“India is committed to eliminating Kala Azar from the country by 2023. 632 (99.8%) endemic blocks have already achieved elimination status (<1 case/10,000). Only one block (Littipara) of Pakur district, Jharkhand is in the endemic category (1.23 case/10,000 population). We are working holistically with the state government and other stakeholders to achieve elimination in Jharkhand”. This was stated by Union Minister of Health and Family Welfare, Dr. Mansukh Mandaviya as he chaired a high level meeting to review the status of Kala-Azar disease in the four endemic states of Bihar, Uttar Pradesh, Jharkhand and West Bengal in the presence of Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar. Shri Tejaswi Yadav, Deputy Chief Minister & Health Minister (Bihar), Shri Brajesh Pathak, Deputy Chief Minister & Health Minister (Uttar Pradesh) and Shri Banna Gupta, Health Minister (Jharkhand), and senior officers from West Bengal were present at the review meeting.

Glimpses of the meeting can be seen through this tweet by Union Health Minister


Dr. Mansukh Mandaviya appreciated the efforts of the State Governments towards meeting elimination target. He said that “under the visionary leadership of our Prime Minister, it remains our target to ensure health for all of our citizens.” Government has taken several steps towards elimination of Kala-Azar by 2023. From interventions like pucca houses through Pradhan Mantri Awas Yojana (PMAY), rural electrification, timely testing, treatment, periodic high-level review, to incentivizing through award distribution for states/districts/blocks, government along with its stakeholders are ensuring a robust ecosystem for early detection of the disease and its timely treatment.” Government of India is supporting the states in active case detection, surveillance, treatment, supply of diagnostic kits, medicines, sprays etc.
Dr. Mandaviya said that “While it is commendable that the endemic states are implementing targeted interventions, and some states have eliminated the disease in their districts, it is equally important to sustain the gain and work towards ensuring that the cases stay below 1 case/10,000 population”. He urged the endemic states to ensure that there is a regular review and micro-stratification in high-risk blocks reporting incidence > 0.5 per 10000 populations. “Since Kala-azar impacts those among the lower socio-economic strata of society, early diagnosis & complete case management, integrated control and surveillance along with capacity building of human resource must be taken up on ground level”, he suggested. “For spreading public awareness, information regarding messages on prolonged fever, associated symptoms and free access to diagnosis and treatment and compensations/incentives, other government interventions are required to be widely disseminated through various mediums”, he further added.
Dr. Bharati Pravin Pawar, MoS (HFW) commended the states on their efforts and urged them to ensure that “Transmission through the notorious sandfly be prevented at any cost. Awareness campaigns regarding the symptoms, early detection and treatment available free of charge at Government health facilities are required to be strengthened for enabling early reporting by the affected persons”.
States informed about the status of disease in their respective states and shared their best practices too. The State Health Ministers expressed gratitude for the support provided by the Centre for detection, surveillance, treatment through various interventions. Health Ministers of states where elimination status is achieved assured to continue working in Mission mode to ensure that the stats is sustained.
About 90% of global cases of kala Azar were reported from eight countries: Brazil, Eritrea, Ethiopia, India, Kenya, Somalia, South Sudan and Sudan in 2021, with India contributing 11.5% of total cases reported globally. Kala-azar is endemic in 633 blocks of 54 districts in four endemic states of Bihar, Jharkhand, Uttar Pradesh and West Bengal. It is a notifiable disease in these states. Currently more than 90% of the Kala-azar cases are contributed by Bihar and Jharkhand. Uttar Pradesh (2019) and West Bengal (2017) states have achieved their elimination targets at the block level.
Shri Rajesh Bhushan, Union Health Secretary, Shri. Rajiv Manjhi, JS and other senior officials of the Health Ministry were present. Senior government officials serving in the States/UTs, NGOs and development partners were also present virtually.
Union Health Secretary, Shri Rajesh Bhushan, chaired a workshop through video conference (VC) today with all States and UTs to review the rollout of vaccination for the age group 15-18 and Precautionary 3rd dose for vulnerable categories; Healthcare workers (HCW), Frontline workers (FLW), and those in the 60+ age group who have comorbidity.

As announced by the Prime Minister Shri. Narendra Modi on 25th December, 2021, the vaccination for the age-group 15-18 yrs is scheduled to begin from Monday, 3rd January, 2022 while administration of precautionary 3rd dose for the vulnerable categories is to commence from Monday, 10th January, 2022. The detailed guidelines regarding this were issued by the Ministry of Health and Family Welfare on 27th December,2021
In context of the vaccination of the age group 15-18 yrs, the Union Health Secretary informed the States/UTs that only ‘Covaxin’ is to be administered in this population category and additional doses of ‘Covaxin’ will be sent to all States and UTs. Union Government will share the supply schedule of ‘Covaxin’ with all the States and UTs in the next few days. Potential beneficiaries can either register themselves on Co-WIN from the 1st of January, 2022 or avail of a walk-in registration when vaccination commences from the 3rd of January. Those with a birth year of 2007 or before will be eligible for vaccination under this category.
