Union Minister of State for Health and Family Welfare Smt. Anupriya Patel today participated in a session on the theme “Innovation to Impact: AI as a Public Health Game-Changer” during the India AI Impact Summit 2026 at Bharat Mandapam. The session spotlighted the transformative role of Artificial Intelligence in advancing public health outcomes and strengthening India’s healthcare delivery systems.
Addressing policymakers, healthcare leaders, technology experts, researchers, and industry stakeholders while delivering the keynote address, Smt. Anupriya Patel, Hon’ble Minister of State for Health and Family Welfare, highlighted that “AI for India, as our Hon’ble Prime Minister Shri Narendra Modi envisions, is not merely Artificial Intelligence but All-Inclusive Intelligence.” She emphasized that when India speaks of AI in healthcare, it is not limited to sophisticated algorithms or the promise of precision alone, but is measured by the extent to which technology touches lives and addresses health inequities across the country.

She stated that as India advances towards the vision of a Viksit Bharat by 2047, health forms one of the most critical pillars of development. India’s vast and diverse population, the rural–urban divide, and the dual burden of communicable and non-communicable diseases present unique challenges. In such a context, she underscored, technology—particularly AI—becomes an indispensable enabler.
She also noted that AI has been integrated across the entire continuum of healthcare—from disease surveillance and prevention to diagnosis and treatment. She highlighted the Media Disease Surveillance System, an AI-enabled tool that monitors disease trends in as many as 13 languages, generates real-time alerts, and strengthens outbreak preparedness. This system, she said, showcases the power of AI in augmenting India’s disease control efforts and enhancing surveillance capacity.

Under the One Health Mission, she further informed that the Indian Council of Medical Research has launched AI-based tools for genomic surveillance, capable of predicting potential zoonotic outbreaks even before transmission from animals to humans occurs. Such predictive capabilities, she emphasized, represent a paradigm shift in preventive public health.
She also highlighted the deployment of AI-enabled handheld X-ray machines and Computer-Aided Detection tools for tuberculosis (CA-TB), which have brought advanced diagnostics closer to communities. These innovations have contributed to approximately 16 percent additional case detection in TB. Furthermore, AI-based tools predicting adverse TB treatment outcomes have helped achieve a 27 percent decline in negative treatment results, strengthening India’s fight against tuberculosis.
Emphasizing scalability and affordability, Smt. Patel stated that in a large population, resource-constrained setting like India, solutions must be scalable, frugal, and capable of addressing systemic gaps. She noted that the Government has actively worked towards building a strong AI ecosystem in healthcare, including the establishment of three Centres of Excellence for AI at AIIMS Delhi, PGIMER Chandigarh, and AIIMS Rishikesh to integrate world-class AI expertise into public healthcare delivery.

Clarifying the role of technology, she asserted that AI is here to augment and assist, not to replace clinicians. By reducing the burden of routine and high-intensity tasks, AI enables doctors to devote more time to complex cases and critical clinical decision-making.
“Medicine is not only a science; it is also an art,” she remarked. Healthcare thrives not merely on algorithms but on human touch, empathy, compassion, and communication—qualities that cannot be replicated by machines and will always remain the domain of clinicians.
Concluding her address, the Minister stressed that future-ready healthcare professionals must be AI-literate. In this regard, she noted that the National Board of Examinations in Medical Sciences has recently launched an online training programme on AI in healthcare to equip doctors across the country with essential digital competencies, ensuring that India’s medical workforce remains prepared for a technology-driven future.
Addressing the summit, Prof. V. K. Paul, Member (Health), NITI Aayog, emphasized that Artificial Intelligence presents a strategic opportunity to transform India’s healthcare landscape and accelerate progress towards universal health coverage. He noted that given India’s scale, diversity, and dual burden of communicable and non-communicable diseases, technology-driven, evidence-based interventions are essential to strengthen service delivery and improve health outcomes.
He highlighted that AI can significantly enhance primary healthcare, enable early diagnosis, strengthen disease surveillance, and support data-driven policy formulation. Integrating AI with India’s growing digital public health infrastructure, he stated, will ensure interoperability, real-time analytics, and more efficient resource allocation across the health system.
Prof. Paul further underscored the importance of robust regulatory frameworks, ethical safeguards, and continuous validation to maintain safety and public trust. He called for sustained collaboration between government, academia, and industry to develop scalable, affordable, and indigenous AI solutions capable of delivering measurable impact at population scale.
Speaking at the Occasion, Shri Roy Jakobs, Chief Executive Officer of Royal Philips, stated that AI will have its greatest impact in the field of healthcare. He observed that health systems across the globe are under immense pressure due to rising demand, workforce shortages, and increasing complexity of care, making the integration of AI not just an opportunity but a necessity.

He emphasized that AI alone cannot transform healthcare; it must be supported by robust data governance, seamless data handling, and strong clinical integration. “Technology must align with clinical needs and workflows,” he noted, underlining that meaningful AI deployment requires quality data, interoperability, and clearly defined use cases.
He further stressed that healthcare runs on trust, and therefore AI systems must be transparent, explainable, and continuously validated to maintain clinical confidence and patient safety.
Commending India’s digital health initiatives, he noted that programmes such as Ayushman Bharat Yojana are laying the groundwork for interoperable data systems and continuity of care at population scale—precisely the kind of foundation AI requires to deliver meaningful and sustainable impact.
He also remarked that solutions built in India are increasingly being deployed globally, demonstrating that technologies designed for scale, diversity, and complexity tend to be resilient and adaptable worldwide. Reaffirming Royal Philips’ commitment to collaborative innovation, Shri Jakobs expressed confidence that partnerships between government and industry will accelerate AI-driven transformation and improve health outcomes globally.

