The flagship scheme Ayushman Bharat Digital Mission (ABDM) is making steady progress in building a comprehensive digital health ecosystem for the country. With over 4 crore digital health records linked to the ABHA accounts of individuals, the scheme has crossed a significant milestone. More than 29 crore citizens have generated their unique Ayushman Bharat Health Accounts (ABHA) so far.
With their health records linked to their ABHA accounts digitally, citizens will be able to access and manage these records as per convenience. This enables citizens to create a comprehensive medical history across various healthcare providers thereby improving clinical decision-making. Further, the citizens can also share relevant health records with ABDM registered healthcare providers digitally.
Elaborating on ABDM’s role in digitizing health records, Dr. R. S. Sharma, CEO, NHA said – “As the implementing agency for ABDM, National Health Authority is working closely with different stakeholders to ensure that the benefits of digital health services can be availed by more citizens. We’re planning to launch an incentive scheme for health facilities such as hospitals, clinics, diagnostic labs etc. to further promote digitization of health records. We’re also encouraging different health locker applications to integrate with ABDM so that citizens get more choice for storing and managing their digital records.”
Adding further he said – “With continuous focus on digitization of health records, we aim to achieve paperless medical consultations and thereby more accuracy in each transaction between the patient and healthcare service provider.”
This digital linking of individual’s health records with ABHA is being carried out extensively across different health facilities of the country with the support of State Governments. The leading contributors to ABHA-linked health records include Government of Andhra Pradesh, Ayushman Bharat PM-JAY, Reproductive and Child Health (RCH) scheme under Ministry of Health and Family Welfare, eHospital and CoWin among others.
“For any society to move forward, research and innovation remain a crucial aspect. India has given push to indigenous research and this is reaping benefits for us now. ICMR-NARFBR has potential to make India a key global player in Biomedical research in 21st-century. Through Provision of quality services in support of biomedical research & training with adherence to highest international standards for human & ethical animal care and use, this resource facility can play a crucial role in improving health and welfare of the nation”, Union Health Minister, Dr. Mansukh Mandaviya stated this while inaugurating ICMR-NARFBR (National Animal Resource Facility for Biomedical Research) at Genome Valley, Hyderabad today in presence of Shri C. Malla Reddy, Minister of Labour and Employment, Telangana.


Speaking on the occasion, Dr. Mandaviya highlighted Government’s push for indigenous research and innovation. He said “during COVID pandemic, our Hon’ble Prime Minister emphasized on making indigenous vaccines. When world was suffering from the shortage of vaccines, India took up this challenge and our scientific community proved their mettle by creating those vaccines. When the import of foreign vaccines would have taken 5-10 years, with the political leadership’s wholehearted support and stakeholders being mobilised, India’s scientific community produced these vaccines in one year’s time.”
Significance of NARFBR can be adjudged from the fact that study of animals in biomedical research becomes crucial in discovering causes, diagnosis and treatment of zoonotic agents and diseases. NARFBR is an apex facility which will provide ethical care and use and welfare of laboratory animals during research. The newly build centre will work as the state-of-the-art facility for not just ethical animal studies but spans from basic, applied to regulatory animal research. It will help in capacity building of new researchers and will create processes for pre-clinical testing of new drugs, vaccines and diagnostics within the country along with quality assurance checks.
Celebrating India’s man power and brain power, Dr. Mandaviya said that Indians have remained at the forefront of creative fields, be it research institutions, Technology, or pharma companies etc. Highlighting India’s key role as pharmacy of the world, Dr. Mandaviya said that “for every four pills made in the world, one is made in India. Thus, we now want to make India a hub not just for medicine manufacturing but for pharma research as well. For this to happen, we need to create robust processes for clinical trials which in turn require animal facilities. Hence, NARFBR will play a key role in making this vision real.” He was also hopeful that through inauguration of NARFBR, this step would give boost to India’s Onehealth approach too.
Reiterating Government of India’s dedication towards creating a vibrant ecosystem of research and innovation in the country, Union Health Minister emphasized that government is taking several steps to ease the access to quality education and research training. He said that from giving boost to traditional medicine and modern medicine system to creating new institutions, and nudging private players, we are holistically moving forward for allowing our younger generation to creatively use their talents.
Dr. Rajiv Bahl, Director General of ICMR and Secretary of the Department of Health Research, termed the facility to be not just best in the country but biggest in the world. “From availability of various animals for ethical research to strengthening various processes under one umbrella, NARFBR would be an asset for the country to deal with zoonotic diseases” he added.
Live coverage of the event can be seen on YouTube here: Inaugurating the ICMR-National Animal Resource Facility For Biomedical Research - YouTube
Facebook: https://www.facebook.com/mansukhmandviya/videos/1413962979432636
Dr. Ramchandra S.G., Director NARFBR along with senior officials from Ministry and state were present at the event.
