramaya” - may all be free from disease - AIIMS Bilaspur has rapidly evolved into a center of excellence for healthcare delivery, education, and research.
Dedicated to advancing healthcare excellence, AIIMS Bilaspur stands at the forefront of patient care, medical education, and research. With a focus on state-of-the-art diagnostic technologies, specialized treatment facilities and holistic patient support, the center continues to set new benchmarks in delivering quality health services for the community and beyond. Every department, from advanced diagnostics to critical care and research, reflects a commitment to patient wellbeing, clinical innovation, and sustainable growth.
Advanced Diagnostics: Equipped with state-of-the-art imaging services including a new 1.5 tesla MRI, 128-SLICE CT Scanner, and Nuclear Medicine Facilities (PET-CT, SPECT-CT, And Radionuclide Therapy).
Specialized Care: Comprehensive cancer treatment with modern Radiotherapy, Oncology Units, And Functional Cath Lab.
Critical & Emergency Services: 64 ICU beds across Cardiac, Medical, NICU, And PICU Wards, along with a dedicated Bronchoscopy suite and uninterrupted oxygen supply through an LMO plant.
Transplants and surgeries: 8 successful kidney transplants completed and eye bank with corneal transplant unit to be commissioned soon.
Research & laboratories: Establishment Of Endocrinology and Immunology Labs, And A Regional Viral Research And Diagnostic Laboratory Costing ₹19.83 Crore, Enhancing Disease Surveillance Capabilities.
Patient support: with three Amrit/Jan Aushadhi Pharmacies, affordable medicine access has expanded. A 250-bed Vishram Sadan (₹26.75 crore) is also under construction to provide accommodation for patient attendants.
Education & Training: Modern lecture hall complex, Simulation, and Skill Labs Strengthening Medical and Nursing training.
Infrastructure Development: residential quarters, hostels with 540+ capacity, rooftop solar power plant, and an indoor stadium under the khelo india initiative are under progress, ensuring sustainable growth of the institute.
AIIMS Bilaspur aims to further its role as a medical hub in North India with upcoming projects including a bone marrow transplant & therapy block (₹18.52 crore), advanced autopsy block (₹34.61 crore), a new trauma & critical care centre, a robotic surgery unit and drone-based logistics system for remote healthcare support. A multilevel car parking facility (624 vehicles, ₹72.97 crore) and a school for staff and students' families are also on the anvil.
With these milestones and ambitious expansion projects, AIIMS Bilaspur will continue to redefine healthcare accessibility and excellence in the region.
Smt. Ankita Mishra Bundela, Joint Secretary (PMSSY), Union Health Ministry; Prof. (Dr.) Narendra Kumar Arora, President, AIIMS Bilaspur; Prof. (Dr.) Dayanand Sharma, Executive Director, AIIMS Bilaspur; Lt. Col. Paranjape, Registrar and other eminent dignitaries were also present on the occasion.
The Department of Health and Family Welfare (DoHFW), along with its Central Government Hospitals, Attached and Subordinate Offices, Autonomous Bodies and CPSUs, is prepared to implement the upcoming Special Campaign 5.0 from 2nd to 31st October, 2025 to institutionalize Swachhata and minimizing pendency in government offices. The campaign, spearheaded by the Department of Administrative Reforms and Public Grievances (DARPG), aims to institutionalize swachhata (cleanliness) and ensure timely and effective disposal of pending matters. DoHFW has set extensive targets under the campaign to accelerate efficiency, streamline processes, and strengthen citizen-centric governance.
Under the Cleanliness Campaign, 1,454 siteshave been identified for cleaning to promote a more organized and hygienic workplace. DoHFW is also prioritizing identification and disposal of e-waste and unserviceable items during which revenue will be generated and valuable office space will also be freed.
To minimize pending matters, a number of public grievances and associated appeals have been earmarked for prompt resolution, reaffirming its commitment to responsive and transparent governance.
In addition, 15,494 physical filesand 3,279 e-fileshave been identified for review. Further, 11 rules/ processes have been identified for simplification to remove procedural bottlenecks and promote ease of governance.
DoHFW reaffirms its dedication to efficient governance, responsive administration and citizen-centric service delivery.
The Agricultural and Processed Food Products Export Development Authority (APEDA), under the Ministry of Commerce & Industry participated as the Export Partner in the 4th edition of World Food India (WFI) 2025, held at Bharat Mandapam, Pragati Maidan, New Delhi.