The overall discussion highlighted that Artificial Intelligence is poised to become a transformative force in public health, provided it is deployed responsibly, ethically, and at scale. Speakers collectively emphasized that AI must move beyond pilot projects to system-level integration, supported by interoperable digital infrastructure, quality data, strong regulatory frameworks, and public-private collaboration. The deliberations underscored that while AI can significantly enhance disease surveillance, diagnostics, clinical decision-making, and health system efficiency, it is ultimately a tool to augment—not replace—clinicians. The session concluded with a shared commitment to harness AI as an inclusive, scalable, and patient-centric solution to address India’s complex healthcare challenges and to strengthen global health resilience.
Basel, 17 February 2026 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the appointment of Mark Dawson, M.D., Ph.D., as the new Head of Roche Pharma Research and Early Development (pRED), effective 1 May 2026. Based in Basel, he will also become a member of the Enlarged Corporate Executive Committee.
Roche CEO Thomas Schinecker commented:
"Mark Dawson is a distinguished physician-scientist with a profound dedication to scientific excellence and patient care. His collaborative spirit and thoughtful leadership make him the ideal choice to lead pRED, and I am delighted to welcome him to our team."
Mark Dawson joins Roche from the Peter MacCallum Cancer Centre, where he serves as Associate Director of Research. A leading expert in cancer biology, his work on chromatin regulation and epigenetics has been instrumental in defining the molecular mechanisms that drive cancer initiation and progression.
In addition to his clinical and research leadership, he holds a professorial appointment at the University of Melbourne and is an elected member of the Australian Academy of Science, the Australian Academy of Health and Medical Sciences, and the European Molecular Biology Organisation (EMBO).
Mark earned his medical degree from the University of Melbourne in 1999, followed by specialist training in haematology and a Ph.D. from the University of Cambridge. His career is marked by prestigious international honors, including:
His extensive background as a clinician-researcher and his history of academic excellence position him to drive the next generation of innovative therapies at Roche pRED.
About Roche
Founded in 1896 in Basel, Switzerland, as one of the first industrial manufacturers of branded medicines, Roche has grown into the world’s largest biotechnology company and the global leader in in-vitro diagnostics. The company pursues scientific excellence to discover and develop medicines and diagnostics for improving and saving the lives of people around the world. We are a pioneer in personalised healthcare and want to further transform how healthcare is delivered to have an even greater impact. To provide the best care for each person we partner with many stakeholders and combine our strengths in Diagnostics and Pharma with data insights from the clinical practice.
For over 125 years, sustainability has been an integral part of Roche’s business. As a science-driven company, our greatest contribution to society is developing innovative medicines and diagnostics that help people live healthier lives. Roche is committed to the Science Based Targets initiative and the Sustainable Markets Initiative to achieve net zero by 2045.
Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan.
For more information, please visit www.roche.com.
All trademarks used or mentioned in this release are protected by law.
Roche Global Media Relations
Phone: +41 61 688 8888 / e-mail: media.relations@roche.com
| Hans Trees, PhD Phone: +41 79 407 72 58 | Nathalie Altermatt Phone: +41 79 771 05 25 |
| Lorena Corfas Phone: +41 79 568 24 95 | Simon Goldsborough Phone: +44 797 32 72 915 |
| Karsten Kleine Phone: +41 79 461 86 83 | Kirti Pandey Phone: +49 172 6367262 |
| Yvette Petillon Phone: +41 79 961 92 50 | Dr Rebekka Schnell Phone: +41 79 205 27 03 |
Attachment
Basel, 16 February 2026 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that the phase III MAJESTY study in adults with primary membranous nephropathy met its primary endpoint, showing statistically significant and clinically meaningful results with Gazyva®/Gazyvaro® (obinutuzumab). Results show that significantly more people achieved complete remission at two years (104 weeks) with Gazyva/Gazyvaro versus tacrolimus. Safety was in line with the well-characterised profile of Gazyva/Gazyvaro and no new safety signals were identified.
“These results demonstrate that Gazyva/Gazyvaro may help more people with primary membranous nephropathy achieve complete remission, maintain kidney function for longer and delay or potentially prevent the onset of life-threatening complications,” said Levi Garraway, MD, PhD, Roche’s Chief Medical Officer and Head of Global Product Development. “If approved, Gazyva/Gazyvaro would be the first therapy specifically indicated for people with primary membranous nephropathy, where there are limited treatment options.”
Analysis of key secondary endpoints showed statistically significant and clinically meaningful benefits with Gazyva/Gazyvaro versus tacrolimus in overall remission (complete or partial remission) at week 104 and complete remission at week 76.
Data will be presented at an upcoming medical meeting and shared with health authorities including the US Food and Drug Administration and the European Medicines Agency.
Primary membranous nephropathy is a chronic autoimmune condition that causes potentially irreversible kidney damage and reduced kidney function, and it is estimated that it affects nearly 88,000 people in the EU and over 96,000 in the US. Up to 30% of people with primary membranous nephropathy will develop kidney failure over 10 years, which requires invasive intervention like dialysis or transplant and has a significant impact on patients and their families, as well as carrying substantial cost to health systems.1,2 Gazyva/Gazyvaro has the potential to address this by targeting an underlying cause of the condition, which may help maintain kidney function for longer and prevent the onset of life-threatening complications.
MAJESTY is the fourth positive phase III study of Gazyva/Gazyvaro in immune-mediated diseases, following REGENCY in lupus nephritis, ALLEGORY in systemic lupus erythematosus and INShore in idiopathic nephrotic syndrome. This growing body of evidence supports Gazyva/Gazyvaro’s potential in addressing disease activity across a spectrum of immune-mediated diseases.
Gazyva/Gazyvaro is approved in the US and EU for the treatment of adults with active lupus nephritis based on data from the REGENCY and NOBILITY studies and is being investigated in a global phase II study of children and adolescents with lupus nephritis.3,4 Beyond Gazyva/Gazyvaro, we have a broad pipeline as part of our ambition to be leaders in immunology, in particular in immune-mediated and kidney-related diseases.
About Gazyva/Gazyvaro
Gazyva®/Gazyvaro® (obinutuzumab) is a humanised monoclonal antibody designed with a Type II anti-CD20 region, for direct B cell death and a glycoengineered Fc region, for higher binding affinity and increased antibody-dependent cellular cytotoxicity (ADCC). CD20 is a protein found on certain types of B cells.
Gazyva/Gazyvaro is approved for adults with lupus nephritis in the US and EU. Gazyva/Gazyvaro is also approved in 100 countries for various types of haematological cancers.
About the MAJESTY study
MAJESTY [NCT04629248] is a phase III, randomised, open-label, multicentre study designed to evaluate the efficacy and safety of Gazyva®/Gazyvaro® (obinutuzumab) in people with primary membranous nephropathy. The study enrolled 142 people who were randomised 1:1 to receive Gazyva/Gazyvaro or tacrolimus. The primary endpoint is the percentage of people who achieve complete remission at two years (week 104).
About primary membranous nephropathy
Primary membranous nephropathy is a chronic autoimmune condition where the body’s immune system attacks the filtering units of the kidney, the glomeruli, causing protein to leak into the urine and potentially a gradual decline in kidney function. Over time, the damage to the kidneys can become irreversible, increasing the risk of life-threatening complications, such as kidney failure, idiopathic nephrotic syndrome, blood clots and cardiovascular disease. Achieving complete remission is critical to help maintain kidney function and delay or prevent the onset of serious and potentially fatal complications.
About Roche in kidney and kidney-related diseases
For more than 20 years, we have combined innovation, scientific expertise and commitment to patients to address unmet needs in kidney diseases. Today, our industry-leading programme includes Gazyva®/Gazyvaro® (obinutuzumab), approved in the US and EU for adults with active lupus nephritis, and more than 10 phase II-III clinical studies in immune-mediated kidney and kidney-related diseases with some of the highest unmet needs.
Our aim is to continue delivering meaningful value for those affected, healthcare systems and society, and help address this growing public health burden.
About Roche
Founded in 1896 in Basel, Switzerland, as one of the first industrial manufacturers of branded medicines, Roche has grown into the world’s largest biotechnology company and the global leader in in-vitro diagnostics. The company pursues scientific excellence to discover and develop medicines and diagnostics for improving and saving the lives of people around the world. We are a pioneer in personalised healthcare and want to further transform how healthcare is delivered to have an even greater impact. To provide the best care for each person we partner with many stakeholders and combine our strengths in Diagnostics and Pharma with data insights from the clinical practice.
For over 125 years, sustainability has been an integral part of Roche’s business. As a science-driven company, our greatest contribution to society is developing innovative medicines and diagnostics that help people live healthier lives. Roche is committed to the Science Based Targets initiative and the Sustainable Markets Initiative to achieve net zero by 2045.
Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan.
For more information, please visit www.roche.com.
All trademarks used or mentioned in this release are protected by law.
References
[1] Keri KC, et al. Primary membranous nephropathy: comprehensive review and historical perspective. Postgrad Med L. 2019 Jan;95(1119). doi: 10.1136/postgradmedj-2018-135729.
[2] Kanigicherla DAK, et al. Long-term outcomes of persistent disease and relapse in primary membranous nephropathy. Nephrol Dial Transplant. 2016 Dec:31(12):2108-2114. doi: 10.1093/ndt/gfv435. Epub 2016 Jan 13.
[3] Furie RA, et al. B-cell depletion with obinutuzumab for the treatment of proliferative lupus nephritis: a randomised, double-blind, placebo-controlled trial. Ann Rheum Dis. 2022 Jan;81(1):100-07.
[4] Furie RA, et al. Efficacy and safety of obinutuzumab in active lupus nephritis. N Engl J Med. 2025 Feb;392:1471-83.
Roche Global Media Relations
Phone: +41 61 688 8888 / e-mail: media.relations@roche.com
| Hans Trees, PhD Phone: +41 79 407 72 58 | Nathalie Altermatt Phone: +41 79 771 05 25 |
| Lorena Corfas Phone: +41 79 568 24 95 | Simon Goldsborough Phone: +44 797 32 72 915 |
| Karsten Kleine Phone: +41 79 461 86 83 | Kirti Pandey Phone: +49 172 6367262 |
| Yvette Petillon Phone: +41 79 961 92 50 | Dr Rebekka Schnell Phone: +41 79 205 27 03 |
Roche Investor Relations
| Dr Bruno Eschli Phone: +41 61 68-75284 e-mail: bruno.eschli@roche.com | Dr Sabine Borngräber Phone: +41 61 68-88027 e-mail: sabine.borngraeber@roche.com |
| Dr Birgit Masjost Phone: +41 61 68-84814 e-mail: birgit.masjost@roche.com |
Investor Relations North America
| Loren Kalm Phone: +1 650 225 3217 e-mail: kalm.loren@gene.com |
Attachment
Table 1: Trade during January 2026*
| January 2026(US$ Billion) | January 2025(US$ Billion) | ||
| Merchandise | Exports | 36.56 | 36.34 |
| Imports | 71.24 | 59.77 | |
| Services* | Exports | 43.90 | 34.75 |
| Imports | 19.60 | 16.71 | |
| Total Trade(Merchandise +Services) * | Exports | 80.45 | 71.09 |
| Imports | 90.83 | 76.48 | |
| Trade Balance | -10.38 | -5.39 |
* Note: The latest data for services sector released by RBI is for December 2025. The data for January 2026 is an estimation. (ii) Data for April-January 2024-25 and April-September 2025 has been revised on pro-rata basis using quarterly balance of payments data.
Fig 1: Total Trade during January 2026*
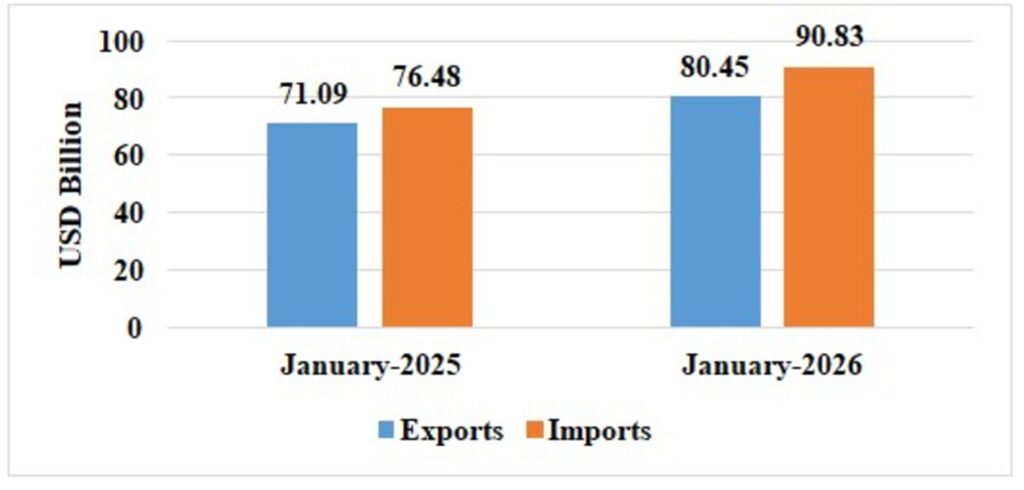
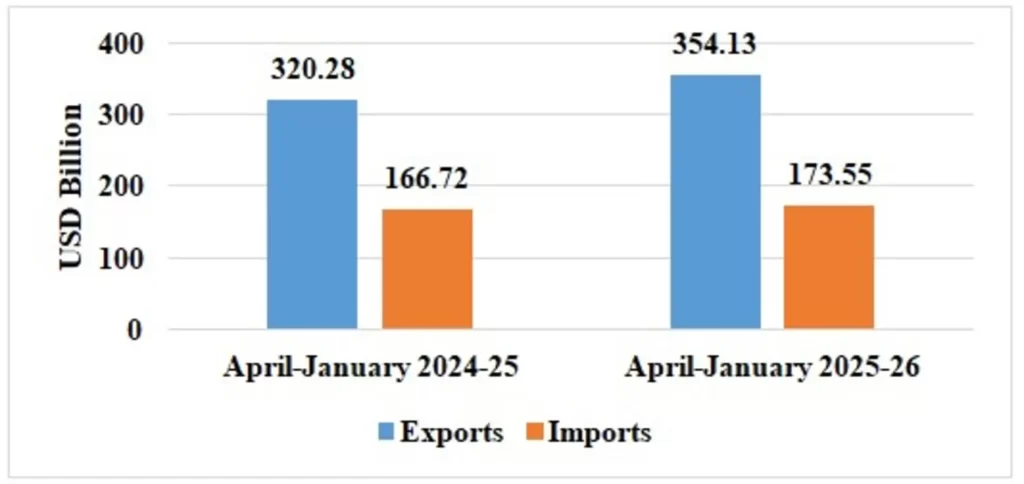
Table 2: Trade during April-January 2025-26*
| April-January 2025-26(US$ Billion) | April-January 2024-25(US$ Billion) | ||
| Merchandise | Exports | 366.63 | 358.75 |
| Imports | 649.86 | 606.13 | |
| Services* | Exports | 354.13 | 320.28 |
| Imports | 173.55 | 166.72 | |
| Total Trade(Merchandise +Services) * | Exports | 720.76 | 679.02 |
| Imports | 823.41 | 772.85 | |
| Trade Balance | -102.65 | -93.83 |
Fig 2: Total Trade during April-January 2025-26*

