Dr. Mansukh Mandaviya, Union Minister of Health and Family Welfare along with Dr. Bharti Pravin Pawar, Minister of State for Health and Family Welfare launched the “ICMR/ DHR Policy on Biomedical Innovation & Entrepreneurship for Medical Professionals, Scientists and Technologists at Medical, Dental, Para-Medical Institutes/Colleges", here today.

Acknowledging research and innovation as primary pillars that propel any country towards growth and development in a competitive global canvas, Dr Mansukh Mandaviya said, “It is time that India also demonstrates its strength and mettle through research, entrepreneurship and innovative initiatives in the health sector, including medical devices. Under the leadership and guidance of Hon’ble Prime Minister, India has taken several notable steps towards self reliance and self-sustenance, especially in vaccine development during the pandemic period. I am very hopeful that this Policy of DHR-ICMR unveiled today will motivate, incentivise and give a fillip to all the stakeholders. It will ensure multi-disciplinary collaboration, promote start-up culture and develop an innovation led ecosystem at the medical institutes across the country by promoting Make-in India, Start-Up-India and Atmanirbhar Bharat initiatives of the Government of India.”
The Union Minister stated that “Our medical workforce comprising of doctors, paramedical staff, technicians have a wealth of knowledge based on their experience of working with fundamental problems at the cutting edge level. They also have ideas for innovations. Till now, these could not get a policy framework and platform for further growth. This Policy will connect the industry, technical institutions and promote commercial translation of these ideas and innovations in the health sector. When our philosophy of Seva Bhaav is clubbed with medical expertise and entrepreneurship, I am confident that this will develop a vibrant ecosystem in India, which will not only benefit our citizens but the society and India as a whole.”
Dr. Bharti Pravin Pawar, Minister of State for Health and Family Welfare appreciated the initiative and congratulated DHR and ICMR for bringing out this very important policy document and stated “I strongly believe that this Policy will create innovation and entrepreneurship ecosystem at Medical Colleges/Institutes and will create a pipeline of healthcare innovations including medical device and diagnostic products in India. Wider dissemination and implementation of this policy will catalyze biomedical innovation & entrepreneurship in the country in line with Hon’ble Prime Minister’s vision of New India.” She added that doctors and medical professionals are ideally positioned to be on the forefront of innovation and research.
Prof. Balram Bharagava, Secretary, DHR and Director General, ICMR said “The ICMR/DHR policy on Biomedical Innovation and Entrepreneurship for Medical Professionals is a game changer. It will enable medical institutions to actively support their personnel in contributing towards the innovation and entrepreneurial ventures with the ultimate goal of positively impacting human-health & well-being. It resonates with the motto of our Hon’ble Prime Minister to “Innovate, Patent, Produce and Prosper” I am very hopeful that this policy will bring about a paradigm shift in the innovation and entrepreneurship culture in the country and will have far reaching impact on all the medical colleges and institutes across the country.”
As per the Policy, the medical professionals/ doctors will be encouraged to pursue entrepreneurial ventures by forming start-up companies, taking adjunct position in Company- Non-Executive Director or Scientific Advisor. The doctors will also be permitted to undertake inter-institutional and industry projects alone or through companies, license technologies to business entities leading to commercialisation, revenue generation for self-sustenance and societal benefit. The medical professionals will also be permitted to take-up sabbatical for translational and commercialisation of their innovation through their start-up company set-up following institute’s permission. The Policy will promote interdisciplinary collaboration, innovation, technology development, skill development and foster entrepreneurship development & Make-in-India product development for societal benefit. DHR- ICMR formulated this Policy in consultation with other Government Department/ Ministries/Organisations such as DPIIT, DST, WIPO, DSIR, AIIMS, IIT Delhi etc. This Policy is an attempt to enable medical institutions to actively support their personnel to contribute in innovation and entrepreneurship associated activities.
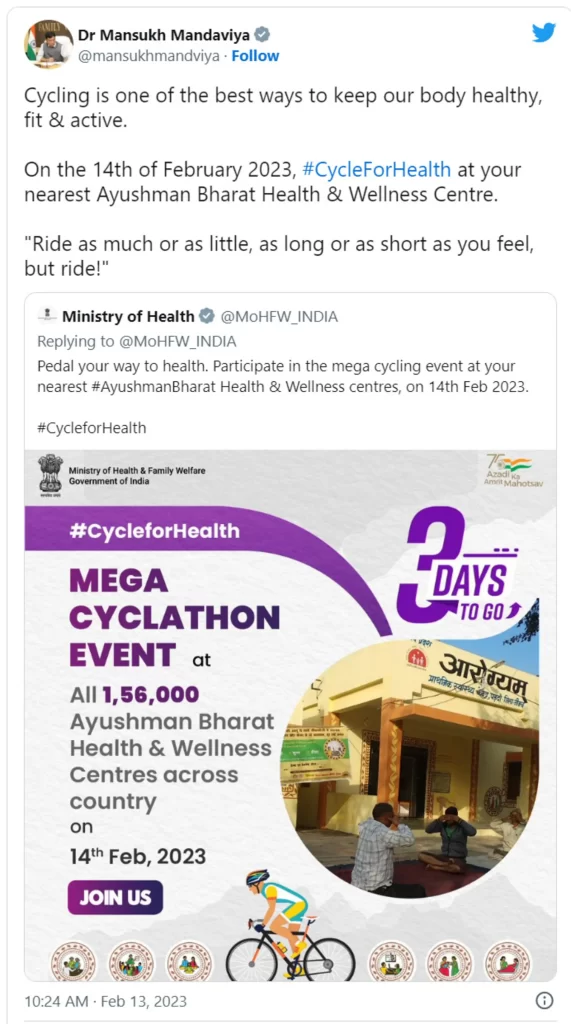
Union Ministry of Health & Family Welfare organises a Cyclathon with the theme, ‘Cycle for Health’ at Lady Hardinge Medical College (LHMC) today. Faculty, staff and students participated with great fervour and in huge numbers. Such rallies are being organised to promote physical and mental well-being and inform our citizens about environment friendly conveyance.
Mega cycling events in the form of Cyclathon, Cycle Rally or Cycle for Health are being undertaken at all 1.56 lakh Ayushman Bharat – Health and Wellness Centres (AB-HWCs) today. These activities are being done as part of the ongoing “Swastha Mann, Swastha Ghar” year-long campaign launched in November last year which aims to promote and enhance awareness surrounding healthy living. In accordance to this, Health Melas will also be organized on 14th of every month at all AB-HWCs across the country where activities such Yoga, Zumba, Teleconsultation, Nikshay Poshan Abhiyan, Non-Communicable Diseases screening and Drug Distribution, Sickle Cell Disease screening will be conducted.
Taking this initiative forward, Union Health Minister Dr. Mansukh Mandaviya who is also known as a “Green MP” for his enthusiasm for cycling, have been urging people to use bicycle to promote health and fitness. Participants at LHMC including younger generation were also encouraged to inculcate healthy practices in their lives to ensure physical and mental benefits. Physical activities help in keeping away many of the non-communicable and life-style diseases.
In a tweet posted by the Union Health Minister yesterday, he encouraged everyone to take part in the initiative.