The National Health Authority (NHA) has released a consultation paper on ‘Operationalising Unified Health Interface (UHI) in India’ that outlines the market rules that will govern the UHI Network. UHI is envisioned as a foundational layer of the Ayushman Bharat Digital Mission (ABDM) and aims to enable interoperability in health services in India through open protocols.
The Consultation Paper focuses on the different elements of UHI and the market rules that will govern them. These include guidelines that will govern the way search & discovery will take place in a fair and transparent manner, payment & settlement processes, rules around cancellation & rescheduling, grievance redressal mechanism, and more. Each section has specific open questions where feedback from stakeholders is sought. Comments from the public are invited to ensure that the UHI Network is designed and operationalised in a collaborative and consultative manner.
Speaking about the importance of this consultation paper, Dr. R. S. Sharma, CEO, National Health Authority, stated, “Unified Health Interface will enable interoperability of health services in India. Since the development of UHI involves several stakeholders, it is important to clearly define how different elements will be operationalised in a fair, efficient and transparent manner. We urge the stakeholders to share their valued comments and play a role in shaping India’s digital healthcare ecosystem. This stakeholder participation can help avoid roadblocks in implementation, and help make adoption faster and easier.”
Full text of the consultation paper is available for download on ABDM’s website at https://abdm.gov.in/publications. Comments and feedback can be submitted here - https://abdm.gov.in/operationalising-uhi-consultation-form till Friday, 13th January 2023.
The incidence of TB in India has reduced by 18% from 256/lakh population in 2015 to 210/lakh population in 2021. At the same time, the estimates of Drug Resistant TB in India have reduced by 20% from 1.49 lakh in 2015 to 1.19 lakh in 2021.
The following measures have been taken by the Government to reduce the number of TB and MDR-TB cases in the country:
The Government of India has developed a National Strategic Plan (2017-25) for Ending TB in the country by 2025.The key focus areas are:
Early diagnosis of all the TB patients, prompt treatment with quality assured drugs and treatment regimens along with suitable patient support systems to promote adherence.
Engaging with the patients seeking care in the private sector.
Prevention strategies including active case finding and contact tracing in high risk / vulnerable population
Airborne infection control.
Multi-sectoral response for addressing social determinants.
Availability of molecular diagnostics has been scaled up to 4761 machines covering all districts in the country. In addition, 79 Line Probe Assay labs and 96 liquid culture testing laboratories have been established in the country for diagnosis of MDR-TB and XDR-TB.
Universal Drug Susceptibility Testing (UDST) is implemented under the NTEP to ensure every diagnosed TB patient is tested to rule out drug resistance before or at the time of treatment initiation itself.
Newer drugs like Bedaquiline and Delamanid have also been made available for management of DRTB.
Decentralized DR TB treatment services are offered through 162 Nodal DR-TB Centres and 614 District DR TB centres across the country.
Pradhan Mantri TB Mukt Bharat Abhiyan (PMTBMBA):
Launched by the Ministry on 9th September 2022 for community support to TB patients with the objective to provide people with TB with additional nutritional, diagnostic and vocational support.
Ni-kshay 2.0 portal has been developed and made available in public domain for facilitating the community to register as Ni-kshay Mitra
Guidance documents have been developed and shared with all State/UTs to implement the initiative.
Periodic reviews are done to monitor the progress of the initiative at National and State/UT levels.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
The National Health Mission (NHM) envisages achievement of universal access to equitable, affordable & quality health care services that are accountable and responsive to people’s needs. The main programmatic components include Health System Strengthening in rural and urban areas, Reproductive-Maternal- Neonatal-Child and Adolescent Health (RMNCH+A), and Communicable and Non-Communicable Diseases.
Under NHM, technical and financial support is provided to States/UTs for strengthening of their healthcare systems based on the proposals submitted by them in their Program Implementation Plans (PIPs), subject to availability of resources.
National Quality Assurance Standards (NQAS) was launched by Ministry of Health for Primary and Secondary cared public health facilities. As per NQAS guidelines for improving quality in public healthcare facilities all the institutions would undergo internal assessments, State assessment (once in year) and national assessment (once facility get 70% and more in State assessments).
The State/UT wise details of CHC quality assessed since 2020 till 30th November 2022 is as attached at Annexure-I.
‘LaQshya’ is a quality improvement initiative, launched in 2017 by MoHFW which aims to accelerate the efforts toward the reduction of preventable maternal mortality and morbidity by aiming to improve in Quality of Care during the delivery and immediate post-partum period. The initiative specifically focuses on the care around birth provided in the Labour room and Maternity OT. It has been initiated in all government medical college hospitals, district hospitals & equivalent health facilities, all designated FRUs and high case load CHCs.