MERCHANDISE TRADE
Fig 3: Merchandise Trade during January 2026

Fig 4: Merchandise Trade during April-January 2025-26
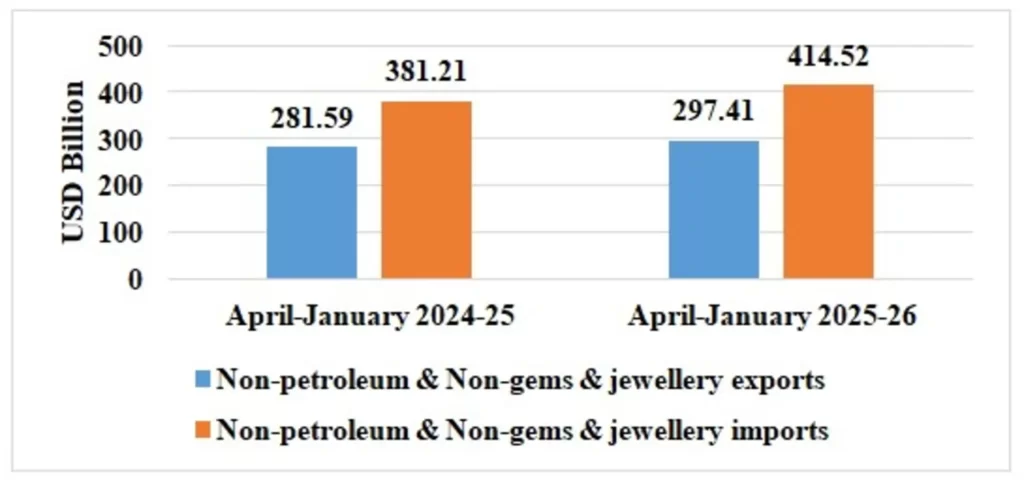
Table 3: Trade excluding Petroleum and Gems & Jewellery during January 2026
| January 2026(US$ Billion) | January 2025 (US$ Billion) | |
| Non- petroleum exports | 32.78 | 32.86 |
| Non- petroleum imports | 57.83 | 46.33 |
| Non-petroleum & Non-Gems & Jewellery exports | 30.47 | 29.86 |
| Non-petroleum & Non-Gems & Jewellery imports | 42.56 | 41.53 |
Note: Gems & Jewellery Imports include Gold, Silver & Pearls, precious & Semi-precious stones
Fig 5: Trade excluding Petroleum and Gems & Jewellery during January 2026