Glimpses of the mega cycling event from different parts of the country, can be seen below.

As the country celebrates ‘Azadi Ka Amrit Mahotsav’ (AKAM), Government of India is taking various steps to fulfil the vision of ensuring Health and Wellness of all citizens and making it an integral part of our daily lives.
Shri Vishal Chauhan, JS, Dr. (Prof). Atul Goel, DGHS, Dr. Subhash Giri, Director (LHMC), other senior officials of Ministry along with faculty, staff and students of LHMC participated in the mega cycling event.
India's journey toward a malaria-free future is a story of remarkable transformation and progress. At the time of independence in 1947, malaria was one of the most pressing public health challenges, with an estimated 7.5 crore cases annually and 800,000 deaths. Over the decades, relentless efforts have drastically reduced these numbers by over 97%, with cases declining to just 2 million and deaths plummeting to just 83 by 2023. This historic achievement underscores India’s commitment to eliminating Malaria and improving public health for its citizens.
The latest World Malaria Report 2024, released by the World Health Organization (WHO), celebrates India’s significant progress. India’s achievements include a significant reduction in malaria cases and malaria-related deaths between 2017 and 2023. This success is further highlighted by India’s exit from the WHO’s High Burden to High Impact (HBHI) group in 2024, signifying a turning point in its fight against malaria. These achievements reflect the nation’s robust public health interventions and its vision to achieve malaria-free status by 2030.
India’s epidemiological progress is particularly evident in the movement of states to lower disease burden categories. From 2015 to 2023, numerous states have transitioned from the higher-burden category to the significantly lower or zero-burden category. In 2015, 10 States and Union Territories were classified as high burden (Category 3), of these, in 2023 only two states (Mizoram & Tripura) remain in Category 3, whereas 4 states such as Odisha, Chhattisgarh, Jharkhand, and Meghalaya, have reduced the case-load and moved to Category 2. Also, the other 4 States, namely, Andaman & Nicobar Islands, Madhya Pradesh, Arunachal Pradesh, and Dadra and Nagar Haveli have significantly reduced the caseload and moved to Category 1 in 2023. In 2015 only 15 states were in Category 1, whereas in 2023, 24 states and UTs (progressed from high/medium-burden categories to Category 1, reporting an API of less than 1 case per 1000 population). As of 2023 Ladakh, Lakshadweep and Puducherry are in Category 0 i.e. zero indigenous Malaria cases. These areas are now eligible for subnational verification of malaria elimination. Additionally, in 2023, 122 districts across various states reported zero malaria cases, which demonstrates the efficacy of targeted interventions.
Both, Malaria cases and deaths have dropped by around 80% from 2015-2023, with cases going down from 11,69,261 in 2015 to 2,27,564 in 2023, while deaths falling from 384 to just 83. This dramatic decline reflects the relentless efforts to combat the disease. Simultaneously, intensified surveillance efforts have led to a significant rise in the Annual Blood Examination Rate (ABER), increasing from 9.58 (2015) to 11.62 (2023). This strengthened surveillance has ensured early detection, timely intervention, and more effective treatment.
The foundation of India’s success lies in its comprehensive and multi-pronged strategy. The National Framework for Malaria Elimination (NFME), launched in 2016, provided a clear roadmap for achieving zero indigenous malaria cases by 2027. Building on this framework, the National Strategic Plan for Malaria Elimination (2023-2027) introduced enhanced surveillance, prompt case management through a "testing, treating, and tracking" approach, and the development of real-time data tracking through the Integrated Health Information Platform (IHIP).
Integrated Vector Management (IVM) has been at the core of India’s malaria control efforts. Strategies such as Indoor Residual Spraying (IRS) and the distribution of Long-Lasting Insecticidal Nets (LLINs) have significantly curtailed mosquito populations and disrupted the transmission cycle. The targeted management of the invasive Anopheles stephensi mosquito has further bolstered urban malaria control efforts.
The government has also focused on strengthening surveillance and diagnostic capacities. The establishment of National Reference Laboratories (NRLs) at the National Centre of Vector Borne Diseases Control (NCVBDC) has ensured high-quality diagnostic services, while localized action plans for high-endemic districts have enabled tailored interventions. District-specific strategies, particularly for tribal and forested areas, have been instrumental in addressing the unique challenges of these regions.
Community integration has played a vital role in India’s malaria elimination journey. The inclusion of malaria prevention and treatment services in Ayushman Bharat health packages has ensured that even the most vulnerable populations have access to essential healthcare. Community Health Officers and Ayushman Arogya Mandirs have been pivotal in delivering these services at the grassroots level.
India’s commitment to capacity building and research has also been a cornerstone of its success. In 2024 alone, over 850 health professionals were trained through National Refresher Trainings, equipping them with the skills needed for effective malaria control. Research initiatives, including studies on insecticide resistance and therapeutic efficacy, have provided critical data to refine intervention strategies.
Collaborations and funding mechanisms have significantly contributed to India’s progress. The Intensified Malaria Elimination Project-3 (IMEP-3) targets 159 districts across 12 states, focusing on vulnerable populations. Resources are allocated for the LLIN distribution, entomological studies, and surveillance systems, enhancing the impact and sustenance of Malaria elimination activities.
Looking ahead, India remains steadfast in its goal to eliminate Malaria by 2030. The government is committed to achieving zero Indigenous cases by 2027 and ensuring prevention of Malaria re-establishment. By combining strategic frameworks, robust interventions, and community engagement, India is setting a global benchmark in malaria elimination and reaffirming its commitment to public health excellence.
The Hon’ble President of India, Smt. Droupadi Murmu addressed the 6th Convocation Ceremony of Vardhman Mahavir Medical College (VMMC) and Safdarjung Hospital, New Delhi in the presence of Shri J P Nadda, Union Minister of Health and Family Welfare, here today.

Addressing the participants, the President of India said, “Medical profession is unlike any other profession, it involves the noble cause of treating and curing people of diseases and saving lives”. The President said that doctors provide the healing touch to humanity and they can make a difference between life and death. “You have a big responsibility being doctors, because you will be taking care of people's health”, she stated.
Crediting Safdarjung Hospital for treating 10,000 people Safdarjung Hospital every day, she said, “it is a matter of pride that Safdarjung Hospital is among the top 20 medical institutes in the country as per the latest NIRF rankings”.

The President urged the medical fraternity and related institutions to motivate people on the noble cause of organ donation. She also noted the achievements in the healthcare sector during the last 10 years. She said, “the number of medical institutions has increased, and the number of PG seats has doubled. New AIIMS have been established, and undergraduate courses have been introduced in these institutions”. “The recently announced Ayushman Vay Vandana scheme will help catering to the needs of the 70+ elderly population”, she said.

A total of 403 Degrees were awarded to students from DM, MCh, MD, MS, and MBBS programs who successfully completed their training. The President also awarded medals to meritorious students in recognition of their exceptional academic achievements. Additionally, the Annual Report of the institution was released by the distinguished dignitaries.

Speaking on the occasion, Shri J P Nadda congratulated all the students and their parents for their hard work and sacrifice. “This celebratory occasion also marks recognition for the numerous achievements made by the students of VMMC and Safdarjung Hospital which are amongst the most prestigious medical institutes in the country” he stated.

The Union Minister said, “VMMC and Safdarjung Hospital are amongst the busiest medical institute in the country and their medical professionalism is recognised everywhere.”
In his message to the students, the Union Minister stated that, “being doctors does not only entail curing diseases, it also involves understanding humanity”. He further stated that, “excellence, values and empathy are key traits to success.”