The MusQan scheme was launched in 2021 to provide high-quality and safe Child-Friendly Services in Public Health Facilities and contribute to preventable new-born child mortality and morbidity. It makes the child-friendly services accessible and available to patients and their parents, as well as provide a child-friendly environment, promote, protect, and support breastfeeding, and provide respectful and dignified care to the mother/parent–attendant.
The Government of India has implemented the following programmes/schemes for ensuring assured, dignified, respectful and quality healthcare services for every pregnant woman.
Surakshit Matritva Aashwasan (SUMAN) provides assured, dignified, respectful and quality healthcare at no cost and zero tolerance for denial of services for every woman and newborn visiting public health facilities to end all preventable maternal and newborn deaths.
Janani Suraksha Yojana (JSY), a demand promotion and conditional cash transfer scheme for promoting institutional delivery.
Under Janani Shishu Suraksha Karyakram (JSSK), every pregnant woman is entitled to free delivery, including caesarean section, in public health institutions along with the provision of free transport, diagnostics, medicines, other consumables & diet
Pradhan Mantri Surakshit Matritva Abhiyan (PMSMA) provides pregnant women a fixed day, free of cost assured and quality antenatal check up by a Specialist/Medical Officer on the 9th day of every month.
Monthly Village Health, Sanitation and Nutrition Day (VHSND) is an outreach activity at Anganwadi centers for provision of maternal and child care including nutrition in convergence with the ICDS.
Delivery Points- Over 25,000 ‘Delivery Points’ across the country have been strengthened in terms of infrastructure, equipment, and trained manpower for provision of comprehensive RMNCAH+N services
MCP Card and Safe Motherhood Booklet are distributed to the pregnant women for educating them on diet, rest, danger signs of pregnancy, benefit schemes and institutional deliveries.
Reproductive and child health (RCH) portal is a name-based web-enabled tracking system for pregnant women and new born so as to ensure seamless provision of regular and complete services to them including antenatal care, institutional delivery and post-natal care.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
With the administration of 68,89,025 vaccine doses in the last 24 hours, India’s COVID-19 vaccination coverage has exceeded 134.61 Cr (1,34,61,14,483) as per provisional reports till 7 am today. This has been achieved through 1,41,10,887 sessions.
The break-up of the cumulative figureas per the provisional report till 7 am today include:
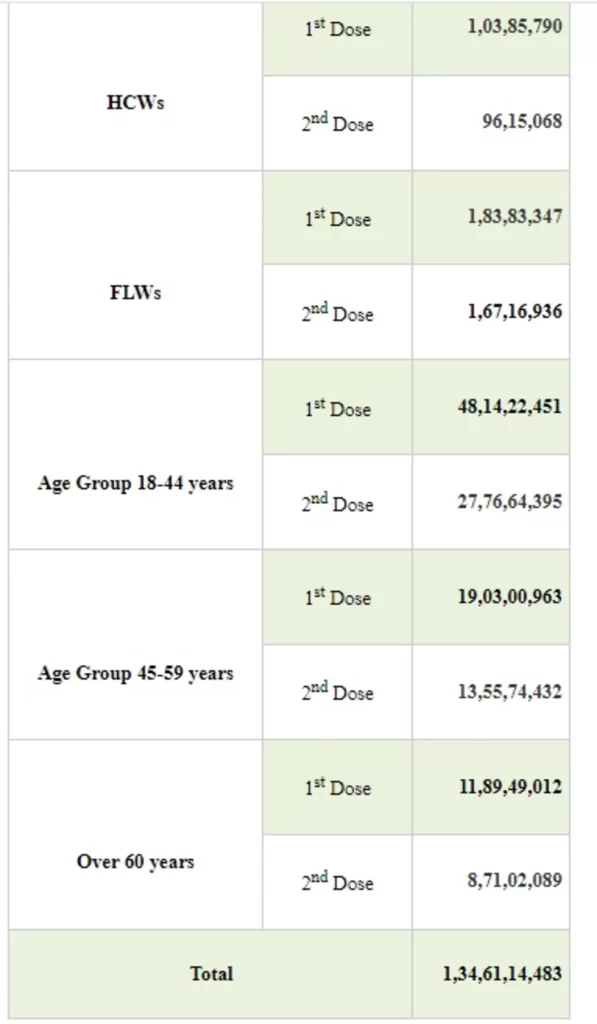
The recovery of 8,168 patients in the last 24 hours has increased the cumulative tally of recovered patients (since the beginning of the pandemic) to 3,41,46,931.
Consequently, India’s recovery rate stands at 98.38%,highest since March 2020.