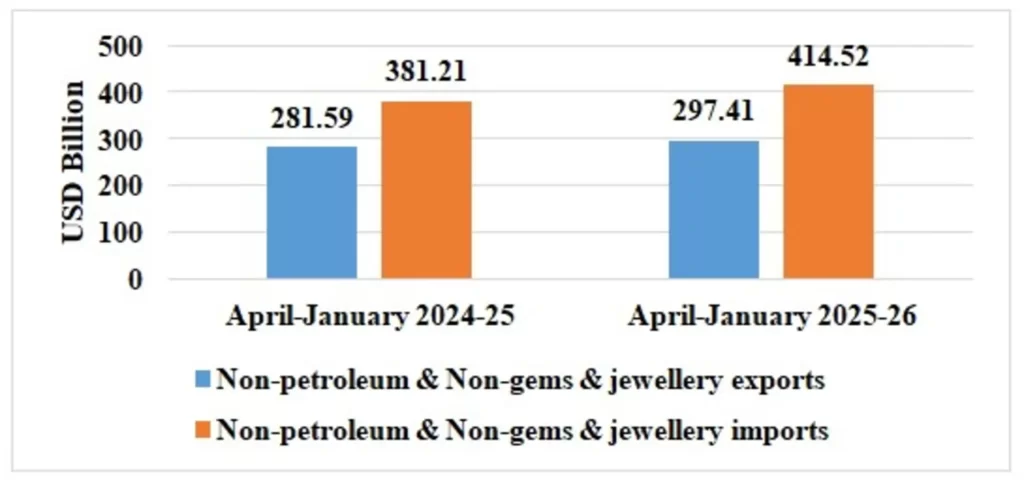
Table 4: Trade excluding Petroleum and Gems & Jewellery during April-January 2025-26
| April-January 2025-26(US$ Billion) | April-January 2024-25(US$ Billion) | |
| Non- petroleum exports | 320.94 | 305.98 |
| Non- petroleum imports | 501.04 | 451.25 |
| Non-petroleum & Non Gems & Jewellery exports | 297.41 | 281.59 |
| Non-petroleum & Non Gems & Jewellery imports | 414.52 | 381.21 |
Note: Gems & Jewellery Imports include Gold, Silver & Pearls, precious & Semi-precious stones
Fig 6: Trade excluding Petroleum and Gems & Jewellery during April-January 2025-26
SERVICES TRADE
Fig 7: Services Trade during January 2026*
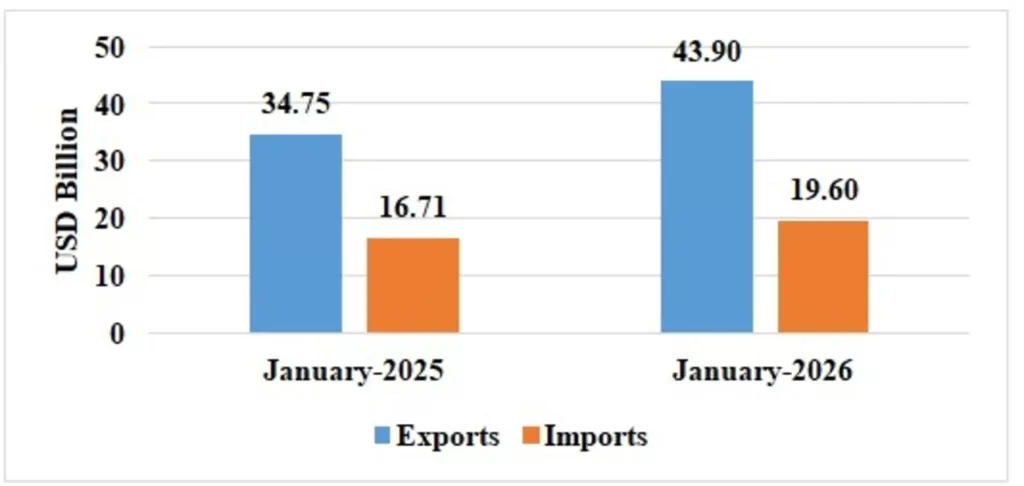
Fig 8: Services Trade during April-January 2025-26*
The Union Minister of Health and Family Welfare, Shri Jagat Prakash Nadda will launch two key national initiatives — the Strategy for Artificial Intelligence in Healthcare for India (SAHI) and the Benchmarking Open Data Platform for Health AI (BODH) at the India AI Summit at Bharat Mandapam, tomorrow.