Background:
Safdarjung Hospital was founded in 1942 during the Second World War as a Base Hospital for the allied forces. It was taken over by the Government of India in 1954 under the Ministry of Health. It has grown over the years into one of the largest, tertiary level, multi-disciplinary healthcare institutions in Asia. Based on the patient needs and developments in medical care, the hospital has been regularly upgrading its facilities from diagnostic and therapeutic aspects, in all the specialities. The hospital which started with only 204 beds, has grown exponentially to almost 3000 beds. It has an annual OPD attendance of more than 25 lakh patients. The hospital provides medical care to millions of citizens not only of Delhi, but also to people of neighbouring states. This institution has speciality and super speciality departments like Cardiothoracic Surgery (CTVS), Cardiology, Neurology, Neurosurgery, Urology, Nephrology, Pulmonary Medicine, Burns & Plastic Surgery, and Paediatric Surgery which provide state of the art facilities like robotic surgery, sleep laboratory, endoscopies, arthroscopies, video EEG, dialysis, spiral CT, MRI angiography, angioplasty, valve replacement and repairs, coronary artery bypass grafting, lithotripsy, , automated blood Test analysers etc.
Vardhman Mahavir Medical College (VMMC) was established at Safdarjung Hospital by the Government of India in November, 2001. Its inauguration was done by the then Honourable Prime Minister Sh. Atal Bihari Vajpayee on 17th December, 2001 in presence of Sh. L.K. Advani, Sh. C. P. Thakur, (then Health Minister) and Founder Principal Dr. Jagdish Prasad. The College building was dedicated to the Nation by the Vice President Sh. M. Hamid Ansari on 20th Nov. 2007 in the presence of Honourable Health Minister Dr. Anbumani Ramadoss. The first batch of MBBS students was admitted in February, 2002. The (MD/MS) course in Pre and Para clinical subjects were started in year 2011, in addition to the pre-existing clinical courses.
VMMC is affiliated to Guru Gobind Singh Indraprastha University; New Delhi. In a short span of time, VMMC has established itself as a prominent medical institution in India. (‘A Medical marvel’ by as India Today Aspire August 2013, Best Medical College established after 2000, India Today August 24.
The hospital has been a centre for training and teaching of post–graduate students since 1962. In 1973. the hospital faculty constituted the clinical faculty of the University College of Medical Sciences (UCMS) to which it was attached till 1990. Even after UCMS shifted to Guru Teg Bahadur Hospital at Shahdara, Delhi, the hospital continued to impart post-graduate training in various specialties, affiliated to Delhi University. From academic year 2008 onwards, all PG seats have been affiliated to GGSIP University and currently the number stands at 325 -MD/MS and 43 Superspeciality (MCh/DM). seats. The hospital has also been a centre for training of interns from various institutions in the country and abroad, since the eighties.
Smt. Roli Singh, Additional Secretary, Health Ministry; Prof. Geetika Khanna, Principal, VMMC; Dr. Sandeep Bansal, Medical Superintendent, VMMC; senior officers of the Union Government; faculty members, students and staff of VMMC and Safdarjung Hospital were also present on the occasion.
Union Minister of Health and Family Welfare, Shri Jagat Prakash Nadda held a meeting with Chief Ministers/LGs and State Health Ministers of States/UTs, requesting their support toward the ongoing 100-day Intensified TB Elimination Campaign via video conference, here today. Union Minister of State for Health and Family Welfare, Shri Prataprao Jadhav joined the meeting virtually. The Chief Ministers/LGs and Health Ministers were provided an overview of the campaign, its objectives, the key strategic activities being undertaken, and the role of states/UTs in ensuring the successful implementation of the campaign.

State Ministers who were present in the meeting included Shri Yogi Adityanath, Chief Minister, Uttar Pradesh; Dr Mohan Yadav, Chief Minister, Madhya Pradesh; Shri Bhupendra Patel, Chief Minister, Gujarat; Shri Bhajan Lal Sharma, Chief Minister, Rajasthan; Shri ManikSaha, Chief Minister, Tripura; Shri Pushkar Singh Dhami, Chief Minister, Uttrakhand; Shri Satya Kumar Yadav, Health Minister (Andhra Pradesh); Shri BiyuramWahge, Health Minister (Arunachal Pradesh); Shri Ashok Singhal, Health Minister (Assam); Smt. Arti Rao, Health Minister (Haryana); Smt. SakeenaItoo, Health Minister (Jammu & Kashmir); Dr. (Col.) Dhani Ram Shandil, Health Minister (Himachal Pradesh); Shri Vishwajit P. Rane, Health Minister (Goa); Shri Dinesh Gundu Rao, Health Minister (Karnataka); Shri P. Paiwang Konyak, Health Minster (Nagaland); Dr. Mukesh Mahaling, Health Minister (Odisha); Dr. Balbir Singh, Health Minister (Punjab); Smt. Veena George, Health Minister (Kerala); Shri Ma. Subramanian, Health Minister (Tamil Nadu); Shri Irfan Ansari, Health Minister (Jharkhand); Shri DamodarRajanarasimha, Health Minister (Telangana); Smt. Mazel AmpareenLyngdoh, Health Minister (Meghalaya); Shri P Lalrinpuii, Health Minister (Mizoram) and Shri Ramakant Goswami, Labour Minister (Delhi);
Shri Nadda urged the States/UTs to monitor the campaign at the state level and ensure the same is done by the political and administrative leadership at the district levels. He suggested that to ensure a whole-of-government approach, other ministries and departments can be roped in to support campaign activities in the spirit of Jan Bhagidari, similar to the approach taken at the national level. He also requested the Chief Ministers to involve elected representatives, especially Members of Legislative Assemblies and Councils, as well as Panchayati Raj Institutions and encourage their active participation to help mobilize communities.