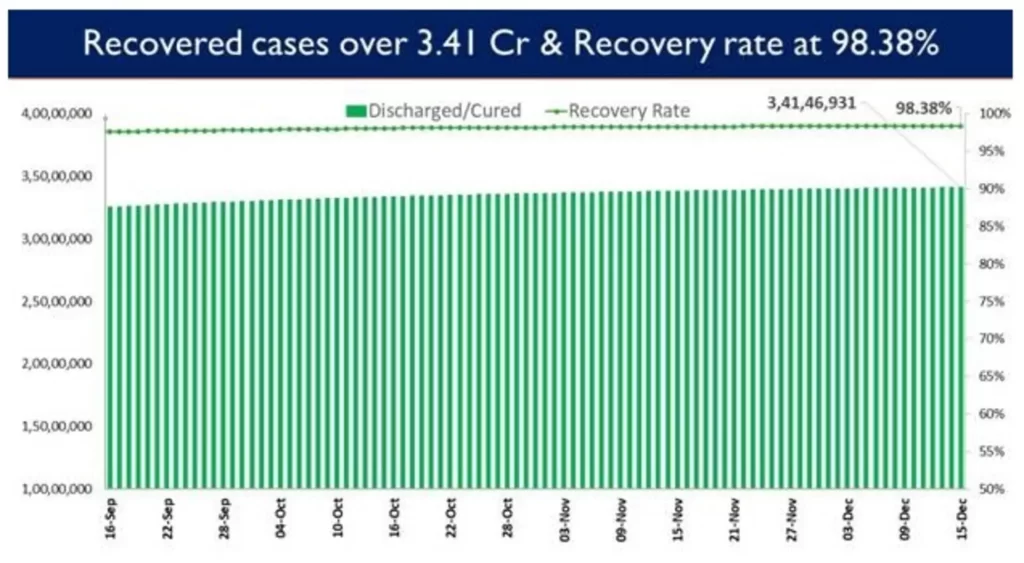
Sustained and collaborative efforts by the Centre and the States/UTs continue the trend of less than 15,000 Daily New Cases that is being reported for last 48days now.
6,984 new cases were reported in the last 24 hours.

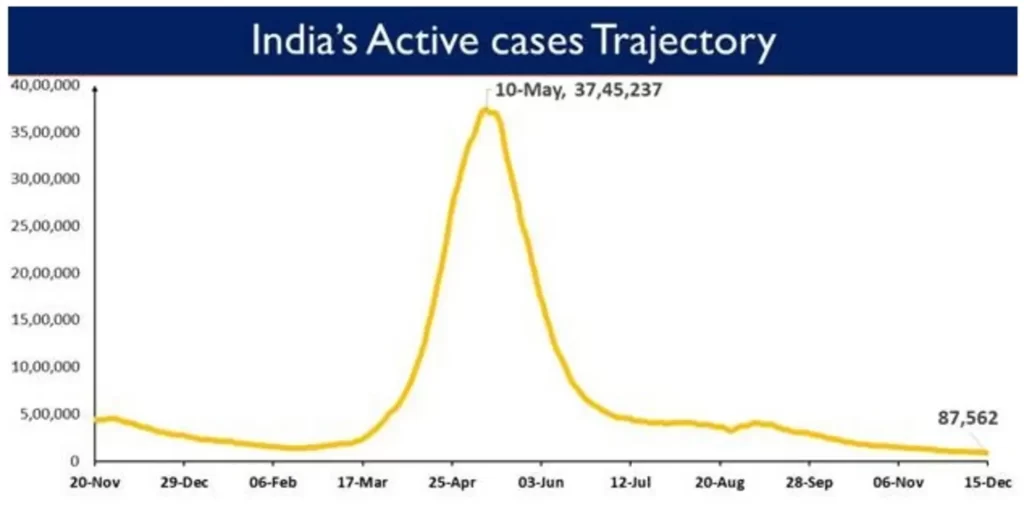
India’s Active Caseloadis presently at 87,562. Active cases constitute 0.25% of the country's total Positive Cases, which is lowest since March 2020.