SAHI is a national guidance framework to enable the safe, ethical, evidence-based, and inclusive adoption of Artificial Intelligence across India’s healthcare system. It aims to provide strategic direction on governance, data stewardship, validation, deployment, and monitoring of AI solutions, while supporting States and institutions in responsible adoption aligned with public health priorities.
BODH, developed by the Indian Institute of Technology Kanpur in collaboration with the National Health Authority, is a privacy-preserving benchmarking platform that enables rigorous evaluation of AI models using diverse, real-world health data without sharing underlying datasets. As a digital public good under the Ayushman Bharat Digital Mission, it is designed to strengthen trust, transparency, and quality assurance in Health AI deployment.
Together, SAHI and BODH mark a significant step in India’s journey towards building a robust, responsible, and globally competitive health AI ecosystem.
The annual rate of inflation based on All India Wholesale Price Index (WPI) number is 1.81% (provisional) for the month of January, 2026 (over January, 2025). Positive rate of inflation in January, 2026 is primarily due to increase in prices of manufacture of basic metals, other manufacturing, non-food articles, food articles and textiles etc. The index numbers and inflation rate for the last three months of all commodities and WPI components are given below:
| Index Numbers and Annual Rate of Inflation (Y-o-Y in %) * | |||||||
| All Commodities/Major Groups | Weight (%) | Nov-25(F) | Dec-25(P) | Jan-26(P) | |||
| Index | Inflation | Index | Inflation | Index | Inflation | ||
| All Commodities | 100.00 | 156.2 | -0.13 | 157.0 | 0.83 | 157.8 | 1.81 |
| I. Primary Articles | 22.62 | 192.9 | -2.53 | 194.2 | 0.21 | 193.9 | 2.21 |
| II. Fuel & Power | 13.15 | 146.4 | -2.33 | 148.3 | -2.31 | 145.9 | -4.01 |
| III. Manufactured Products | 64.23 | 145.2 | 1.47 | 145.6 | 1.82 | 147.5 | 2.86 |
| Food Index | 24.38 | 195.2 | -2.50 | 196.0 | 0.00 | 194.2 | 1.41 |
Note: F: Final, P: Provisional, *Annual rate of WPI inflation is calculated over the corresponding month of previous year
The month over month change in WPI for the month of January, 2026 stood at 0.51% as compared to December, 2025. The monthly change in WPI for last six-month is summarized below:
| Month Over Month (M-o-M in %) change in WPI Index# | |||||||
| All Commodities/Major Groups | Weight | Aug-25 | Sep-25 | Oct-25 | Nov-25(F) | Dec-25(P) | Jan-26(P) |
| All Commodities | 100.00 | 0.52 | -0.13 | 0.06 | 0.71 | 0.51 | 0.51 |
| I. Primary Articles | 22.62 | 1.33 | -0.84 | -0.37 | 2.23 | 0.67 | -0.15 |
| II. Fuel & Power | 13.15 | -0.14 | -0.07 | 1.26 | 0.83 | 1.30 | -1.62 |
| III. Manufactured Products | 64.23 | 0.28 | 0.14 | 0.07 | -0.07 | 0.28 | 1.30 |
| Food Index | 24.38 | 1.10 | -0.72 | -0.05 | 1.67 | 0.41 | -0.92 |
Note: F: Final, P: Provisional, #Monthly rate of change, based on month over month (M-o-M) is calculated over the preceding month
Month-over-Month Change in Major Groups of WPI:
WPI Food Index (Weight 24.38%): The Food Index consisting of 'Food Articles' from Primary Articles group and 'Food Product' from Manufactured Products group have decreased from 196.0 in December, 2025 to 194.2 in January, 2026. The rate of inflation (Y-o-Y) based on WPI Food Index increased to 1.41% in January, 2026.
Final Index for the month of November, 2025 (Base Year: 2011-12=100): For the month of November, 2025, the final Wholesale Price Index and inflation rate for 'All Commodities' (Base: 2011-12=100) stood at 156.2 and (-) 0.13% respectively.
The details of all India Wholesale Price Indices and Rates of Inflation for different commodity groups based on updated figures are at Annex I. The Annual rate of Inflation (Y-o-Y) based on WPI for different commodity groups in the last six months is at Annex II. WPI for different commodity groups in the last six months is at Annex III.
Response Rate: The WPI for January, 2026 has been compiled at a weighted response rate of 84.2 per cent, while the final figure for November, 2025 is based on the weighted response rate of 93.4 per cent. The provisional figures of WPI will undergo revision as per the revision policy of WPI. This
press release, item indices, and inflation numbers are available at our website https://eaindustry.nic.in.
Next date of Press Release: WPI for the month of February, 2026 would be released on 16/03/2026.
Note: DPIIT releases index number of wholesale price in India on monthly basis on 14th of every month (or next working day, if 14th falls on holiday) with a time lag of two weeks of the reference month, and the index number is compiled with data received from institutional sources and selected manufacturing units across the country. This press release contains WPI (Base Year 2011-12=100) for the month of January, 2026 (Provisional), November, 2025 (Final) and other months/years. Provisional figures of WPI are finalised after 10 weeks (from the month of reference), and frozen thereafter.
Annex-I
All India Wholesale Price Indices and Rates of Inflation (Base Year: 2011-12=100) for January, 2026
| Commodities/Major Groups/Groups/Sub-Groups/Items | Weight | IndexJan-26* | Latest month over Month (MoM) | Inflation (YoY) | Rate of Inflation (YoY) | |||
| Dec-Jan 2024-25 | Dec-Jan2025-26* | 2024-25 (Apr-Jan) | 2025-26 (Apr-Jan) | January 2025 | January2026* | |||
| ALL COMMODITIES | 100.00 | 157.8 | -0.45 | 0.51 | 2.26 | 0.24 | 2.51 | 1.81 |
| I. PRIMARY ARTICLES | 22.62 | 193.9 | -2.12 | -0.15 | 5.80 | -2.22 | 4.58 | 2.21 |
| A. Food Articles | 15.26 | 202.9 | -3.71 | -1.79 | 8.27 | -3.12 | 5.83 | 1.55 |
| Cereals | 2.82 | 209.3 | 0.43 | 0.19 | 8.25 | 0.45 | 7.33 | -1.41 |
| Paddy | 1.43 | 204.9 | -1.07 | -0.34 | 9.24 | -0.01 | 6.22 | 0.89 |
| Wheat | 1.03 | 214.5 | 1.90 | 1.13 | 7.40 | 2.37 | 9.75 | -2.32 |
| Pulses | 0.64 | 193.1 | -3.08 | 0.10 | 13.37 | -13.53 | 5.13 | -11.05 |
| Vegetables | 1.87 | 237.7 | -22.84 | -14.62 | 21.37 | -19.27 | 8.11 | 6.78 |
| Potato | 0.28 | 178.9 | -19.88 | -20.70 | 76.79 | -37.21 | 72.57 | -38.84 |
| Onion | 0.16 | 210.8 | -23.66 | 11.48 | 43.51 | -48.49 | 28.33 | -33.42 |
| Fruits | 1.60 | 200.2 | 1.76 | 1.57 | 10.32 | 1.30 | 15.30 | 1.78 |
| Milk | 4.44 | 191.7 | 0.75 | 0.05 | 3.34 | 2.73 | 2.58 | 2.51 |
| Eggs, Meat & Fish | 2.40 | 181.1 | 0.00 | 2.49 | 0.63 | 0.77 | 3.56 | 3.66 |
| B. Non-Food Articles | 4.12 | 180.2 | 0.78 | 5.32 | -1.14 | 3.46 | 3.01 | 7.58 |
| Oil Seeds | 1.12 | 218.7 | 0.33 | 4.19 | -2.35 | 9.47 | 0.16 | 19.25 |
| C. Minerals | 0.83 | 256.2 | -1.26 | -0.47 | 5.03 | 6.69 | 1.56 | 12.76 |
| D. Crude Petroleum & Natural gas | 2.41 | 139.1 | 6.34 | 4.27 | -0.65 | -9.33 | -0.53 | -7.82 |
| Crude Petroleum | 1.95 | 115.4 | 8.79 | 7.35 | -1.06 | -13.21 | -0.76 | -11.23 |
| II. FUEL & POWER | 13.15 | 145.9 | 0.13 | -1.62 | -1.51 | -3.16 | -1.87 | -4.01 |
| LPG | 0.64 | 114.2 | -0.72 | 6.63 | 3.23 | -5.08 | 2.23 | -7.68 |
| Petrol | 1.60 | 143.9 | 1.07 | -1.71 | -3.67 | -5.03 | -3.64 | -4.58 |
| HSD | 3.10 | 158.5 | 0.61 | -1.67 | -3.47 | -3.73 | -3.61 | -4.29 |
| III. MANUFACTURED PRODUCTS | 64.23 | 147.5 | 0.28 | 1.30 | 1.46 | 2.14 | 2.65 | 2.86 |
| Mf/o Food Products | 9.12 | 179.6 | 0.40 | 0.67 | 6.37 | 4.71 | 10.73 | 1.18 |
| Vegetable & Animal Oils and Fats | 2.64 | 188.5 | 1.02 | 1.02 | 12.96 | 13.29 | 33.74 | 0.53 |
| Mf/o Beverages | 0.91 | 135.8 | -0.07 | 0.30 | 1.97 | 1.22 | 1.51 | 1.04 |
| Mf/o Tobacco Products | 0.51 | 183.7 | 0.50 | 0.38 | 2.37 | 2.52 | 4.02 | 1.38 |
| Mf/o Textiles | 4.88 | 140.4 | 0.15 | 0.93 | 1.14 | 1.24 | 2.24 | 2.48 |
| Mf/o Wearing Apparel | 0.81 | 157.4 | -0.13 | 0.38 | 1.69 | 1.83 | 2.19 | 2.08 |
| Mf/o Leather and Related Products | 0.54 | 128.5 | 1.19 | 0.71 | 0.74 | 2.10 | 3.24 | 0.78 |
| Mf/o Wood and Products of Wood and Cork | 0.77 | 151.3 | 0.88 | 0.07 | 2.10 | 0.92 | 1.01 | 1.14 |
| Mf/o Paper and Paper Products | 1.11 | 140.3 | 0.87 | 0.07 | -1.36 | 0.96 | 0.58 | 0.57 |
| Mf/o Chemicals and Chemical Products | 6.47 | 137.2 | 0.22 | 0.15 | -0.56 | 0.52 | 1.03 | 0.29 |
| Mf/o Pharmaceuticals, Medicinal Chemical and Botanical Products | 1.99 | 145.7 | 0.69 | -0.41 | 1.03 | 1.21 | 1.40 | 0.48 |
| Mf/o Rubber and Plastics Products | 2.30 | 128.2 | 0.23 | 0.23 | 1.15 | 0.12 | 1.65 | -0.85 |