Shri Nadda highlighted that the rate of TB decline in India from 2015 to 2024 is 17.7% which is double that of the global average of 8.3%. He also informed that deaths due to TB have reduced significantly in India by 21.4% in the last 10 years.
Crediting the state ministers for the achievements made towards TB eradication,the Union Minister thanked them for their support towards the campaign. He highlighted the importance of test, track and diagnostics for active TB testing, screening and diagnosis of patients and urged the dignitaries to actively monitor the campaign in the identified districts in their respective states. Underlining the support that state ministers can provide on the TB campaign, he requested them to promote the campaign in their meetings and rallies and also urged them to come forward as Ni-KshayMitras to adopt TB patients.
The Union Health Minister noted that states already have nearly two months stock of TB medicines and stated that the Centre is working towards ensuring advance stock of at least 6 months of TB medicines available in the states. He concluded his address by encouraging all the stakeholders to reiterate their commitment to end TB.
An overview of the 100-day campaign which aims to reduce TB incidence and mortality due to TB in 347 priority districts across the country was presented at the event. It was informed that to increase detection, intensified case finding drives will be conducted using advanced screening and diagnostic technologies to reduce delays in diagnosis and treatment initiation. Parallelly, to reduce mortality due to TB, the programme will expand access to novel initiatives such as the Differentiated TB Care to provide specialised care for high-risk patients and increased nutritional support through Ni-kshayPoshan Yojana.
The State Ministers were also informed about the various community mobilization activities that should be undertaken during the campaign, including over 80,000 Ni-kshayShivirs to monitor progress. Building on the Jan Bhagidaari approach, the campaign aims to mobilize community members to undertake Ni-kshayShapaths, urge community leaders, individuals, NGOs and corporates to become Ni-kshayMitras. Alongside, TB Vijaetas (TB champions) and Ni-kshayMitras will be recognized for their contributions that will further inspire collective action. Lastly, involvement of Panchayati Raj Institution members will be critical and regular Gram Sabhas on TB will be organized to raise community awareness while ensuring the uptake of essential TB services.

The Chief Ministers expressed their appreciation for the Union Government's intensified efforts in the TB campaign and pledged their support toward achieving the goal of TB elimination. The Chief Minister of Madhya Pradesh shared details about the state's Jan Jagrukta Abhiyan, aimed at raising awareness about TB, and highlighted its leadership role in providing food baskets to TB patients. Chief Ministers from Gujarat and Uttar Pradesh also discussed their progress in enhancing TB testing and screening services. The dignitaries further shared their experiences with the TB campaign and offered valuable feedback and suggestions.
Background:
Since the launch of the campaign, a total of 53 lakh vulnerable individuals have been screened. Out of these, 2.21 lakh individuals have been tested, leading to the diagnosis of 22,178 new patients. Through the NikshayPoshan Yojana, 17,621 beneficiaries have received Direct Benefit Transfers (DBT), and in total, 16 lakh beneficiaries have been paid 416 crore rupees in the year. Additionally, 1,630 new Ni-kshayMitras were registered, collectively adopting over 12,000 patients during the campaign period. Cumulatively, 1.8 lakh Ni-kshayMitras have adopted 8.63 lakh patients. During the campaign, 13,066 food baskets were distributed, and a total of 22 lakh food baskets have been distributed cumulatively.
Smt. Punya Salila Srivastava, Union Health Secretary; Smt. Aradhana Patnaik, Addl. Secretary, Union Health Ministry; senior officers of the Union Health Ministry and Mission Directors (NHM) from stateswere present on the occasion.
Union Ministers of State for Health and Family Welfare, Shri Prataprao Jadhav and Smt. Anupriya Patel sensitized the Members of Parliament from across party lines on the 100-day Intensified TB Elimination Campaign, at the Balayogi Auditorium, Parliament Library building, today. The MPs were informed about the campaign objectives, key activities being undertaken, and their role in supporting the campaign.
Around 250 Members of Parliament were present at the event and pledged to support campaign efforts and drive community mobilization efforts in their constituencies.

Addressing the gathering, the Union Ministers underscored the important leadership roles of the MPs in achieving the goal of TB elimination. They urged the MPs to monitor the 100 days intensified anti -TB campaign in their respective constituencies, raise awareness and reduce stigma around the disease, and mobilize the community to actively participate in the campaign.
The Union Ministers highlighted the government’s successes in reducing TB incidence since 2015, which is more than double the pace of decline observed globally. The MPs were informed about the several novel interventions that have been rolled out in the last ten years, including engagement with the private health care sector, introduction of Ni-kshay Poshan Yojana, the recent doubling of Direct Benefit Transfer support to patients, and the upgradation of diagnostic and treatment capacity of the National TB Elimination Programme. The Ministers also highlighted the central role of the 1.75 lakh Ayushman Arogya Mandirs in taking TB care closer to patients.
Shri Prataprao Jadhav emphasized on the critical role of Panchayats and TB champions in the fight against TB. He noted the importance of further penetration of TB screening services and awareness generation at the ground level for the success of the TB elimination efforts. He encouraged the Hon’ble MPs to support these efforts in order to make it a Jan Andolan.