With 142 exhibitors and 120 stalls, APEDA’s pavilion showcased the diversity of India’s agri-food sector, including GI-tagged products, Basmati rice, millets, organic produce, livestock products, and value-added foods. The pavilion also featured dedicated thematic zones such as the GI Gallery, Fresca Zone, Basmati Export Development Foundation stall, and a BHARTI Zone focused on startups and innovation.
As Export Partner, APEDA organized the Reverse Buyer-Seller Meet (RBSM), which attracted more than 530 international buyers from 68 countries, including leading supermarket chains such as Walmart, Nesto, Al Madina, Mustafa Singapore, and Choithram. Over 4,654 curated B2B meetings were held during the event, providing Indian exporters and startups an unprecedented platform to explore new markets, showcase innovative products, and establish long-term business linkages.
APEDA further strengthened global market access by signing a strategic Memorandum of Understanding (MoU) with LuLu Hypermarket LLC on September 25, 2025. The collaboration, under the BHARTI (Bharat’s Hub for Agritech Resilience, Advancement and Incubation for Export Enablement) Initiative, will provide Indian agri-food startups shelf space across 252 LuLu outlets in the GCC, along with product sampling campaigns, consumer engagement, and access to LuLu’s export division for global distribution.
The BHARTI Initiative was another major highlight at WFI 2025, with a dedicated stall and an interactive seminar on “From Farm to Global Markets: Startup-Driven Innovation for Agri-Food Exports.” The session brought together policymakers, industry leaders, and innovators to deliberate on strategies for empowering 100 agri-food ventures through mentorship, branding, technology adoption, and global market intelligence.
Speaking on the occasion, Chairman, APEDA, Shri Abhishek Dev said that World Food India 2025 has been a milestone in showcasing India’s agri-food potential to the world. As Export Partner, APEDA successfully brought together more than 140 exhibitors and over 500 buyers from 68 countries, enabling thousands of business meetings and new opportunities for exporters. The Reverse Buyer-Seller Meet, the MoU with LuLu Hypermarket, and the BHARTI Initiative together reflect APEDA’s commitment to empowering startups, strengthening international partnerships, and building a future-ready, innovation-driven export ecosystem.
Through its active participation at World Food India 2025, APEDA reaffirmed its mission to build a globally competitive agri-food export sector. By combining large-scale buyer engagement, strategic international collaborations, and startup-driven innovation, APEDA continues to position India as a trusted partner in global food trade and a leader in sustainable, innovation-led growth.
The landmark Trade and Economic Partnership Agreement (TEPA) between India and the EFTA States (Iceland, Liechtenstein, Norway and Switzerland) entered into force on 1 October 2025, representing a significant milestone in the trade and economic relations between the parties. India is the fastest‑growing large economy and is on course to become the world’s third‑largest economy. The EFTA States collectively rank among the global leaders in both merchandise and services trade. Together India and the EFTA States represent a combined GDP of about USD 5.4 trillion, providing the scale for deeper integration.
The occasion was marked by a high-level event, the Prosperity Summit, in New Delhi hosted by Shri Piyush Goyal, Minister of Commerce & Industry of India. EFTA was represented by Helene Budliger Artieda, Swiss State Secretary at the State Secretariat for Economic Affairs ; Ragnar Kristjánsson, Director General of External Trade and Economic Affairs at the Icelandic Ministry for Foreign Affairs; Christine Lingg, Deputy Director of the Office for Foreign Affairs of Liechtenstein; May-Elin Stener, Ambassador of Norway to India; and Markus Schlagenhof, Deputy Secretary-General of EFTA. The event also brought together a range of business representatives from all Parties, providing an opportunity to establish new ties and strengthen existing partnerships. Business engagement will facilitate co‑production in precision manufacturing and clean technologies, as well as scaling services through predictable mobility and recognition of qualifications.
The dignitaries welcomed the shared objectives to mobilise USD 100 billion of investments in India over fifteen years and to support the creation of one million direct jobs, with an investment facilitation mechanism to monitor delivery.
The entry into force of the TEPA is a concrete step towards deepening the relationships between the EFTA States and India, building bridges and encouraging trade and investment. The parties will pursue balanced and sustainable outcomes by expanding total trade and strengthening value‑chain integration across goods, services and investment, supported by standards cooperation and regulatory dialogue.