| Mf/o other Non-Metallic Mineral Products | 3.20 | 133.4 | 0.38 | 0.53 | -2.78 | 1.45 | -1.64 | 0.91 |
| Cement, Lime and Plaster | 1.64 | 131.8 | 0.00 | 0.92 | -5.54 | 1.39 | -5.10 | 1.23 |
| Mf/o Basic Metals | 9.65 | 145.4 | -0.22 | 5.82 | -1.13 | -0.76 | -1.15 | 5.98 |
| Mild Steel - Semi Finished Steel | 1.27 | 117.8 | 0.43 | 2.70 | -2.16 | -1.43 | 0.09 | 0.43 |
| Mf/o Fabricated Metal Products, Except Machinery and Equipment | 3.15 | 136.5 | -0.44 | 0.15 | -2.15 | 0.63 | -1.81 | 0.89 |
Note: * = Provisional. Mf/o = Manufacture of
Annex-II
WPI Inflation (Base Year: 2011-12=100) for last 6 months
| Commodities/Major Groups/Groups/Sub-Groups/Items | Weight | WPI based inflation (YoY) figures for last 6 months | |||||
| Aug-25 | Sep-25 | Oct-25 | Nov-25 | Dec-25* | Jan-26* | ||
| ALL COMMODITIES | 100.00 | 0.52 | 0.19 | -1.02 | -0.13 | 0.83 | 1.81 |
| I. PRIMARY ARTICLES | 22.62 | -2.10 | -3.12 | -5.93 | -2.53 | 0.21 | 2.21 |
| A. Food Articles | 15.26 | -3.11 | -5.12 | -8.31 | -4.07 | -0.43 | 1.55 |
| Cereals | 2.82 | 1.03 | -0.24 | -0.91 | -1.66 | -1.18 | -1.41 |
| Paddy | 1.43 | -0.84 | -1.38 | -1.17 | -1.07 | 0.15 | 0.89 |
| Wheat | 1.03 | 4.75 | 3.02 | 0.81 | -1.12 | -1.58 | -2.32 |
| Pulses | 0.64 | -15.23 | -17.19 | -16.59 | -15.21 | -13.88 | -11.05 |
| Vegetables | 1.87 | -14.34 | -24.38 | -35.00 | -19.87 | -3.50 | 6.78 |
| Potato | 0.28 | -44.56 | -42.03 | -39.56 | -36.19 | -38.21 | -38.84 |
| Onion | 0.16 | -50.46 | -64.12 | -66.06 | -64.70 | -54.40 | -33.42 |
| Fruits | 1.60 | -4.86 | -4.16 | -7.60 | -0.45 | 1.97 | 1.78 |
| Milk | 4.44 | 2.58 | 3.08 | 3.23 | 3.35 | 3.23 | 2.51 |
| Eggs, Meat & Fish | 2.40 | 0.06 | 1.56 | 1.93 | 2.08 | 1.14 | 3.66 |
| B. Non-Food Articles | 4.12 | 5.56 | 3.64 | 2.10 | 3.93 | 2.95 | 7.58 |
| Oil Seeds | 1.12 | 14.00 | 9.70 | 6.90 | 9.54 | 14.82 | 19.25 |
| C. Minerals | 0.83 | 4.70 | 8.65 | 10.32 | 12.25 | 11.86 | 12.76 |
| D. Crude Petroleum & Natural gas | 2.41 | -9.87 | -3.83 | -7.54 | -8.66 | -5.99 | -7.82 |
| Crude Petroleum | 1.95 | -13.68 | -7.45 | -13.56 | -13.92 | -10.04 | -11.23 |
| II. FUEL & POWER | 13.15 | -3.24 | -2.58 | -2.42 | -2.33 | -2.31 | -4.01 |
| LPG | 0.64 | -1.22 | -7.96 | -9.52 | -12.78 | -14.04 | -7.68 |
| Petrol | 1.60 | -5.72 | -3.82 | -2.60 | -1.75 | -1.88 | -4.58 |
| HSD | 3.10 | -3.54 | -3.09 | -1.89 | -1.64 | -2.07 | -4.29 |
| III. MANUFACTURED PRODUCTS | 64.23 | 2.62 | 2.33 | 1.68 | 1.47 | 1.82 | 2.86 |
| Mf/o Food Products | 9.12 | 7.27 | 4.62 | 1.76 | 0.73 | 0.90 | 1.18 |
| Vegetable & Animal Oils and Fats | 2.64 | 23.06 | 14.80 | 4.83 | 1.53 | 0.54 | 0.53 |
| Mf/o Beverages | 0.91 | 1.19 | 1.04 | 1.04 | 0.89 | 0.67 | 1.04 |
| Mf/o Tobacco Products | 0.51 | 3.24 | 2.14 | 3.18 | 2.77 | 1.50 | 1.38 |
| Mf/o Textiles | 4.88 | 1.32 | 1.77 | 1.91 | 1.91 | 1.68 | 2.48 |
| Mf/o Wearing Apparel | 0.81 | 1.90 | 1.63 | 1.62 | 2.28 | 1.55 | 2.08 |
| Mf/o Leather and Related Products | 0.54 | 2.32 | 1.92 | 1.35 | 1.59 | 1.27 | 0.78 |
| Mf/o Wood and Products of Wood and Cork | 0.77 | 0.00 | 1.08 | 1.61 | 1.41 | 1.96 | 1.14 |
| Mf/o Paper and Paper Products | 1.11 | 0.07 | 0.29 | 0.36 | 1.37 | 1.37 | 0.57 |
| Mf/o Chemicals and Chemical Products | 6.47 | 0.37 | 0.37 | 0.37 | 0.22 | 0.37 | 0.29 |
| Mf/o Pharmaceuticals, Medicinal Chemical and Botanical Products | 1.99 | 0.55 | 1.25 | 1.88 | 1.60 | 1.60 | 0.48 |
| Mf/o Rubber and Plastics Products | 2.30 | 0.08 | 0.31 | -0.54 | -0.08 | -0.85 | -0.85 |
| Mf/o other Non-Metallic Mineral Products | 3.20 | 3.08 | 2.07 | 1.69 | 0.76 | 0.76 | 0.91 |
| Cement, Lime and Plaster | 1.64 | 4.54 | 2.87 | 1.94 | 0.31 | 0.31 | 1.23 |
| Mf/o Basic Metals | 9.65 | -0.51 | 0.00 | -1.58 | -1.23 | -0.07 | 5.98 |
| Mild Steel - Semi Finished Steel | 1.27 | 1.31 | 1.14 | -3.05 | -3.49 | -1.80 | 0.43 |
| Mf/o Fabricated Metal Products, Except Machinery and Equipment | 3.15 | 0.29 | 0.51 | 1.26 | 0.74 | 0.29 | 0.89 |
Note: * = Provisional. Mf/o = Manufacture of
Annex-III
Wholesale Price Indices (Base Year: 2011-12=100) for last 6 months
| Commodities/Major Groups/Groups/Sub-Groups/Items | Weight | WPI Numbers for last 6 months | |||||
| Aug-25 | Sep-25 | Oct-25 | Nov-25 | Dec-25* | Jan-26* | ||
| ALL COMMODITIES | 100.00 | 155.2 | 155.0 | 155.1 | 156.2 | 157.0 | 157.8 |
| I. PRIMARY ARTICLES | 22.62 | 191.0 | 189.4 | 188.7 | 192.9 | 194.2 | 193.9 |
| A. Food Articles | 15.26 | 202.5 | 200.0 | 199.8 | 205.0 | 206.6 | 202.9 |
| Cereals | 2.82 | 206.7 | 206.3 | 206.7 | 207.5 | 208.9 | 209.3 |
| Paddy | 1.43 | 200.3 | 200.6 | 202.0 | 203.7 | 205.6 | 204.9 |
| Wheat | 1.03 | 211.8 | 211.6 | 211.3 | 211.4 | 212.1 | 214.5 |
| Pulses | 0.64 | 198.1 | 196.6 | 195.6 | 195.7 | 192.9 | 193.1 |
| Vegetables | 1.87 | 259.8 | 235.1 | 234.6 | 268.1 | 278.4 | 237.7 |
| Potato | 0.28 | 218.2 | 218.1 | 227.0 | 245.1 | 225.6 | 178.9 |
| Onion | 0.16 | 193.8 | 177.0 | 162.3 | 175.0 | 189.1 | 210.8 |
| Fruits | 1.60 | 197.6 | 200.6 | 194.5 | 197.5 | 197.1 | 200.2 |
| Milk | 4.44 | 190.7 | 191.0 | 191.6 | 191.4 | 191.6 | 191.7 |
| Eggs, Meat & Fish | 2.40 | 173.2 | 175.3 | 174.3 | 176.7 | 176.7 | 181.1 |
| B. Non-Food Articles | 4.12 | 169.1 | 168.1 | 165.3 | 169.2 | 171.1 | 180.2 |
| Oil Seeds | 1.12 | 203.6 | 202.5 | 198.2 | 203.3 | 209.9 | 218.7 |
| C. Minerals | 0.83 | 238.3 | 242.5 | 253.3 | 257.5 | 257.4 | 256.2 |
| D. Crude Petroleum & Natural gas | 2.41 | 139.7 | 140.5 | 136.2 | 134.0 | 133.4 | 139.1 |
| Crude Petroleum | 1.95 | 113.6 | 114.3 | 109.0 | 107.6 | 107.5 | 115.4 |
| II. FUEL & POWER | 13.15 | 143.5 | 143.4 | 145.2 | 146.4 | 148.3 | 145.9 |
| LPG | 0.64 | 113.0 | 107.5 | 108.4 | 107.8 | 107.1 | 114.2 |
| Petrol | 1.60 | 145.1 | 145.9 | 146.0 | 146.1 | 146.4 | 143.9 |
| HSD | 3.10 | 160.8 | 160.0 | 161.1 | 161.7 | 161.2 | 158.5 |
| III. MANUFACTURED PRODUCTS | 64.23 | 145.0 | 145.2 | 145.3 | 145.2 | 145.6 | 147.5 |
| Mf/o Food Products | 9.12 | 178.6 | 178.9 | 179.0 | 178.8 | 178.4 | 179.6 |
| Vegetable & Animal Oils and Fats | 2.64 | 185.2 | 186.9 | 186.8 | 186.0 | 186.6 | 188.5 |
| Mf/o Beverages | 0.91 | 135.6 | 135.7 | 135.9 | 135.9 | 135.4 | 135.8 |
| Mf/o Tobacco Products | 0.51 | 181.7 | 181.3 | 181.6 | 181.9 | 183.0 | 183.7 |
| Mf/o Textiles | 4.88 | 137.7 | 138.2 | 138.5 | 138.7 | 139.1 | 140.4 |
| Mf/o Wearing Apparel | 0.81 | 155.8 | 156.1 | 156.4 | 157.2 | 156.8 | 157.4 |
| Mf/o Leather and Related Products | 0.54 | 127.8 | 127.4 | 127.4 | 127.8 | 127.6 | 128.5 |
| Mf/o Wood and Products of Wood and Cork | 0.77 | 149.5 | 150.2 | 151.1 | 150.6 | 151.2 | 151.3 |
| Mf/o Paper and Paper Products | 1.11 | 139.9 | 140.2 | 140.3 | 140.4 | 140.2 | 140.3 |
| Mf/o Chemicals and Chemical Products | 6.47 | 137.2 | 137.0 | 136.8 | 136.7 | 137.0 | 137.2 |
| Mf/o Pharmaceuticals, Medicinal Chemical and Botanical Products | 1.99 | 145.6 | 145.9 | 146.2 | 146.4 | 146.3 | 145.7 |
| Mf/o Rubber and Plastics Products | 2.30 | 129.2 | 129.1 | 128.9 | 128.5 | 127.9 | 128.2 |
| Mf/o other Non-Metallic Mineral Products | 3.20 | 133.8 | 133.3 | 132.6 | 132.4 | 132.7 | 133.4 |
| Cement, Lime and Plaster | 1.64 | 133.5 | 132.6 | 131.3 | 130.5 | 130.6 | 131.8 |
| Mf/o Basic Metals | 9.65 | 137.6 | 137.7 | 137.1 | 136.9 | 137.4 | 145.4 |
| Mild Steel - Semi Finished Steel | 1.27 | 115.9 | 115.4 | 114.4 | 113.4 | 114.7 | 117.8 |
| Mf/o Fabricated Metal Products, Except Machinery and Equipment | 3.15 | 137.0 | 137.0 | 136.7 | 136.3 | 136.3 | 136.5 |
Note: * = Provisional. Mf/o = Manufacture of
The National Health Authority (NHA), in collaboration with the ICMR–National Institute for Research in Digital Health and Data Science (ICMR–NIRDHDS) and Indian Institute of Technology Kanpur organized the Federated Intelligence Hackathon for Healthcare at the IIT Kanpur campus. The national-level initiative aims to advance secure and scalable artificial intelligence solutions for India’s healthcare ecosystem.
The Federated Intelligence Hackathon for Healthcare commenced on 19 January 2026, with Hackathon Week (19–23 January 2026) featuring intensive technical development, mentoring and evaluations at IIT Kanpur. The two-day on-site programme, beginning on 23 January 2026, brought together policymakers, clinicians, researchers, start-ups and industry experts for expert sessions, technical presentations and panel discussions.