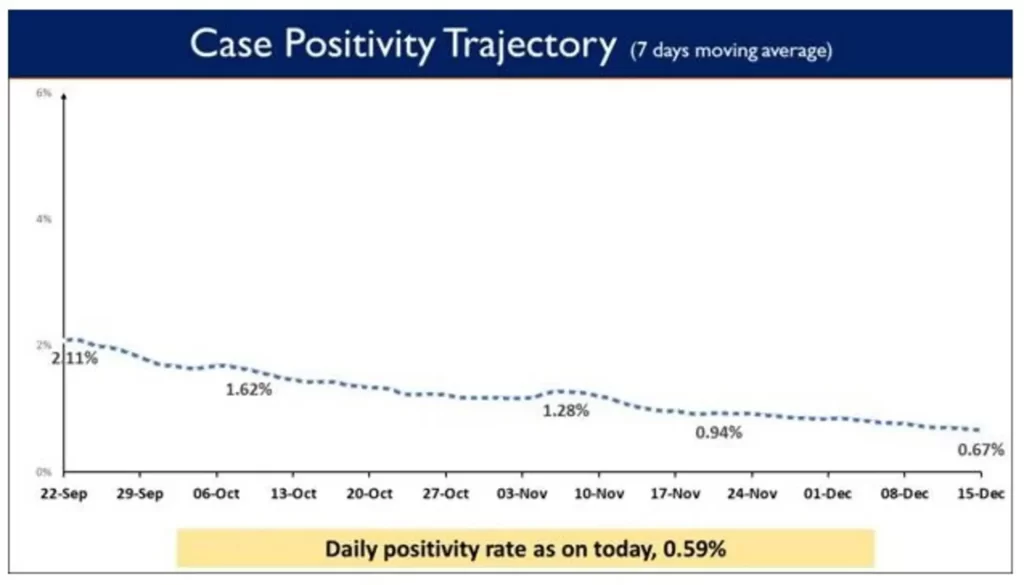
The testing capacity across the country continues to be expanded. The last 24 hours saw a total of 11,84,883 tests being conducted. India has so far conducted over 65.88 Cr (65,88,47,816) cumulative tests.
While testing capacity has been enhanced across the country, Weekly Positivity Rate at 0.67% remains less than 1% for the last 31 days now. The Daily Positivity rate reported to be0.59%. The daily Positivity rate has remained below2% for last 72 days and below 3% for 107 consecutive days now.
Medical oxygen is an essential public health commodity andits uninterrupted supply in sufficient quantity is of critical importance to tackle the pandemic. This was underlined by Shri Rajesh Bhushan, Union Health Secretary as he addressed the meeting with States and UTs to review the status and their preparedness regarding Medical Oxygen devices and systems (PSA Plants, LMO Plants, Oxygen Concentrators, Medical Gas Pipeline System), through a video conference today.
Informing the States and UTs that the Union Government has assisted them through availability of equipment, technical and financial support for PSA plants, oxygen concentrators, ventilators, oxygen cylinders, Liquid Medical Oxygen (LMO) plants and Medical Gas Pipeline systems (MGPS), the States were urged to review and monitor the status of these on a daily basis to ensure that the gap between the equipments and systems delivered to the Districts and installed at the healthcare facilities is reduced to zero. While equipment and systems have been sanctioned and delivered, in many states these have not been sent to the district healthcare facilities and when delivered, some have still not been made functional. State nodal officers were requested to streamline coordination with Defence Research and Development Organization (DRDO), HLL Infra Tech Services Limited (HITES) and Central Medical Services Society (CMSS) etc., for resolution of electricity related and site related issues for ensuring quick operationalization of the entire medical oxygen supply infrastructure supplied to them.
As on date, a total of 3236 PSA plants have been installed in the country from various sources with a total commissioned oxygen capacity of 3783 MT. Moreover, 1,14,000 Oxygen Concentrators are being provided to States under PM CARES (1 lakh) and ECRP-II (14,000).

It was also pointed out to the States that ECRP-II funds have been sanctioned to them for installation of 958 LMO Storage Tanks and medical gas pipeline systems in 1374 hospitals. States were advised to utilize this opportunity to enhance domestic oxygen production capacity and ensure the completion, installation and commissioning of medical gas pipelines in government hospitals expeditiously.
The States were strongly urged to schedule and conduct mock drills of all installed and commissioned PSA Plants to ensure that they are in fully operational status, so that the oxygen with the required quantity, pressure and purity reaches the intended patients at their bedside. These drills are to be completed by the end of December 2021. The report to this effect is to be submitted to the Union Health Ministry through designated portals for live tracking and monitoring of the functional status of these equipments. States were also requested to complete the pending Oxygen Audit reports and submit this through the designated portal by the end of December 2021.
The Union Health Ministry is conducting comprehensive training programmes to build and enhances capacities of technicians and clinicians for operation and maintenance of the PSA plants and other medical oxygen related infrastructure. States that have yet to complete the scheduled training programmes were urged to expedite them and complete the pending ones by December end, in coordination with the District Skill development Councils.
Dr Manohar Agnani, AS (HFW), Principal Secretary (Health), Mission Director (NHM) and State Surveillance Officers of all States participated in the review meeting. Representatives from Ministries of Coal, Power, Railways, Petroleum & Natural Gas were also present.