The agreement is set to deliver substantial and long-lasting benefits, including more resilient and better integrated supply chains, increased trade and investment flows, new job opportunities, and sustained economic growth. It provides enhanced market access and streamlines customs procedures, making it easier for Indian and EFTA businesses to expand their operations into growing markets.
The agreement also aims to facilitate and promote investment opportunities between the Parties, creating an environment for Indian and EFTA businesses to innovate, expand and prosper. The Parties note that trade in goods has increased steadily over time and trade in services has roughly doubled in the last decade. The Parties will strive to increase total two‑way trade substantially.
The India-EFTA TEPA was signed on 10 March 2024 in New Delhi. This partnership aligns with the trajectory of the parties for building resilient and reliable global supply chains and expanding the links between them as trusted partners.
Department for Promotion of Industry and Internal Trade (DPIIT), Ministry of Commerce & Industry, signed a Memorandum of Understanding (MoU) with Thermo Fisher Scientific (TFS) to accelerate innovation in India’s biotechnology sector and nurture a strong pipeline of high-growth startups.
The partnership aims to support over 500 biotech startups in the next three years through strategic advisory, technology access, mentorship, and investor connect. As part of the MoU, TFS under Startup India, DPIIT’s initiative Bharat Startup Grand Challenge (BSGC) will launch the BioVerse Challenge, a nationwide platform to identify and nurture India’s most promising biotech entrepreneurs, and the BioVerse Mentors Circle to provide skill development and technical training across leading bio-incubators.
Under this partnership:
Speaking on the occasion, Shri Sanjiv, Joint Secretary, DPIIT, said, “The collaboration between Startup India, DPIIT and Thermo Fisher Scientific marks a significant step towards strengthening India’s biotechnology ecosystem. By combining Thermo Fisher’s global expertise with Startup India’s strong innovation network, we aim to create new opportunities for early-stage biotech entrepreneurs to scale their solutions and contribute to India’s bioeconomy.”
Highlighting TFS’s commitment, Shri Srinath Venkatesh, Managing Director, India & South Asia, Thermo Fisher Scientific, said, “We are honoured to formalize our collaboration with DPIIT through this MOU, and to partner on the BioVerse Challenge. The BioVerse Challenge reflects our commitment to working closely with early-stage start-ups and bio-incubators across India. Through our upcoming facilities in Genome Valley, Hyderabad, we will give founders access to technologies and guidance that can help them move from prototype to product faster. It will enable them to compete globally.”
Thermo Fisher Scientific brings unmatched expertise in life sciences research, diagnostics, and biotechnology. Together with Startup India, DPIIT this initiative will catalyze India’s position as a global hub for biotech innovation.
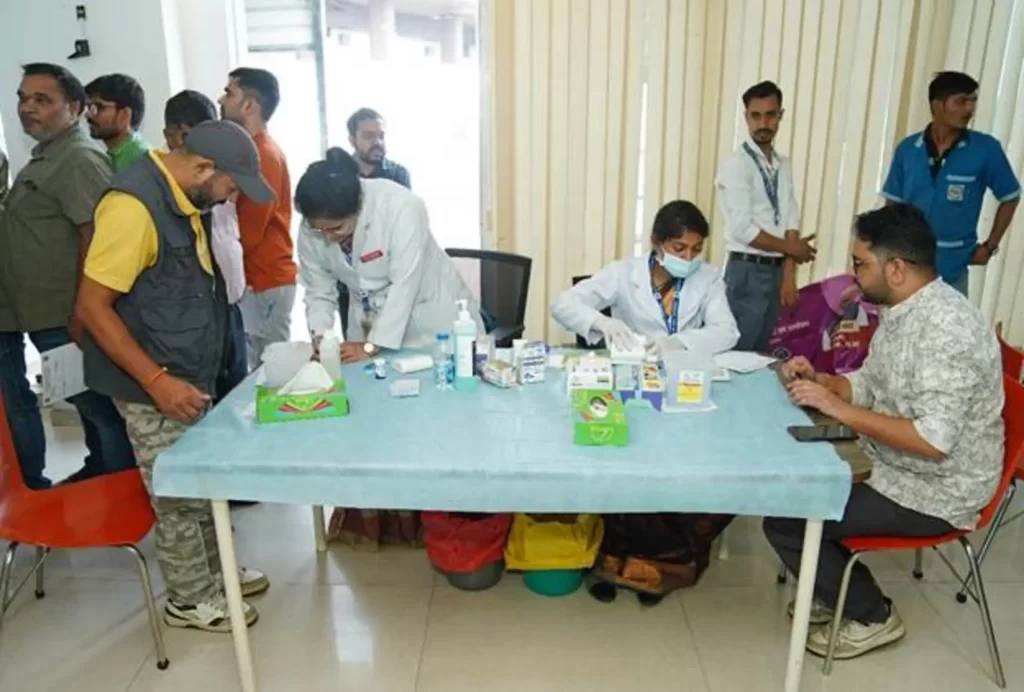
The Press Information Bureau (PIB), in collaboration with the All India Institute of Ayurveda, Delhi (AIIA) under the Ministry of Ayush, and Dr. Ram Manohar Lohia Hospital, New Delhi, under the Ministry of Health and Family Welfare, organized a preventive health check-up and consultation camp for Safai Mitras and PIB officials at Shastri Bhawan and the National Media Centre (NMC) today.