Prof. Sandeep Verma, Head of the Gangwal School of Medical Sciences and Technology
The sessions commenced with a keynote address by Prof. Sandeep Verma, Head of the Gangwal School of Medical Sciences and Technology. He highlighted the pivotal role of interdisciplinary research in driving AI‑enabled healthcare innovation. Prof. Verma underscored the necessity of cross‑domain collaboration, emphasizing that meaningful progress in Health AI can only be achieved through the integration of medicine, engineering, data science, and policy. He noted that such convergence is essential to building sustainable and impactful healthcare systems for the future.

Prof. Phaneendra Kumar Yalavarthy, Chief PM, TANUH, IISc Bengaluru
This was followed by technical sessions by Prof. Phaneendra Kumar Yalavarthy, Chief PM, TANUH, IISc Bengaluru, covering traditional machine learning methods, clinical evaluation of AI systems and regulatory considerations for healthcare deployment. He opined that, “AI in healthcare has evolved from rule‑based systems to data‑driven learning, where deep learning enables autonomous feature discovery for complex tasks like medical image segmentation. By balancing bias–variance, applying methods such as regression, SVMs, and regularization, we can build robust, reliable, and clinically sustainable digital health solutions.”
The programme also featured a live Health AI demonstration by Google Health AI and the Wadhwani Foundation, presented by Manish Kumar, Vice President – AI Platform, Wadhwani Foundation. Panel discussions on Health Technology Assessment (HTA) for algorithms and data sourcing for Health AI in India brought together experts from government, industry and academia, including representatives from Samsung India, Google Health AI, CoRover.ai, ArtPark@IISc and NITI Aayog. The discussions focused on validation pathways, regulatory readiness, data standardisation and large-scale deployment of AI tools within public health systems.

Domain-specific sessions showcased datasets and AI use cases in ophthalmology, bone age detection and computational pathology, highlighting emerging opportunities for population-scale screening and diagnostics. The technical programme concluded with remarks on responsible AI deployment in healthcare by Prof. Nisheeth Srivastava, IIT Kanpur.
The hackathon has received 191 registrations, comprising 76 individual participants and 115 teams, reflecting strong nationwide participation from health-tech start-ups, AI/ML researchers and developers, clinicians, and medical institutions. The collaboration brings together NHA’s digital health and governance perspective through initiatives such as the Ayushman Bharat Digital Mission, ICMR–NIRDHDS’s clinical research and data science expertise, and IIT Kanpur’s advanced capabilities in artificial intelligence and federated systems, with a shared objective of strengthening data readiness, validation frameworks and clinical applicability of Health AI tools.