All established protocols regarding vaccination are to be followed for the 15-18 yrs age group; beneficiaries are to wait for half-an-hour when they will be monitored for AEFI and will be eligible for the 2nd dose only after 28 days. States were informed that they have the option to designate some COVID Vaccination Center (CVC)s as dedicated CVC for the 15-18 age group exclusively which can also be reflected on Co-WIN. Dedicated CVCs would ensure that there is no confusion in administering the vaccines. For CVCs that are intended to serve categories in addition to 15-18 age group, the States were requested to ensure separate queues for the 15-18 age group and separate vaccination teams. States were advised to form two separate vaccination teams at the same CVC, one for 15-18 age groups and other for all adults to avoid confusion in administration of the right vaccines.
With regards to the administration of the precautionary dose, the Union Health Secretary highlighted that 9 month (39 weeks) must have elapsed since the administration of the 2nd dose to make the beneficiary eligible. Drawing attention to misinformation being propagated through various media regarding the requirement of a doctor’s certificate to establish co-morbidity at the CVC, he categorically emphasized that the Union Government has not issued any directions on the said matter and prescriptions/certificates are NOT mandated to be produced at the CVC for administration of the precaution dose. He also informed that CoWIN will send reminder messages to all those eligible for Precaution dose and Precaution dose will be reflected in the digital vaccination certificates.
States/UTs were further advised to ensure the orientation of vaccinators and vaccination team members for vaccination of 15-18 years age-group and the identification of dedicated session sites for vaccination of 15-18 years age-group. States were exhorted to undertake proper planning in advance for distribution of Covaxin to identified session sites. To avoid the mixing-up of vaccines during administration, separate CVCs, separate session sites, separate queue (if at same session where adult vaccination is ongoing) and separate vaccination team (if at same session site) are to be strived for. States/UTs were also advised to share their requirement of vaccine doses through district wise estimation of beneficiaries using Co-WIN. They are to publicize sessions where vaccination will be available for 15-18 years age-groups. Sufficient vaccines will be provided to states to cover these beneficiaries
Dr. Manohar Agnani, Additional Secretary (Health), Shri Vishal Chauhan, Joint Secretary (Health) were present in the meeting along with Principal Secretary (Health), Additional Chief Secretary (Health) and State Surveillance Officer of the concerned states/UTs.
Union Health Secretary Shri Rajesh Bhushan held a high level meeting with the five poll bound states of Uttarakhand, Goa, Manipur, Uttar Pradesh and Punjab to review the public health response measures for containment and management of COVID19, and the vaccination status in these states.
While Uttarakhand and Goa have reported vaccination coverage for the first and second doses higher than the national average, Uttar Pradesh, Punjab and Manipur have COVID19 vaccination coverage numbers below the national average. Total of 142.38 crore vaccination doses have been administered as on date, of which more than 83.80 crore vaccine doses are for the first dose and more than 58.58 crore are second doses of COVID19 vaccine.
The States were advised to speedily ramp up the COVID19 vaccination of all eligible population for the first dose and ensure that those who were due for the second dose are administered the second dose. District-wise weekly vaccination implementation plans are required to be drawn up for this purpose. The state authorities were advised to review the implementation status on a daily basis.
The poll-bound states were also advised to exponentially increase the testing to ensure that the infected cases are identified promptly for timely initiation of public health response measures, and to ensure that there is no sudden upsurge in numbers due to low testing.
The state authorities were strongly advised to ensure that recommended COVID Appropriate Behavior is strictly followed and adequate measures are undertaken for their effective enforcement.
The Union Government continues to support efforts of States/UTs towards management of COVID – 19 pandemic under the ‘Whole of Government’ approach.
“Centre and States need to work in tandem and in a collaborative spirit as was done during the previous surges for COVID19 prevention and management”. This was stated by Dr. Mansukh Mandaviya, Union Minister of Health and Family Welfare as he virtually interacted with State Health Ministers and Principal Secretaries/ Additional Chief Secretaries and Information Commissioners, here today. The virtual meeting was held in the presence of Dr. Bharati Pravin Pawar, Union Minister of State for Health and Dr V K Paul, Member (Health), NITI Aayog to review the public health preparedness for containment and management of COVID-19 and progress of national COVID-19 vaccination campaign in view of the recent upsurge in cases in some countries like China, Japan, Brazil and the United States.

Shri N Rangaswamy, Chief Minister, Puducherry, Dr Manik Saha, Chief Minister and Health Minister, Tripura, Shri Manoj Sinha, Lieutenant Governor, Jammu & Kashmir, Shri Parsadi Lal Meena, Health Minister (Rajasthan), Shri Dhan Singh Rawat, Health Minister (Uttarakhand), Shri Keshab Mahanta, Health Minister (Assam), Dr K Sudhakar, Health Minister (Karnataka), Shri Banna Gupta, Health Minister (Jharkhand), Shri Prabhuram Choudhury, Health Minister (Madhya Pradesh), Shri S Chetan Singh Jauramajra, Health Minister (Punjab), Shri T S Singh Deo, Health Minister (Chhattisgarh), Shri Sapam Ranjan Singh, Health Minister (Manipur), Shri Anil Vij, Health Minister (Haryana), Shri Thiru Ma Subramanian, Health Minister (Tamil Nadu), Smt. Vidadala Rajini, Health Minister (Andhra Pradesh), Smt. Veena George, Health Minister (Kerala), Smt. Chandrima Bhattacharya, Health Minister (West Bengal) and Shri Manish Sisodia, Deputy Chief Minister (Delhi) joined the high-level review meeting.