This camp, held as part of the Swachhata Hi Seva Campaign 2025 (observed from 17th September to 2nd October), aimed to promote health, well-being, and service-oriented initiatives.
The integrative health camp featured two streams of medical consultation. Ayurveda services at Shastri Bhawan were led by Dr. Rama Kant Yadava, Additional Professor, and Rajaram Mahto, Associate Professor, Department of Kayachikitsa, All India Institute of Ayurveda, Delhi. Concurrently, allopathy services at the National Media Centre were overseen by Dr. Shailesh Kumar, Professor of Medicine, and Dr. Ramesh Chand Meena, Associate Professor & Nodal Officer from Dr. Ram Manohar Lohia Hospital. Medical teams from both institutions, including consultants and nursing staff, provided comprehensive health assessments and consultations.
Over 50 Safai Mitra and 200 officials availed consultations at the Ayurvedic Health Camp, while approximately 195 officials underwent preventive health check-ups and consultations at the Allopathy Camp.
Leading by example, Principal Director General (PrDG), PIB, Shri Dhirendra Ojha, reached the venue early and was the first to avail consultation at the Ayush camp. His proactive participation, along with that of other senior officials, set a commendable precedent and encouraged wider engagement among staff.
The camp also actively promoted the Swasth Nari Sashakt Parivar Abhiyaan, with enthusiastic participation from female officials and staff, highlighting the government’s focus on women’s health and family well-being.
Ms. Anupama Bhatnagar, Director General, PIB, felicitated the doctors and medical staff from both AIIA and Dr. Ram Manohar Lohia Hospital, presenting them with certificates of appreciation in recognition of their invaluable contributions during the health camp.
This initiative underscores the Government’s unwavering commitment to the holistic well-being of Safai Mitras and PIB personnel by ensuring access to preventive and integrated healthcare services.





Department for Promotion of Industry and Internal Trade (DPIIT), Ministry of Commerce and Industry, has successfully completed the migration of Industrial Entrepreneur Memorandum (IEM) services from the existing G2B Portal to the National Single Window System (NSWS).
This important reform initiative is aimed at furthering the Government of India’s agenda of Ease of Doing Business and providing a seamless digital experience to entrepreneurs and investors across the country.
All services relating to IEM, including IEM Part-A acknowledgement, IEM Part-B filing and IEM Part-A amendments, will now be available exclusively on NSWS. The NSWS platform provides single-point access to multiple approvals and registrations required by businesses. It also enables auto-population of data from government databases to minimize duplication of entries, allows real-time application tracking for greater transparency, and has an integrated payment gateway for convenient online fee submission.
All new IEM applications must be filed only through the NSWS portal (https://www.nsws.gov.in). Applicants must register and create a new profile on NSWS, as earlier G2B portal login credentials will not be valid. Applicants are required to complete the profile on NSWS by entering their entity details such as name, contact details, email, CIN, PAN and authorized signatories.
For IEM Part-A filing, applicants must select the “Industrial Entrepreneur Memorandum (IEM)” service under Central Approvals. They are required to fill in project details such as sector, NIC code, item description, production capacity, investment, employment and annual turnover, along with GST details to fetch the manufacturing unit’s location address, and make the prescribed payment online. The system will generate an e-signed acknowledgement which will be available for download from the dashboard.
After commencement of commercial production, applicants must log in to NSWS with the registered user credentials and submit Part-B with relevant details. They are required to select the IEM Part-B filing option and provide the actual production commencement date, actual production capacity and other details.