Smt. Patel highlighted that the Union Government’s efforts in reducing both TB incidence and mortality has been recognized in the recently published WHO Global TB Report. “India has also achieved a significant decline in mortality rate of TB patients from 28% to 22% as well as expanded TB treatment coverage by 32%”, she stated.

Union Health Secretary, Smt. Punya Salila Srivastava underscored the importance of the campaign for reducing the TB burden in the country. She also noted that the recently organized friendly cricket match between the two houses of the Parliament gave a lot of impetus to the National TB elimination mission.
The Additional Secretary and Mission Director, National Health Mission, provided an overview of the 100-day campaign which aims to reduce TB incidence and mortality due to TB in 347 priority districts across the country.
To increase detection, intensified case finding drives will be conducted using advanced screening and diagnostic technologies to reduce delays in diagnosis and treatment initiation. Parallelly, to reduce mortality due to TB, the programme will expand access to novel initiatives such as the Differentiated TB Care to provide specialised care for high-risk patients and increased nutritional support through Ni-kshay Poshan Yojana.
The MPs were informed about their role, including holding regular Ni-kshay Shivirs to monitor progress. Building on the Jan Bhagidaari approach, the MPs were encouraged to mobilize community members to undertake Ni-kshay Shapaths, urge community leaders, individuals, NGOs and corporates to become Ni-kshay Mitras. Alongside, MPs were requested to honour TB Vijaetas (TB champions) and Ni-kshay Mitras for their contributions that will further inspire collective action. Lastly, the MPs were encouraged to work with Panchayati Raj Institution members to work toward TB-free Panchayat certification, organize regular Gram Sabhas on TB, and raise community awareness while ensuring the uptake of essential TB services.

Dr Rajiv Bahl, Secretary, Department of Health Research and DG, ICMR and Dr Atul Goel, Director General for Health Services were also present at the gathering.
National Health Mission (NHM) encompasses its two Sub-Missions, the National Rural Health Mission (NRHM) and the National Urban Health Mission (NUHM). Under NHM, Ministry of Health and Family Welfare provides technical and financial support to the States/UTs to strengthen the public healthcare system towards the objective of providing accessible, affordable and quality healthcare to all those who access public health facilities. Support under NHM is provided to States/ UTs to supplement the efforts of the State/UT governments, based on the requirements posted by them in their Programme Implementation Plans (PIPs) and within their overall resource envelope.
The various initiatives carried out under NHM by the Government of India in various States are operationalisation of Ayushman Arogya Mandir, National Ambulance Services, Mobile Medical Units, ASHAs, 24 x 7 Services and First Referral facilities, Prime Minister's National Dialysis Programme, Free Diagnostics Service Initiative and Free Drugs Service Initiative, various activities under Reproductive & Child Health, Anemia Mukt Bharat (AMB) strategy, Pradhan Mantri TB Mukt Bharat Abhiyaan (PMTBMBA) and Universal Immunization programme.
The target under National Health Policy and current achievements are as below:
Maternal Mortality Ratio reduced from 103 per 100,000 live births in 2017-2019 to 97 per 100,000 live births in 2018-20 (against target of 100 by year 2020), Infant Mortality Rate reduced from 32 per 1000 live births in 2018 to 28 per 1000 live births in 2020 (against target of 28 by year 2019) and Total Fertility Rate is reduced from 2.2 in 2015-16 as per NFHS-4 to 2.0 in 2019-21 as per NFHS-5 (against target of 2.1 by year 2025).
The details of expenditure under NHM incurred on rural and urban healthcare delivery system during the last five years and the current year, State-wise is attached at Annexure.
Pradhan Mantri Jan Arogya Yojana provides financial protection to deprived rural families and identified occupational categories of urban workers’ families. As per PMJAY dashboard, as on 12.12.2024, 36.16 cr. Ayushman Cards have been created for such beneficiaries. Of these, 29.87 Cr. cards have been created for the beneficiaries residing in rural areas. It offers a benefit cover of Rs. 5,00,000 per family per year (on a family floater basis). Services include a range of procedures covering all the costs related to treatment, including drugs, supplies, diagnostic services, physician's fees, room charges, surgeon charges, OT and ICU charges etc.
The Union Minister of State for Health and Family Welfare, Shri Prataprao Jadhav stated this in a written reply in the Rajya Sabha today.
Annexure
State/UT-wise expenditure under NHM incurred on rural and urban healthcare delivery system from FY 2019-20 to FY 2024-25