Union Health Minister referred to the message of Hon. Prime Minister from the high level review meeting held yesterday and advised States to be on the alert and keep all preparedness for COVID19 management. States were requested to continue with pre-emptive & proactive approach. He urged States to strengthen the surveillance system for whole genome sequencing of positive case samples to track the variants through Indian SARS-CoV-2 Genomics Consortium (INSACOG) network to ensure timely detection of newer variants, if any, circulating in the country. Focus is to be given to health facility-based sentinel surveillance; pan-respiratory virus surveillance; community-based surveillance; and sewage/wastewater surveillance. He highlighted the “Need to collectively reinvigorate the system & remove any sense of complacency and fatigue”.
Dr. Mandaviya said that irrespective of the new COVID variants, ‘Test-Track-Treat-Vaccinate & Adherence to COVID Appropriate Behaviour’ continue to remain the tested strategy for COVID management. This would facilitate undertaking of appropriate public health measures, he said. States/UTs were also requested to expeditiously increase the rate of testing from the current rate of 79 tests per million, as on week ending 22nd December, 2022. They were further advised to increase the share of RT-PCR in tests.
Union Health Minister advised States/UTs to ramp up vaccination of all eligible population, especially of the elderly and vulnerable population group. He cautioned against spreading of misinformation by ensuring dissemination of factually correct information in a timely manner. In view of the upcoming festive season, he stressed on the importance of public awareness campaigns regarding adherence to Covid Appropriate Behaviour. Dr Mandaviya requested the State Health Ministers to personally monitor and review the preparedness of all infrastructure and ensure there is adequate stock of essential medicines.
States/UTs were briefed on the global COVID-19 situation and the domestic scenario. They were reminded that Union Health Ministry has already issued “Operational Guidelines for Revised Surveillance Strategy in context of COVID-19” in June 2022 which calls for early detection, isolation, testing and timely management of suspected and confirmed cases to detect and contain outbreaks of new SARS-CoV-2 variants. Union Health Minister requested States/UTs to ensure effective implementation of the same. There was comprehensive and detailed discussion on various aspects of COVID management including ramping up of hospital infrastructure; increased testing.
States appreciated the timely review meetings chaired by the Hon. Prime Minister and the Union Health Minister and advisories from the Union Health Ministry. They assured that they will work with the Centre for effective prevention and management of COVID-19. They informed that they are maintaining vigil and are reviewing the present situation. States also assured they will hold mock drill for readiness of hospital infrastructure on 27th December, 2022.
Dr. Manohar Agnani, AS (Health Ministry), Shri Lav Agarwal, AS (Health Ministry), Shri Mandeep Bhandari, JS (Health Ministry), Dr. Atul Goel, DGHS, and senior officials of the ministry were present in the meeting.
In the year 2022, various programs and initiatives were implemented in the Department of Pharmaceuticals. Major achievements of the Department this year include schemes like 'Pradhan Mantri Bhartiya Janaushadhi Pariyojana' to provide quality generic medicines at affordable prices to the poor and underprivileged and PLI scheme to strengthen India's manufacturing capacity in the pharmaceutical sector by increasing investment and production. Apart from this, the department also laid special emphasis on promoting domestic manufacturing of medical equipment and strengthening the pharmaceutical industry.
Under Pradhan Mantri Bhartiya Janaushadhi Pariyojana scheme, dedicated outlets known as Pradhan Mantri Bhartiya Janaushadhi Kendras (PMBJK) are opened all over the country to provide generic medicines at affordable prices to the masses. Till 30th November 2022, 8916 PMBJKs have been opened across the country. Target is to increase these kendras to 10500 by March 2025. Prices of the Jan Aushadhi medicines are generally 50%-90% less than that of branded medicines’ which are available in the open market. Medicines are procured from World Health Organization – Good Manufacturing Practices (WHO-GMP) certified suppliers only for ensuring the quality of the products.
The Product basket of PMBJP comprises 1759 drugs and 280 surgical. The target is to enhance the product basket to include 2000 medicines and 300 surgical products by March 2025 so that all essential medicines covering therapeutic groups, like - Anti Diabetics, Cardiovascular Drugs, Anti-Cancer, Analgesics & Antipyretics, Anti Allergic, Gastro Intestinal Agents, Vitamins, Minerals & Food supplements, Tropical Medicines, etc. are provided.
To ensure easy availability of the menstrual health services to all women across the country, “Janaushadhi Suvidha Oxy-Biodegradable Sanitary Napkin” are available for sale in all PMBJP Kendras across the country at ₹1.00 per sanitary pad. Till November, 2022, more than 31.00 Crore pads have been sold through these kendras.
In the financial year i.e. 2021-22, Pharmaceuticals & Medical Devices Bureau of India (PMBI) had made sales of Rs. 893.56 Crore which led to savings of approximately Rs. 5300 Cr. to the citizens. In the current financial year 2022-23 till 30th November 2022, PMBI has made sales of Rs. 758 Crore which has led to savings of approximately Rs. 4500 Cr. to the citizens. Thus, in all, approximately Rs. 18,000 Crore have been saved under this Pariyojana in last 8 years.