Any amendments to IEM Part-A can also be filed through NSWS along with the applicable fees. Applicants must select “IEM Part-A Amendment Application” under Central Approvals and pay the amendment fee. Existing applicants registered on the G2B Portal are required to register themselves afresh on NSWS for IEM acknowledgments. All future filings and amendments must be made through NSWS.
For technical support, applicants may contact the NSWS Helpdesk at nsws@investindia.org.in or toll-free number 1800 102 5841. For any further guidance, they may contact the DPIIT helpdesk at email id: iem-section-dpiit@gov.in or call 011 23061177.
The migration of IEM services to NSWS represents a major milestone in digital governance and investor facilitation, bringing all IEM-related services onto a single-window platform. This step will further enhance the ease of doing business in India and strengthen the country’s position as an attractive global investment destination.
Department of Pharmaceuticals, Government of India has invited applications for research and innovation projects under its Promotion of Research and Innovation in Pharma-MedTech Sector (PRIP) scheme, a landmark initiative to transform the sector into a globally competitive, innovation-driven sector. With an approved outlay of ₹5,000 crore, the scheme is expected to catalyse a Pharma-MedTech innovation pipeline by supporting around 300 projects involving total R&D investment of about ₹11,000 crore in new medicines, complex generics, biosimilars and novel medical devices.
While inviting applications, the Department has notified amendments to the scheme as notified earlier and issued revised guidelines with a view to enhance the impact of the scheme and make it better suited to address the requirements of all stakeholders.
Under the amended scheme, for early stage projects, MSMEs and startups may apply for projects costing up to ₹9 crore for assistance of up to ₹5 crore. For later stage projects, projects of industry, MSMEs and startups costing up to ₹285 crore may apply for assistance up to ₹100 crore. The scale of financial assistance for early stage projects is 100% for cost up to ₹1 crore and 50% of additional cost beyond ₹1 crore, subject to a maximum up to ₹5 crore. The scale for financial assistance for later stage projects is 35% of project cost, subject to a maximum of ₹100 crore.
Further, with a view to strengthen India’s health security framework in areas of high public health significance but relatively lower market potential, identified as Strategic Priority Innovation (SPI) areas, the amended scheme provides that assistance for later stage projects may be to the extent of 50%, subject to a maximum of ₹100 crore. Such SPI areas include specified rare diseases, antimicrobial resistance, vaccine-preventable diseases, tropical vector-borne diseases and outbreak/pandemic-causing pathogens.
Moreover, the amended scheme offers several specific incentives to industry, MSMEs and startups to collaborate with academic and research government institutions of national repute, by providing for preference in selection for up to nine projects each at early and later stages, subject to collaborative development being significant and the collaborative partners/team having strong credentials. Further, industry, MSMEs and startups may use assistance provided under the scheme to in-licence research outputs developed by such institutions, thereby linking the strengths of academia in research with those of industry and startups in developing these into viable technologies and products and taking them to market. In addition, use of funds by an assisted applicant for creating public R&D assets in such institutions as part of their approved project is incentivised under the scheme.
Besides financial assistance, the amended scheme provides for development of strong institutional enablers to further nurture the innovation journey. Dedicated industry-focussed Centres of Excellence at the seven National Institutes of Pharmaceutical Education and Research (NIPERs) will serve as hubs of advanced research, while the NIPER Academia-Industry Coordination Committee recently constituted under Secretary, Department of Pharmaceuticals with joint membership from all industry associations and NIPERs will institutionalise industry-institute linkages in these institutes. Further, the scheme envisions the development of a pan-India digital Pharma-MedTech innovation exchange to connect innovators with investors, mentors, relevant government initiatives such as Patent Mitra and MedTech Mitra and global opportunities, ensuring that innovators are fully supported along their R&D journey from idea to market and a vibrant innovation ecosystem develops.
The application window has opened on 1st October 2025 through a dedicated portal to drive a fully digital application process. Detailed guidelines, eligibility criteria, timelines, instructions etc. are available on the PRIP portal (https://prip.pharma-dept.gov.in). By encouraging participation from both global and domestic players, the scheme seeks to channel greater investment into R&D, while building deeper collaborations across the ecosystem. This forward-looking approach will accelerate delivery of affordable, high-quality solutions, strengthen public health resilience and establish India as a pivotal hub for cutting-edge healthcare innovation. Above all, the scheme is an investment in shaping a healthier and more secure future for India and enhancing its contribution to a healthier world.
The notification, guidelines and website can be accessed through the links