Note:
1. Expenditure includes expenditure against Central Release, State release & unspent balances at the beginning of the year. Expenditure is as per FMRs submitted by States/UTs and is provisional. The expenditure for 2024-25 w.r.t. flexible pool for RCH, etc. is updated up-to 30.09.2024 except Arunachal Pradesh, Punjab (updated up-to 31.08.2024), Karnataka, Mizoram (updated up-to 31.07.2024) & Meghalaya (updated up-to 30.06.2024).
2. After the reorganization of the State of Jammu and Kashmir (J&K) into the Union Territory of J&K and the Union Territory of Ladakh, NHM funds to the UT of Ladakh were disbursed for the first time during 2020-21.
The Government of India launched the Production Linked Incentive (PLI) Scheme for Promoting Domestic Manufacturing of Medical Devices in 2020, to promote indigenous manufacturing of medical devices. Total financial outlay of the scheme is Rs. 3,420 crore with production tenure from FY 2022-2023 to FY 2026-27. The scheme provides incentive to selected companies at the rate of 5% on incremental sales of medical devices manufactured in India and covered under the four Target segments of the scheme, for a period of five (5) years.
Under the scheme, 19 green-field projects have been commissioned and production of 44 products including high end medical devices such as Linear Accelerator, MRI machines, CT-Scans, Mammograms, C- Arms, Ultrasound machines etc., which were previously imported into the country has started. The cumulative sales made by the applicants under the scheme upto September, 2024 is Rs 8,039.63 crore (which includes exports worth Rs 3,844.01 crore).
Four greenfield projects under the scheme have been established so far in the state of Gujarat. The details of the projects in Annexure.
Annexure