IT-enabled End to End Supply Chain system has been implemented and one central warehouse at Gurugram and three regional warehouses at Chennai, Guwahati & Surat have been established. Further, it has been planned to open two more warehouses in Western and Central India.
FDI inflows in pharmaceutical sector (in both pharmaceuticals and medical devices) was Rs 12,097 crore in the financial year 2021-22. During current financial year of 2022-23 from April 2022 to September 2022, FDI inflows has been Rs 8,081 crore. Further, the Department of Pharmaceuticals has approved 21 FDI proposals worth Rs. 4,681 crore for brownfield projects during 1st January 2022 to 30th November 2022.
Department of Pharmaceuticals (DoP) notified the amended Schedule-I of Drugs Prices Control Order (DPCO) 2013 on 11th November 2022 based on National List of Essential Medicines 2022 Notified by Ministry of Health and Family Welfare on 13th September 2022. Based on the same, National Pharmaceutical Pricing Authority (NPPA), an attached office under DoP is under process of revising the Ceiling Prices of the drugs coming under the Schedule-I as per extant provisions of DPCO, 2013.
On the occasion of 25th Foundation day NPPA on 29th August 2022, an updated version of Integrated Pharmaceutical Database Management System 2.0 (IPDMS) was launched which is an important step towards bringing in enhanced technology to facilitate the interface between Government and the stakeholders. On the same occasion, updated version of Pharma Sahi Dam Mobil App 2.0 was also launched, which empowers the consumers.
The Scheme would be operational over a period of five years from FY 21-22 to 25-26 and has an outlay of Rs.500 cr. The Scheme has 3 components / sub-schemes:
Assistance to Pharmaceutical Industry for Common Facilities (APICF)
Pharmaceutical Technology Upgradation Assistance Scheme (PTUAS)
Pharmaceutical & Medical Devices Promotion and Development Scheme (PMPDS)
Under sub-scheme Assistance to Pharmaceutical Industry for Common Facilities (APICF), 20 project proposals have been received of which 17 were found eligible under the scheme. Of these 17 project proposals, 7 have been shortlisted and requested to submit the DRP by 15th December, 2022 for further examination and finalization for approval of projects.
Under Sub-Scheme Pharmaceutical Technology Upgradation Assistance Scheme (PTUAS) more than 60 applications have been registered.
The Scheme was approved with the objective of attaining self-reliance and reducing import dependence in critical KSMs/DIs/APIs. The scheme will boost domestic manufacturing of identified KSMs, DIs and APIs by attracting large investments in the sector and thereby reduce India’s import dependence in critical APIs.
The tenure of the sub-scheme is from financial year 2020-21 to 2029-30, with the total financial outlay of Rs. 6,940 crore. The Financial incentive under the sub-scheme is provided on sales of 41 identified products categorized into four Target Segments. Total 249 applications were received in four Rounds. 51 applicants have been approved with committed investment of Rs 4,138.41 cr. against which investment of Rs. 1707 cr. has already been incurred. These 51 projects are expected to generate an employment of around 10,598 persons. The work on these projects have already generated employment of 1,907 persons up to September, 2022. Based on Quarterly Review Report (QRR) of September, 2022, 21 project has been commissioned with actual investment of Rs. 890.82 cr. as against total committed investment of Rs. 843.79 cr.
The Scheme envisages boosting domestic manufacturing and attracting large investments in the Medical Devices Sector. The tenure of the scheme is from FY 2020-21 to FY 2027-28 with total financial outlay of Rs. 3,420 crore. The financial incentive is to be given to selected companies at the rate of 5% on incremental sales of medical devices manufactured in India and covered under the Target segments of the scheme, for a period of five (5) years.
The identified products under this Scheme have been categorized into four Target Segments which is “Cancer care/Radiotherapy medical devices, Radiology & Imaging medical devices (both ionizing & non-ionizing radiation products) and Nuclear Imaging devices, Anaesthetics & Cardio-Respiratory medical devices including Catheters of Cardio Respiratory Category & Renal Care medical devices and All Implants including implantable electronic devices”.
Total 42 applications were received in two round of application window. Out of 42 applications, 21 applicants have been approved with committed investment of Rs 1,058.97 cr and expected employment generation of around 6,411pesons. 13 projects have already been commissioned for 31 products as on September 2022. The actual employment generated up to September 2022 is 2,892 persons.
The objective of this scheme is to enhance India’s manufacturing capabilities by increasing investment and production in the sector and contributing to product diversification towards high value goods in the pharmaceutical sector.
The scheme covers pharmaceutical goods under following three categories-
Category 1: Biopharmaceuticals; Complex generic drugs; Patented drugs or drugs nearing patent expiry; Cell based or gene therapy drugs; Orphan drugs; Special empty capsules like HPMC, Pullulan, enteric etc.; Complex excipients; Phyto-pharmaceuticals; Other drugs as approved.
Category 2: Active Pharmaceutical Ingredients / Key Starting Materials / Drug Intermediates (except for the 41 eligible products already covered under the “PLI Scheme for promotion of domestic manufacturing of critical KSMs / DIs / APIs” at sl.no.(i) above).
Category 3 (Drugs not covered under Category 1 and Category 2): Repurposed drugs; Auto immune drugs, anti-cancer drugs, anti-diabetic drugs, anti-infective drugs, cardiovascular drugs, psychotropic drugs and anti-retroviral drugs; In vitro diagnostic devices; Other drugs as approved; Other drugs not manufactured in India.