The hackathon carries a total prize pool of ₹12 lakh, with post-hackathon assessment and award ceremonies scheduled on 23–24 January 2026, respectively. Discussions on 23rd January 2026 highlighted the need to address foundational challenges in validating Health AI at scale. Programmes scheduled for 24th January 2026 will focus on policy-level deliberations, responsible AI frameworks, presentations by top-performing teams of the hackathon across priority healthcare use cases, announcement of hackathon prize winners, certificate handover, and the valedictory session.
Recombination of monkeypox virus (MPXV) strains has been documented in recent months, with two cases of a recombinant strain comprising clade Ib and IIb MPXV reported. Recombination is a known natural process that can occur when two related viruses infecting the same individual exchange genetic material, producing a new virus. The first case was detected in the United Kingdom of Great Britain and Northern Ireland (hereafter “United Kingdom”), with travel history to a country in South-East Asia, and the second in India, with travel history to a country in the Arabian Peninsula. Detailed analysis of the virus genomes shows that the two individuals fell ill several weeks apart with the same recombinant strain, suggesting that there may be further cases than are currently reported. Both cases had similar clinical presentation to that observed for other clades. Neither patient experienced severe outcomes. Contact tracing for both cases in the reporting countries has been completed; no secondary cases were detected. Based on available information, the overall WHO public health risk assessment for mpox remains unchanged: the risk is assessed as moderate for men who have sex with men with new and/or multiple partners and for sex workers or others with multiple casual sexual partners, and low for the general population without specific risk factors.
Union Minister for Health and Family Welfare, Shri Jagat Prakash Nadda, today addressed the 8th Convocation Ceremony of Swami Rama Himalayan University, Dehradun, and congratulated the graduating students on successfully completing their academic journey.

Addressing the gathering, Shri Nadda described the convocation as both special and significant—special as it represents years of dedication, perseverance, and hard work, and significant as it marks the beginning of a new phase of professional responsibility and service. He urged the graduates to uphold the highest ideals of the medical profession, continuously strive for excellence, and dedicate their skills and knowledge to the service of humanity.
Highlighting the transformative strides made in India’s health sector over the past eleven years, the Union Health Minister stated that the number of AIIMS has increased from 6 to 23, substantially expanding access to advanced tertiary healthcare services across the country. He further informed that institutional deliveries have risen to nearly 89%, reflecting strengthened maternal healthcare systems.

The Minister noted that the Maternal Mortality Ratio (MMR) has declined from 130 per lakh live births a decade ago to 88 per lakh live births, while the Infant Mortality Rate (IMR) has reduced from 39 per thousand live births to 27 per thousand live births, demonstrating sustained progress in maternal and child health outcomes.
Citing estimates from the World Health Organization and UN agencies, Shri Nadda stated that India has achieved a significantly faster decline in the Under-5 Mortality Rate over the past decade compared to the global average, underscoring the impact of focused policy interventions and expanded healthcare access. Referring to tuberculosis control efforts, he noted that India has recorded a substantial decline in TB incidence, outperforming the global average reduction through sustained public health measures and community-based interventions.
The Union Health Minister further highlighted India’s historic COVID-19 vaccination drive, under which more than 220 crore vaccine doses, including precautionary and booster doses, have been administered nationwide, demonstrating the scale, resilience, and efficiency of India’s public health system.

Emphasizing financial protection in healthcare, Shri Nadda spoke about Ayushman Bharat–Pradhan Mantri Jan Arogya Yojana (AB-PMJAY), which provides health cover of ₹5 lakh per family. He stated that the scheme now benefits nearly 62 crore people, covering approximately 40% of India’s population. Referring to evidence from reputed international medical journals and independent evaluations, he noted that AB-PMJAY has significantly improved access to timely cancer care and strengthened financial protection for eligible beneficiaries across the country.
He further stated that India has witnessed a substantial reduction in Out-of-Pocket Expenditure on healthcare over the past decade, easing the financial burden on households, particularly among economically vulnerable sections. Despite accounting for nearly one-sixth of the global population, India has made significant progress in reducing malaria incidence and mortality through sustained vector-borne disease control efforts in line with WHO-reported trends.
Underscoring the importance of comprehensive primary healthcare, the Minister informed that more than 1.82 lakh Ayushman Arogya Mandirs have been operationalized nationwide as the first point of contact for citizens. Of these, 50,000 centres have already been certified under National Quality Assurance Standards (NQAS), with a target to scale up to 1 lakh NQAS-certified Ayushman Arogya Mandirs in the near future.

Concluding his address, Shri Nadda reiterated that the strength of India’s healthcare system ultimately rests on the commitment, competence, and compassion of its medical professionals.
Speaking on the occasion, Chief Minister of Uttarakhand, Shri Pushkar Singh Dhami, congratulated the graduating students and highlighted the State Government’s efforts to strengthen healthcare infrastructure, medical education, and emergency services, particularly in remote and hilly regions. He emphasized the role of institutions such as Swami Rama Himalayan University in developing a skilled healthcare workforce for the State and the nation.
Shri Dhan Singh Rawat, Hon’ble Minister of Medical Health & Higher Education, Uttarakhand, also addressed the gathering and appreciated the University’s contribution to advancing medical and higher education. He encouraged graduates to actively contribute to research, innovation, and public health services, especially in underserved areas.
Established with the vision of advancing excellence in healthcare and higher education, Swami Rama Himalayan University has emerged as a leading institution in the Himalayan region, offering a wide range of undergraduate, postgraduate, and doctoral programmes across medical, paramedical, nursing, management, engineering, and allied sciences.
The ceremony witnessed conferment of degrees across various disciplines, marking a significant academic milestone for the graduating cohort.
Dr. Vijay Dhasmana, President, Swami Rama Himalayan University; Dr. Rajendra Dobhal, Vice-Chancellor; senior dignitaries, faculty members, parents, and students were present on the occasion.
In a major boost to India’s growing startup ecosystem, the Union Cabinet chaired by the Prime Minister, Shri Narendra Modi, has approved the establishment of the Startup India Fund of Funds 2.0 (Startup India FoF 2.0) with a total corpus of Rs. 10,000 crore for the purpose of mobilizing venture capital for the startup ecosystem of the country.
The Scheme is designed to accelerate the next phase of India’s startup journey by mobilising long-term domestic capital, strengthening the venture capital ecosystem, and supporting innovation-led entrepreneurship across the country.
Launched under the Startup India initiative, Startup India FoF 2.0 builds on nearly a decade of sustained efforts to make India one of the world’s leading startup nations. Since the launch of Startup India in 2016, India’s startup ecosystem has witnessed an extraordinary transformation growing from fewer than 500 startups to over 2 lakh Department for Promotion of Industry and Internal Trade (DPIIT)-recognised startups today, with 2025 marking the highest ever annual startup registrations.
Building on Fund of Funds for Startups 1.0
The Startup India FoF 2.0 follows the strong performance of the Fund of Funds for Startups (FFS 1.0), which was launched in 2016 to address funding gaps and catalyse the domestic venture capital market for startups.
Under FFS 1.0, the entire corpus of Rs. 10,000 crore has been committed to 145 Alternative Investment Funds (AIFs). Such supported AIFs have invested over Rs. 25,500 crore in more than 1,370 startups across the country in sectors such as agriculture, artificial intelligence, robotics, automotive, clean tech, consumer goods & services, e-commerce, education, fintech, food & beverages, healthcare, manufacturing, space tech, and biotechnology amongst others.
FFS 1.0 played a pivotal role in nurturing first-time founders, crowding in private capital, and helping build a strong foundation for India’s venture capital ecosystem.
Key Features of the Scheme:
While the first phase built the ecosystem, Startup India FoF 2.0 is designed to take Indian innovation to the next level. The new fund will have a targeted, segmented funding approach to support:
Startup India FoF 2.0 is expected to play a pivotal role in shaping India’s economic trajectory, leading to transformational impact.
Startup India Fund of Funds 2.0 is expected to play a critical role in advancing India’s innovation-led growth agenda. By supporting startups that build globally competitive technologies, products, and solutions, the Fund will contribute to strengthening India’s economic resilience, boosting manufacturing capabilities, generating high-quality jobs, and positioning India as a global innovation hub.
Aligned with the national vision of Viksit Bharat @ 2047, the Fund represents the Government’s continued commitment to empowering entrepreneurs, fostering innovation, and unlocking the full potential of India’s startup ecosystem.