This information was given by the Union Minister of State for Chemicals and Fertilizers Smt Anupriya Patel in Rajya Sabha in a written reply to a question today.
Ayushman Bharat Digital Mission (ABDM) has been launched with an aim to create an online platform enabling interoperability of health data within the health ecosystem to create longitudinal electronic health record of every citizen. The mission aims to develop the backbone necessary to support the integrated digital health infrastructure of the country.
ABDM comprises key registries which intended through building registries such as Ayushman Bharat Health Account (ABHA), healthcare professional registry (HPR), health facility registry (HFR), and drug registry.
ABDM intends to make healthcare more transparent, secure, inclusive, accessible, timely delivery, and most importantly citizen centric.
As on 10th December 2024, a total of 71,16,45,172 (~71.16 Cr) ABHA have been created, 3,54,130 (~3.54 Lac) health facilities have been registered on HFR, 5,37,980 (~5.37 Lac) healthcare professionals have been registered on HPR and 45,99,97,067 (~45.99 Cr) health records have been linked with ABHA.
ABDM is voluntary in nature. Multiple awareness campaigns and efforts are underway to raise awareness about the benefits of ABHA among the public, healthcare providers, and institutions along with capacity building support being extended to hospitals and health systems to integrate ABHA seamlessly into their workflows. As implementing agency, National Health Authority (NHA) has initiated awareness campaigns in healthcare facilities through publicity materials. ABDM actively promotes awareness and citizen engagement through participation in public events such as trade fairs, marathons, medical conferences, technology events etc. encouraging the creation of ABHA and adoption of digital healthcare. States/UTs also undertake targeted IEC activities and capacity building to enhance local adoption. So far, significant progress has been observed in the adoption of ABHA with over 70 crore ABHAs created. Integration with government schemes such as PM-JAY, reproductive child health (RCH), TB Nikshay, national programme for prevention and control of cancer, diabetes, cardiovascular diseases and stroke (NPCDCS) and initiatives such as QR based services for quick OPD registration and faster payments in hospitals, AIIMS and various government hospitals, have enhanced the adoption of digital health accounts. Private healthcare providers are also increasingly adopting ABHA-integrated solutions.
The government recognizes the importance of digital record-keeping in enhancing the healthcare ecosystem and is strengthening digital healthcare infrastructure, ensuring capacity building, and building the necessary digital health solutions and frameworks through ABDM.
The Union Minister of State for Health and Family Welfare, Shri Prataprao Jadhav stated this in a written reply in the Rajya Sabha today.
India’s pharmaceutical market for FY 2023-24 is valued at USD 50 billion with domestic consumption valued at USD 23.5 billion and export valued at USD 26.5 billion. India’s pharma industry is considered to be the world's third largest by volume and 14th in terms of value of production. With an extremely diversified product base covering generic drugs, bulk drugs, over-the-counter drugs, vaccines, biosimilars, and biologics, the Indian pharmaceutical industry has a strong presence at the global level. According to National Accounts Statistics 2024, published by the Ministry of Statistics and Programme Implementation, total output for industry i.e. Pharmaceuticals, medicinal and botanical products is Rs. 4,56,246 crores for FY 2022-23 at constant prices, of which value added is Rs. 1,75,583 crores. 9,25,811 number of persons are engaged in Pharmaceuticals, medicinal and botanical products industry during FY 2022-23.
Research & Development (R&D) and innovation in Pharma Sector is done by number of institutions and organizations under various scientific Ministries/Departments. The Department of Pharmaceuticals has set up seven National Institutes of Pharmaceutical Education & Research (NIPERs) as institutes of national importance, which besides imparting postgraduate and doctorate education, conduct high end research in various pharma specializations. Further, Department has framed a “National Policy on Research & Development and Innovation in Pharma-MedTech Sector in India” to encourage R&D in pharmaceuticals and medical devices and to create an ecosystem for innovation in the sector in order for India to become leader in drug discovery and innovative medical devices through incubating an entrepreneurial environment to build a robust ecosystem to ensure the holistic development of R&D and Innovation. The policy was notified on 18.08.2023.
The policy postulates three main areas of focus to achieve the objectives
To create a regulatory environment that facilitates innovation and research in product development, expanding the traditional regulatory objectives of safety and quality.
To incentivize private and public investment in innovation through a mix of fiscal and non-fiscal measures.