The tenure of the Scheme is from Financial Year 2020-21 to Financial Year 2028-29. The scheme provides for incentives on incremental sales to selected participants under these categories at varying rate over the years ranging from 10% to 3%.
The scheme is expected to bring in investment of more than 17,000 crore in the pharmaceutical sector, promote the production of high-value products in the country and increase the value addition in exports. The actual investment of Rs 15,164 cr. have already been made by these 55 applicants.
The support under PLI schemes is expected to promote the production of high-value products in the country as well as generate employment for both skilled and unskilled personnel, estimated at 20,000 direct and 80,000 indirect jobs. The actual employment generated up to September 2022 is 22,560 persons.
The medical devices sector is an essential and integral constituent of the healthcare sector. The current market size of the medical devices sector in India is estimated to be USD 11 Bn and its share in the global medical device market is estimated to be 1.5%.
The contribution of India’s medical devices sector to health become more evident during COVID-19 pandemic where role of medical devices and diagnostic kits, such as Ventilators, IR Thermometers, PPE Kits & N-95 masks, Rapid Antigen Test Kits, and RT-PCR kits etc. were of critical significance. Taking cognizance of the importance of the sector, few additional note-worthy interventions for the Medical Devices Sector, in addition to PLI and Medical Device parks, were also taken in 2022 which are:
Reconstitution of National Medical Devices Promotion Council - On 05.08.2022, the Department reconstituted the National Medical Devices Promotion Council as an inter-departmental council to interact frequently with the medical devices industry to take up matters for resolution, which are regulatory in nature and are spread over different departments. This institutional set-up is expected to resolve the issue of the medical devices sector, which is multi-disciplinary in nature.
Export Promotion Council for Medical Devices - Department of Commerce vide O.M. dated 21st September, 2022 has approved establishment of a separate Export Promotion Council (EPC) for Medical Devices at YEIDA, Uttar Pradesh. The Department has notified the OSD for carrying out further action. The EPC-MD will give the boost to the Medical Devices Sector.
“Clubbing the spirit of Azadi ka Amrit Mahotsav along with expertise from the land of Saint Ramanuja, India is working towards a Swasthya and Samrudh Bharat. Government of India is on a mission to safeguard and enhance public health by ensuring top notch quality of drugs, cosmetics, and medical devices along with maintaining their safety and efficacy in the country. The new building of CDSCO, South Zone will further facilitate Government’s vision of providing safety and regulatory best practices, especially for the southern States/UTs including Tamil Nadu, Puducherry, Kerala and Lakshadweep”. This was stated by Union Minister of Health & Family Welfare Dr. Mansukh Mandaviya as he virtually inaugurated the new building of CDSCO Bhawan, South Zone at Chennai today.

Highlighting CDSCO’s significance, Dr. Mandaviya said that “CDSCO is playing a crucial role in manufacturing, importing and distribution of health products along with ensuring their safety efficacy and quality. They have facilitated right medicine at the right time for our citizens, especially during the COVID pandemic.” He further added that “owing to the significance of CDSCO, Government of India has expanded its capacities. As part of strengthening the drug regulatory system in the country, Government of India has approved various projects, construction of new CDSCO offices, new drug testing labs and upgrading of existing labs, mini labs at ports etc., under the 12th Five Year Plan.”

Given the rapid progress in the field of pharmaceuticals, diagnostics and medical devices sector, Dr. Mandaviya reiterated the clarion call made by Hon’ble Prime Minister for “Make in India” and “Aatmanirbhar Bharat” which has provided impetus for manufacturing medical products indigenously and foster public health goals. He said that “Government of India is focusing on the key areas like quality, accessibility, affordability of medicines as well as encouraging the industry players and other stakeholders.” He also highlighted Government’s willingness towards adapting new technologies and innovations like drug trials through humanoid chips. He said that “Government is also bringing new Drugs, cosmetics and medical device bill which will replace the existing act and rules. These steps will further help us in creating ease of doing business, preventing harassment of innovators and in turn, create a vibrant drug and cosmetics Industry along with a robust regulatory system.”
With 578 Blood centres,700 Drug Manufacturing units, 251 Cosmetics manufacturing units, 9 Vaccine manufacturing units, 85 Medical Devices Manufacturing units, 40 Analytical Labs and 12 BA/BE centres, CDSCO Bhawan South Zone will help in monitoring the quality of drugs through joint inspections and other licenses like Blood Bank license, Vaccines and Sera, Large Volume Parenterals, r-DNA products, Medical Devices etc. They will also assist in inspection of clinical trial facilities, and Public Drug Testing Laboratory along with drawing of regular drugs samples and carrying out surprise checks/Raid jointly/independently. CDSCO plays a crucial role in collaborating with international regulatory agencies too.
Dr. Mandeep K. Bhandari, Joint Secretary MoHFW, Dr. V.G. Somani, Drugs Controller General of India, Dr. B. Kumar Deputy Drugs Controller, and senior officials of the Health Ministry were present at the event.