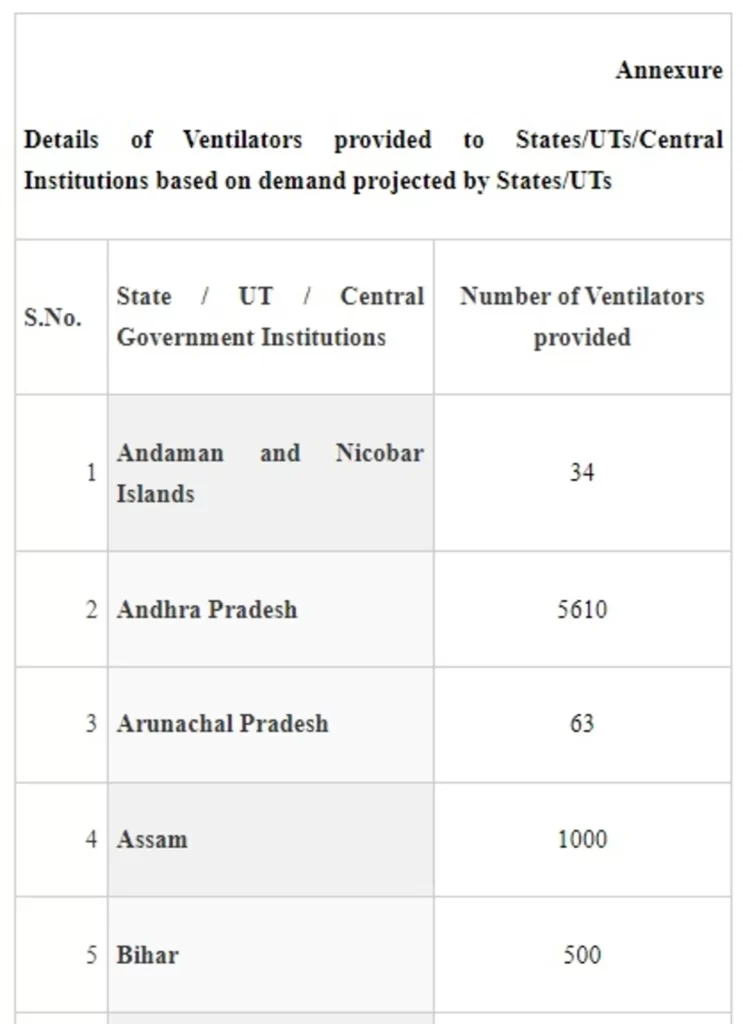
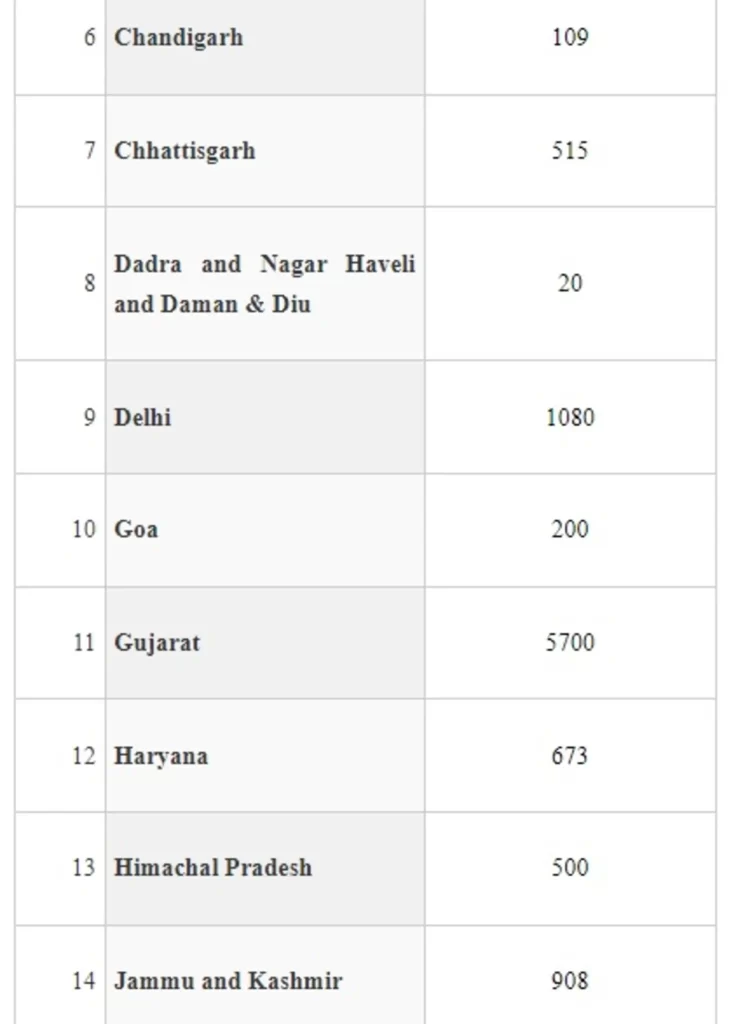
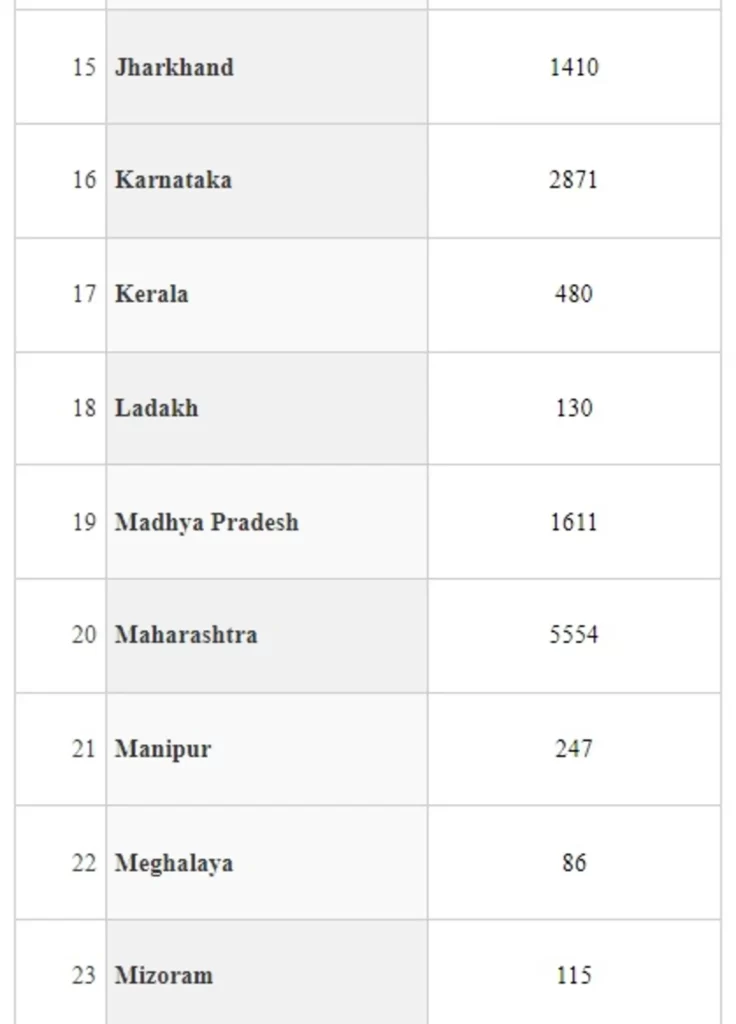
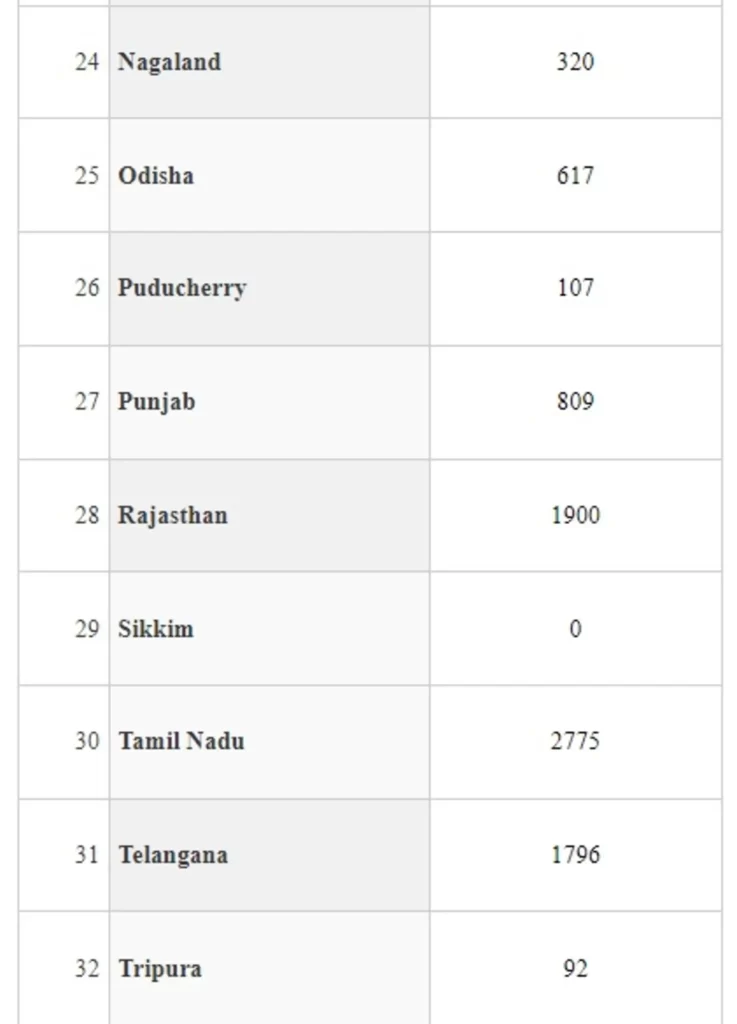
In order to ensure availability of Ventilators across the country for management of COVID-19 pandemic, Ministry of Health and Family Welfare ordered procurement of Ventilators centrally for supplying to the States/UTs based on the demand received from the States/UTs. The number of ventilators provided to State/UT is annexed at annexure.