To build an enabling ecosystem designed to support innovation and cross-sectoral research as strong institutional foundation for sustainable growth in the sector.
The details of the policy document may be perused at the Gazette notification dated 18.08.2023 available in the public domain (https://pharmaceuticals.gov.in/policy). The Department has also framed a Scheme for Promotion of Research & Innovation in Pharma Sector (PRIP) with an outlay of Rs. 5000 crore for a period of 5 years i.e. 2023-24 to 2027-28, which was notified on 17.08.2023 with the objective of transforming Indian Pharma MediTech sector from cost-based to innovation-based by strengthening research infrastructure in the country. PRIP Scheme has two components:
Component A: Strengthening the Research Infrastructure by establishing Centres of Excellence (CoEs) in the seven existing National Institutes of Pharmaceutical Education & Research (NIPERs).
Component B: Promotion of Research in Pharma MediTech sector wherein financial assistance will be provided to the companies/projects for both in-house and academic R&D in six specified priority areas. The details of the scheme may be perused at the gazette notification dated 17.08.2023 available in the public domain (https://pharmaceuticals.gov.in/schemes)
Apart from the above, New Drugs and Clinical Trials Rules, 2019 have been notified on 19/03/2019, which contains various provisions for encouraging research and development and innovation of new drugs in the country. Key provisions are: -
• Disposal of clinical trial and new drug applications by way of approval or rejection or seeking further information within a period of 90 days.
• In case of application to conduct clinical trial of a new drug or investigational new drug as part of drug discovery, research and manufacture in India, the application is to be disposed of within a period of 30 days.
• In case of Clinical Trials application, if no communication is received from CDSCO within the prescribed timelines, the application will be deemed to have been approved.
• Provisions for accelerated/ expedited approval process in certain situation like unmet need, orphan drugs for rare diseases etc.
• Provisions for pre-submission and post submission meetings of the applicants with CDSCO for formal discussion and case specific regulatory pathway.
For export, the drug is required to be manufactured under manufacturing license as per provision of Drugs and Cosmetics Act and Rules thereunder. Further, the manufacturer is required to comply with the requirements of importing country.
As informed by the Department for Promotion of Industry & Internal Trade (DPIIT), Government has not taken any specific initiative especially for Pharmaceutical industry to strengthen their Intellectual property regime. However, several initiatives irrespective of the industry type has been undertaken to strengthen the Intellectual Property regime. The main initiatives undertaken in this regard are outlined below:-
1. Amendment of Rules i.e. Patents, Geographical Indication, The Trademark, Design and Copyright which are as under: -
Patents rules : Since 2014, Patents Rules have been amended several times to streamline and simplify filing and processing of patent applications, remove irregularities, redress procedural delays, and complexities in patent granting procedure, streamline the use of IT and digital technologies and provide certain benefits to the sectors that are critical for Indian economy. Some of the key changes made are viz: fee rebates of at least 80% for filing and processing of patent applications and maintenance of patents have been given to Startups, small entities (MSMEs) and educational institutes, facility of expedited examination has been given to Startups, small entities (MSMEs), applicants electing India an authority for international applications, female applicants, government institutions/ departments, electronic submission of documents by patent agents has been made mandatory and timelines have been streamlined, requirements for filing of a priority document and form 27 (statement regarding working of patents) have been streamlined, the time to submit request for examination has been reduced to 31 months from 48 months to fast-track the patent examination process and 10% reduction in the official fee for patent renewal is available if the fees for at least four years are paid in advance through electronic mode etc.
Start-Ups Intellectual Property Protection (SIPP) Scheme - “Start-Ups Intellectual Property Protection (SIPP)” scheme facilitates protection of Patents, Trademark and Designs by interested Startups, and all Indian innovators/ creators, educational institutes using the services of the Technology and Innovation Support Centers (TISCs) established in India. The Scheme was started in 2016 and has been further extended up to 31-03-2026. As on date, there are more than 2700 empanelled facilitators to help start-ups to file applications. The scheme has been revised recently and facilitation fees have been notably increased by at least 100%. The revised applicable fee structure is as under:

This information was given by the Union Minister of State for Chemicals and Fertilizers Smt Anupriya Patel in Rajya Sabha in a written reply to a question today.