In order to provide facilities for treatment and care of patients suffering from rare diseases,eight (08) Centres of Excellence have been notified, which are premier Government tertiary hospitals with facilities for diagnosis, prevention and treatment of rare diseases.List of Centres of Excellence (CoEs) is given below:
1) All India Institute of Medical Sciences, New Delhi
2)Maulana Azad Medical College, New Delhi
3) Sanjay Gandhi Post Graduate Institute of Medical Sciences, Lucknow
4) Post Graduate Institute of Medical Education and Research, Chandigarh
5) Centre for DNA Fingerprinting & Diagnostics, Hyderabad
6) King Edward Medical Hospital, Mumbai
7) Institute of Post-Graduate Medical Education and Research, Kolkata
8) Center for Human Genetics (CHG) with Indira Gandhi Hospital, Bengaluru
On the basis of provisions envisaged under National Policy for Rare Diseases, 2021, with regard to providing financial assistance to the patients suffering from rare diseases, it is submitted that for the diseases/ disorders amenable to one time curative treatment (listed under Group -1), financial support will be provided under umbrella scheme of RAN i.e. an amount up toRs. 20 lakh only for entitled beneficiaries shall be provided by the Central Government. The diseases requiring long term / lifelong treatment having relatively lower cost of treatment (listed under Group 2), the financial support is envisaged to be provided by the State Governments. The diseases for which definite treatment is available but involves very high cost and lifelong therapy (listed under Group 3), the policy provides assistance to the patients by creation of Digital Platform for inviting voluntary donations from individuals and corporate donors. Donors will have a choice to make donations to different Centres of Excellence (CoEs) and for the patients’ treatment by these CoEs. Funds are to be utilized in a decentralised manner, i.e. every Centre of Excellence (CoE) will have its own Rare Disease Fund which is to be utilized with the approval of in charge concerned.
The Department of Biotechnology under the Unique Methods of Management of Inherited Disorders (UMMID) initiative has supported the establishment of Genetic Diagnostic Units i.e. National Inherited Disorders Administration Kendras (NIDAN Kendras) to provide comprehensive clinical care including diagnosis, management, multidisciplinary care, counseling, prenatal testing; Training Program for producing skilled clinicians in the area of Human Genetics (Biochemical Genetics, Cytogenetics, Molecular Genetics, Clinical Genetics and Comprehensive Clinical Care); and Screening Program for pregnant women and newborn babies for diagnosis of inherited genetic diseases in aspirational districts to provide comprehensive clinical care. Autonomous institutions of the Department of Biotechnology viz. Centre for DNA Fingerprinting and Diagnostics (CDFD), Hyderabad and National Institute of Biomedical Genomics (NIBMG), Kalyani are also providing genetic testing and counselling services for rare and genetic disorders.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
Under the National Health Mission (NHM), Primary Health Centre (PHC) are established to cover a population of 30,000 in rural areas and 20,000 in hilly, tribal and desert areas.
As per the Rural Health Statistics 2019-20, as on 31.03.2020, a total of 24,918 rural PHCs and 5,895 urban PHCs have been functional in the country. State-wise details are given in Annexure-I.
State-wise details of State Program Implementation Plan approvals for the Primary Health Centres under NHM for the year 2018-19 to 2020-21 and current year 2021-22 are given at Annexure-II.
Annexure-I
NUMBER OF PRIMARY HEALTH CENTRES (PHCs) FUNCTIONING IN RURAL & URBAN AREAS (as on 31.03.2020)

The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
As per the budget announcement 2017-18, 1.5 lakh Sub-Health Centres (SHCs) and Primary Health Centres (PHCs) are to be transformed into Health and Wellness Centres (HWCs) by December, 2022.
As on 14th December 2021, 80,764 AB-HWCs have been operationalized. This includes- Sub Health Centres (SHCs)-54,707, Primary Health Centres (PHCs)-21,894, Urban Primary Health Centres (UPHC-4,163) (Source- AB-HWC Portal). State-wise figures are at Annexure-I.
The expenditure under the scheme since inception is at Annexure-II.