The States have been provided with extensive training on operation of these ventilators and more than 19,000 Doctors and Para-medical workers have been trained on them. States have been advised to ensure that the ventilators are kept fully functional at all times. They have also been advised to ensure maintenance and up-keep of ventilators, preparedness of hospital infrastructure including optimum oxygen pressure in medical Gas pipeline Systems, sufficient availability of consumables for ventilators and operation of these ventilators by the trained manpower.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya
National Leprosy Eradication Programme (NLEP) is a Centrally Sponsored Scheme under the umbrella of National Health Mission (NHM). India has achieved the elimination of leprosy as a public health problem i.e., defined as less than 1 case per 10,000 populations, at the National level.
The NLEP aims at eliminating leprosy in each of the districts by 2030. NLEP Programme Progress from 2014 to 2021 (up to Sept.), Indicator wise is placed at Annexure.
Under the National Leprosy Eradication Programme action is taken for early case detection; complete treatment of detected cases, and to contain the onset of disease in close contacts of the index cases (persons diagnosed with leprosy).
Leprosy Case Detection Campaigns (LCDC) for 14 days in high endemic districts ASHA Based Surveillance for Leprosy Suspects (ABSULS)
Focused Leprosy Campaign (FLC) in low endemic districts for case detection Special plans for Hard-to-Reach areas for early case detection and treatment on time
The first two activities, namely, LCDC and ABSULS have now been clubbed together as “Active Case Detection and Regular Surveillance” (ACDRS) both in rural and urban areas in order to ensure detection of leprosy cases on regular basis and at an early stage in order to prevent Grade II disabilities.
Sparsh Leprosy Awareness Campaigns on 30th January
Convergence of leprosy screening under Rashtriya Bal Swasthya Karyakram (RBSK) for screening of children (0-18 years) and under Ayushman Bharat for screening of people above 30 years of age
NIKUSTH - A real time leprosy reporting software implemented across India Joint Monitoring and Investigation Group (JMIG) formed.
For encouraging the district heath functionaries, provision of certification and award to the districts for achieving leprosy elimination. Under two categories :1) Gold Category 2)Silver category
Contact tracing is done and Post Exposure Prophylaxis (PEP) with Single dose of Rifampicin (SDR) is administered to the eligible contacts of index case in order to interrupt the chain of transmission
Various services are provided under the programme for Disability Prevention and Medical Rehabilitation (DPMR), i.e., reaction management, provision of Microcellular Rubber (MCR) footwear, Aids & Appliances, self-care kits, etc. Reconstructive Surgeries are conducted at District Hospitals/Medical Colleges/ Central Leprosy Institutes, and welfare allowance @ Rs 8000 is paid to each patients undergoing RCS.
NLEP Programme Progress from 2014 to 2021 (September), Indicator wise.

The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
As on 3rd December 2021, 14,15,49,620 health IDs have been created in the country under the Ayushman Bharat Digital Mission (ABDM).
Ayushman Bharat Digital Mission (AB-DM) aims to create an online platform enabling interoperability of health data within the health ecosystem to create longitudinal electronic health record of every citizen, make healthcare accessible for citizens including reducing the cost of care and enabling greater efficiencies in health service delivery.
The digital health ecosystem created by ABDM supports continuity of care across primary, secondary and tertiary healthcare in a seamless manner. It ensures availability of health care services through electronic means particularly in remote and rural areas where generally such specialist care may not be available. As it reduces the need for repeated diagnostics through availability of longitudinal health records, it helps in reducing the cost of care.
Doctors, hospitals and others concerned can access the medical and other records of the individual only after consent for access is provided by the individual. Moreover, ABDM has been built on the principle of ‘privacy by design’ and federated architecture to facilitate secure data exchange between the intended stakeholders after the citizens’ consent.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