Annexure-I
State-wise figures for Operational no. of AB-HWCs


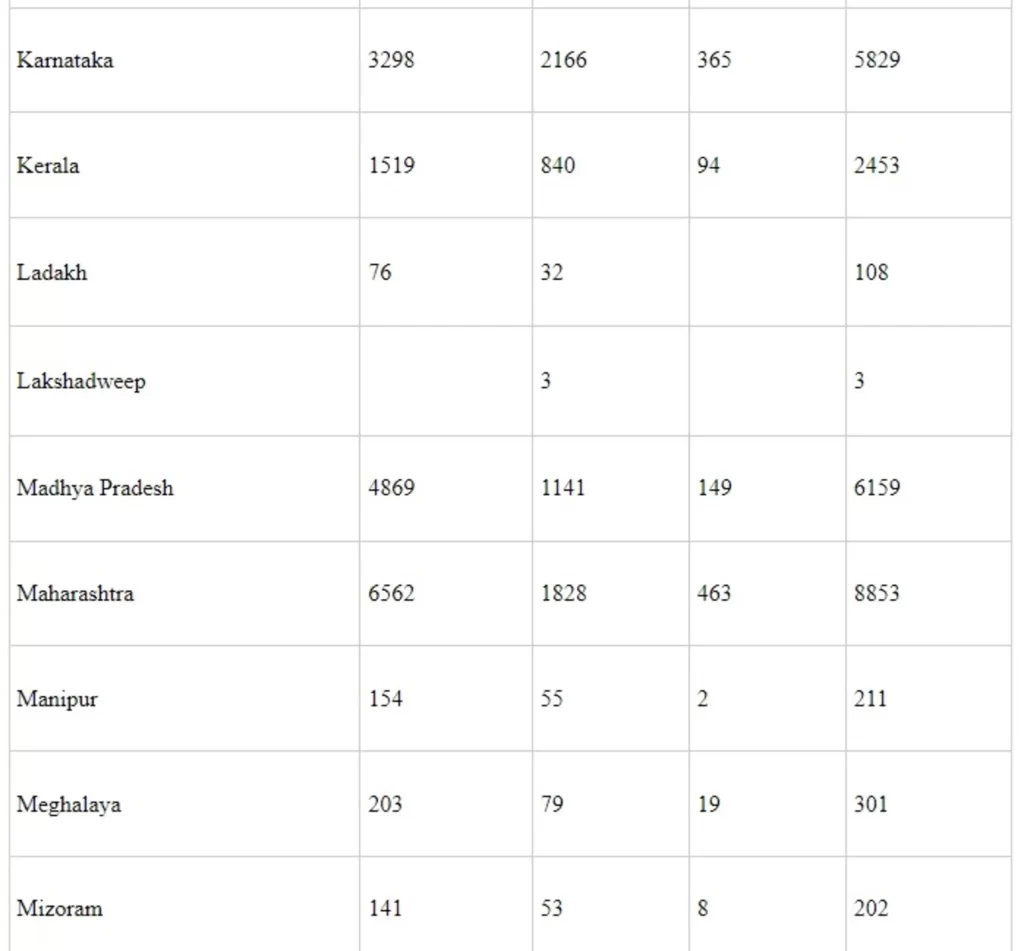
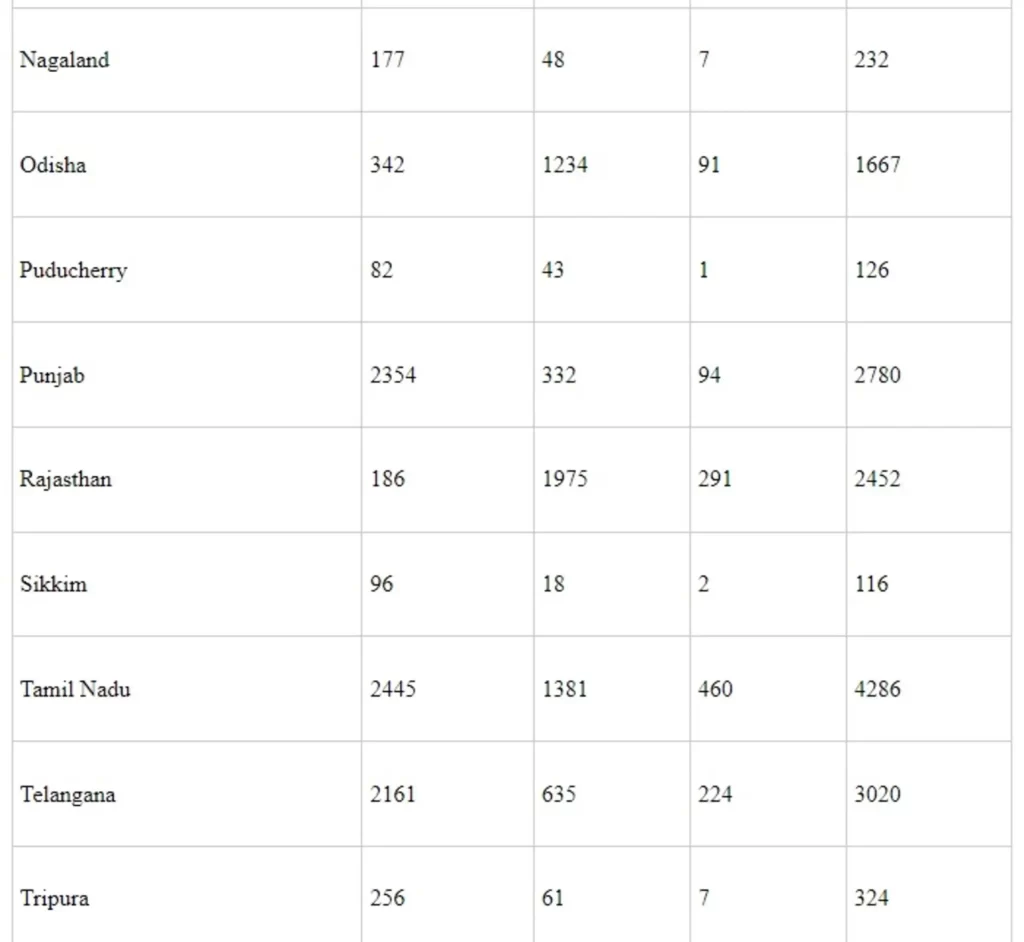
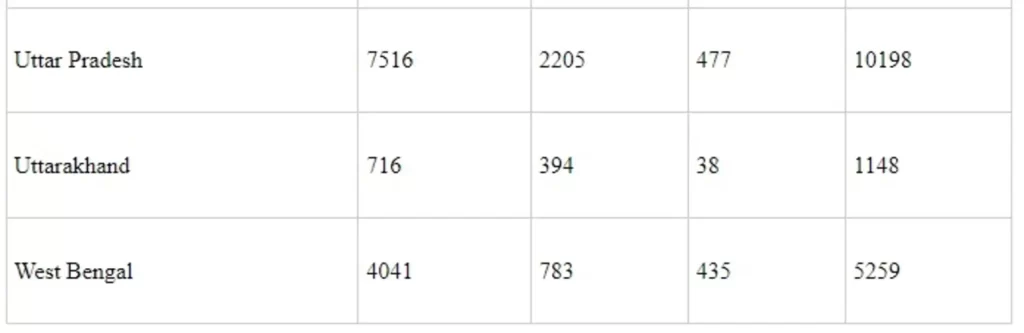
Annexure-II
State/UT wise Utilization under Ayushman Bharat Yojana - Health & Wellness Centres (AB-H&WCs) under NHM from the F.Y. 2018-19 to 2021-22





The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The Union Minister of Health & Family Welfare, Dr. Mansukh Mandaviya inaugurated the Ayushmann Bharath Digital Mission (ABDM) services of AIIMS Bibinagar today and witnessed the practical demonstration of real time video specialist consultation. ABDM is the initiative started by the Government to digitalise health records across the country and includes QR code-based patient registration, Health Management Information System (HMIS) a software to manage data in any Hospital including many digital services like patient registration, patient Que management, Lab information system, Doctors desk, OP Billing etc. Dr. Mandaviya said that “with the help of Ayushman Bharat Health Account (ABHA) card, patients can access their medical records anywhere, anytime without losing them all over India. This would enhance accessibility to their health records on the palm of their hands.”
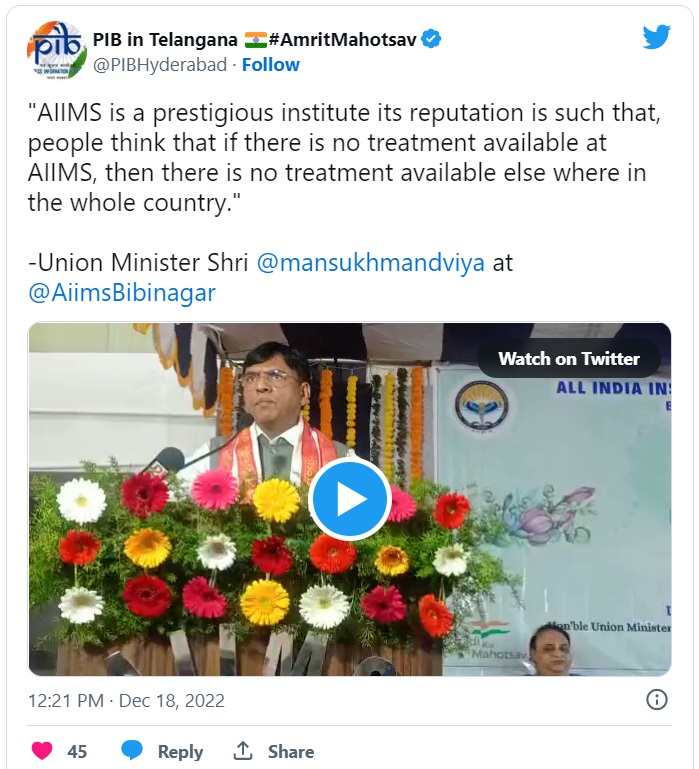
The Union Health Minister delivered the Maharshi Charak shapath the newly joined batch of MBBS students (2022-23) and congratulated the first- year students. He said that "AIIMS is a prestigious institute. Its reputation is such that, people think that if there is no treatment available at AIIMS, then there is no treatment available elsewhere in the whole country."

Speaking at the occasion, the Union Health minister called upon every medical student to inculcate the two important traits for success, commitment and dedication. Shri Mandaviya reiterated that health is never to be treated as business, it is a service to humanity. Recalling how India supplied critical medicines to rest of the world at the pre-COVID prices without taking advantage of the crisis, he said that was in sync with the principle of ‘Vasudaiva Kutumbakam’. He exhorted the teachers at AIIMS to ensure that the young doctors accord highest priority to serve the poorest of the poor. Union Health Minister also had lunch with students in the Institute’s canteen and he interacted with the students on various issues.

Under Swacch Bharath Abhiyaan, Herbal Plantation drive was taken up in the presence of Health Minister, where 101 herbal plants were planted by the newly joined 1st year MBBS students of AIIMS Bibinagar. The Minister was presented with progress report of AIIMS Bibinagar by Executive Director Dr. Bhatia later Dr. Mandaviya visited the project site of construction to review the work under progress.
Shri Komatireddy Venkat Reddy, Member of Parliament, Bhuvanagiri constituency, Executive Director Prof (Dr.) Vikas Bhatia. Dean (Academics) Prof (Dr.) Rahul Narang and Medical Superintendent, Prof (Dr.) Neeraj Agarwal also participated in the event.
References:YouTube Live link: https://youtu.be/SDaO_GexZjo
Tweet Links of AIIMS Bibi Nagar event: