The Government has put in place a Uniform Code for Pharmaceutical Marketing Practices (UCPMP) for Pharmaceutical companies, which is in operation since 01.01.2015, to prevent unethical practices by the pharmaceutical companies. This code governs the conduct of pharmaceutical companies in their marketing practices, duly covering the various aspects such as medical representatives, textual and audio-visual promotional materials, samples, gifts, etc. Further, the code establishes relationship with healthcare professionals, wherein the provisions related to travel facilities, hospitality and cash or monetary grants to physicians or their families have been elaborated.
The code also details the mode of operation of the code, responsibilities of the Pharmaceutical Associations in constituting the Ethics Committee for Pharmaceutical Marketing Practices (ECPMP) for handling the complaints and Apex Ethics Committee for Pharmaceutical Marketing Practices (AECPMP) for review, procedure of lodging a complaint, procedure of handling of complaints by the Pharmaceutical Associations and various penalty provisions.
The code has been adopted by the all the major associations of pharmaceutical companies and the Department on various instances has reviewed implementation of the code by the Pharmaceuticals associations.
The complaints of violation of the voluntary UCPMP by pharma companies which are perceived by the Department are forwarded to the concerned associations for taking necessary action.
The Indian Medical Council (Professional Conduct, Etiquette and Ethics) Regulations, 2002 formed under Indian Medical Council Act, 1956 (102 of 1956), provides for conduct for doctors and professional association of doctors in their relationship with pharmaceutical and allied health sector industry. Under this, any complaint of professional misconduct of a medical practitioner or professional is to be addressed by the respective State Medical Councils.
Besides UCPMP, there exists sufficient and enforceable legal regime to counter, control and dis-incentivize the unethical marketing practices such as “Indian Medical Council Professional Conduct, Etiquette and Ethics) Regulations, 2002” under the Indian Medical Council Act, 1956, provisions available under the Income Tax Act, Drugs and Cosmetics Act, Prevention of Corruption Act, etc.
The Department has not received any representations in the recent past from civil societies and patents group to make the UCPMP code mandatory. Further, in the writ petition (Civil) No. (s).323/2021 filled by federation of the Medical and Sales Representatives Associations of India & others against Union of India and others, the matter is under consideration of Hon’ble Supreme Court of India.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Lok Sabha today.
The Ministry of Health and Family Welfare, Government of India supports ‘Free Diagnostics Service Initiative’ (FDSI) programme under National Health Mission. The programme was launched in July 2015 with the aim to provide accessible and affordable pathological and radiological diagnostics services closer to the community which in turn reduces the Out-of-Pocket Expenditure (OOPE). A minimum set of essential diagnostic tests has been recommended as per the Essential Diagnostic List at each level of public health facility.
A guidance document for implementing laboratory services has been shared with all states and UTs. The guidelines also recommend expanded range of diagnostics at all levels of public health facilities (14 tests at Sub Centres (SCs), 63 tests at Primary Health Centres (PHCs), 97 tests at Community Health Centres (CHCs), 111 tests at Sub-District Hospitals (SDHs) and 134 tests at District Hospitals (DHs).
Under NHM, ‘Free Diagnostic Service initiative’ is delivered through in-house, Public Private Partnership (PPP) and Hybrid mode by the States/UTs in the country. Free Laboratory service is operational in 33 States, out of which 11 States/UTs (Arunachal Pradesh, Assam, Delhi, Himachal Pradesh, Jharkhand, Maharashtra, Manipur, Meghalaya, Odisha, Tripura, and Uttarakhand) are providing the services through PPP/hybrid mode and 22 States/UTs (Andhra Pradesh, A&N Island, Bihar, Chandigarh, Chhattisgarh, D&N Haveli-Daman & Diu, Goa, Gujarat, Haryana, Jammu & Kashmir, Karnataka, Kerala, Lakshadweep, Madhya Pradesh, Nagaland, Puducherry, Punjab, Sikkim, Tamil Nadu, Telangana, Uttar Pradesh, West Bengal) provide the services through In-house mode.
Further, free Tele-Radiology (Tele-reporting of X-rays) service is operational in 13 states/UTs (Andhra Pradesh, Assam, Bihar, Himachal Pradesh, Jammu & Kashmir, Maharashtra, Meghalaya, Odisha, Rajasthan, Uttarakhand, Uttar Pradesh, and West Bengal, Tripura). Free CT Scan service is operational in 27 States/UTs out of which 17 States (Andhra Pradesh, Assam, Chandigarh, Chhattisgarh, Delhi, Haryana, Himachal Pradesh, Jharkhand, Karnataka, Madhya Pradesh, Meghalaya, Maharashtra, Odisha, Rajasthan, Tripura, Uttar Pradesh, West Bengal) are providing it in PPP mode and 10 states (A&N island, Bihar, D&N Haveli-Daman & Diu, Goa, Jammu & Kashmir, Lakshadweep, Kerala, Puducherry, Tamil Nadu, Telangana) provide through In-house mode.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
The Government is implementing the National Leprosy Eradication Programme (NLEP) with the goal of making India leprosy free. NLEP is a Centrally Sponsored Scheme under the umbrella of National Health Mission (NHM). This programme is implemented in all the States/UTs.
The major focus of the Programme is to detect the cases of leprosy at an early stage and to provide complete treatment, free of cost, in order to prevent the occurrence of disability.
Major initiatives under NLEP are as follows:
Leprosy Case Detections Campaign (LCDC) in high endemic districts.
Focused Leprosy Campaign (FLC) in low endemic districts for case detection.
Special plans for Hard-to-Reach areas for early case detection and treatment.
ASHA Based Surveillance for Leprosy Suspects (ABSULS).
Sparsh Leprosy Awareness Campaign on 30th January every year.
Active Case Detection and Surveillance both in rural and urban areas.
Convergence of leprosy screening under Rashtriya Bal Swasthya Karyakram (RBSK) for screening of children (0-18 years) and under Ayushman Bharat for screening of people above 30 years of age.
Monitoring of the programme by Joint Monitoring Investigation and Advisory Group (JMIAG).
Certificate, award and performance linked financial incentive are given to the districts for achieving leprosy elimination.
Contact tracing is done and Post Exposure Prophylaxis (PEP) with Single dose of Rifampicin (SDR) given to the eligible contacts of index case in order to interrupt the chain of transmission.
Further, 12 packs of MDT (Multi Drug Therapy) blister packs are given to Multi Bacillary Leprosy cases as per the Standard Treatment Protocol.
India has achieved the elimination of leprosy as a public health problem as per WHO criteria of less than 1 case per 10,000 population at the National level in 2005. However, there are few districts within States where leprosy is still endemic.
As per Monthly Progress Report of Central Leprosy Division, Directorate General of Health Services (as on 31st May 2022), 72,914 leprosy patients, as informed by the States/UTs, are under treatment in the country.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
Ministry of Health & Family Welfare (MoH&FW) publishes the National List of Essential Medicines (NLEM), which is incorporated in the Schedule-I of the Drugs Prices Control Order (DPCO). National Pharmaceutical Pricing Authority (NPPA) under the aegis of Department of Pharmaceuticals fixes the ceiling price of scheduled formulations as per the provisions of the DPCO, 2013. Schedule-I of DPCO, 2013 was last amended by adopting NLEM, 2015 consisting of 377 medicines.
NPPA has fixed the ceiling prices of 890 scheduled formulations of medicines under NLEM, 2015 till 30.06.2022. In addition, NPPA has fixed retail price of 2,023 new drugs under DPCO, 2013. Further, NPPA has also capped the Trade Margin of non-scheduled formulations of 42 Anti-cancer medicines under ‘Trade Margin Rationalization’ approach as a Pilot for proof of concept, wherein price of more than 500 brands of medicines were reduced up to 90%.
The percentage reduction in prices varies based on the previous ceiling price fixation of 890 scheduled formulations. The details of price of drugs regulated by NPPA are available on its website, viz., www.nppaindia.nic.in.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Lok Sabha today.
The Department implements the scheme “Promotion of Medical Devices Parks”, with a total financial outlay of Rs. 400 crore and the maximum assistance under the scheme for one Medical Device Park would be limited to Rs. 100 crore. The tenure of the scheme is from FY 2020-2021 to FY 2024-2025 and the selected Medical Device Park project will be implemented by a State Implementing Agency (SIA). Under the scheme, Department of Pharmaceuticals has received proposals from 16 States/Union Territories. The proposals were evaluated as per the criteria given in the scheme guidelines and final approval for financial assistance of Rs. 100 crore each has been given to the States of Uttar Pradesh, Tamil Nadu, Madhya Pradesh and Himachal Pradesh. There is no proposal to establish more medical device parks in the Country, under the scheme.
Further, the Department has provided grant-in-aid to the project of the Superconducting Magnet Testing, Validation and Integration Centre at AMTZ, Andhra Pradesh under the scheme “Assistance to Medical Device Industry for Development of Common Facility Centres”.
The Government of India has taken several measures to encourage domestic manufacturing of Pharmaceutical drugs including bulk drugs and medical devices to reduce import dependence. The Programmatic interventions to support Pharma and Medical Devices Industries are as follows;
The Production Linked Incentive (PLI) Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) in India, with a financial outlay of Rs. 6,940 crores and the tenure from FY 2020-2021 to FY 2029-30, provides for financial incentive for 41 identified products. A total of 51 applicants have been selected under the scheme.
The Production Linked Incentive Scheme for Pharmaceuticals, with a financial outlay Rs. 15,000 crores and the tenure from FY 2020- 2021 to FY 2028-29, provides for financial incentive to 55 selected applicants for manufacturing of identified products under three categories for a period of six years, including five (5) industry applicants selected for In-vitro diagnostic medical devices.
The Scheme for Promotion of Bulk Drug Parks, with a financial outlay of Rs. 3,000 crores and the tenure from FY 2020-2021 to FY 2024-25, provides for financial assistance to three States for establishing Bulk Drug Parks. The proposals received are under evaluation.
The Department has launched the scheme of Strengthening of Pharmaceutical Industry (SPI), with a financial outlay of Rs. 500 crores and the tenure from FY 2021-2022 to FY 2025-26 and this scheme has three components, to provide infrastructure support for pharma MSMEs in clusters and to address the issues of technology upgradation of individual pharma MSMEs.
Under the scheme “Promotion of Medical Devices Parks”, final approval for financial assistance of Rs. 100 crore each, has been given to the States of Uttar Pradesh, Tamil Nadu, Madhya Pradesh and Himachal Pradesh for establishment of common facilities in their Medical Device Parks.
Further, under the sub-scheme “Assistance to Medical Device Industry for Common Facility Centre”, grant-in-aid of ₹ 25 crore was provided to Andhra Pradesh Medtech Zone Ltd. (AMTZ), Andhra Pradesh for establishment of Common Facility for Super conducting magnetic coil testing and research facility.
The Production Linked Incentive (PLI) Scheme for Promoting Domestic Manufacturing of Medical Devices, with a financial outlay of Rs.3,420 Cr and with the tenure from FY 2020-21 to FY 2027-28, provides for financial incentives to selected companies at the rate of 5% of incremental sales of medical devices manufactured in India and covered under the four Target segments of the scheme, for a period of five (5) years. A total of 21 Applicants have been selected under the scheme.
The non-schematic interventions are as follows:
In order to attract investments in this sector, the Government has allowed 100% foreign direct investments (FDI) in medical devices sector. Similarly, the Government has allowed 100% FDI in pharma sector for greenfield projects under automatic route. For the brownfield projects, upto 74%, FDI investments are allowed under automatic route and beyond 74% to 100%, FDI investments are allowed under government approval route.
To redress the specific challenges of the MedTech Industry, in view of the diversity and multi-disciplinary nature of the sector, the institutional mechanism of Standing Forum of Medical Devices Associations, has been set up to deliberate on various issues with all the stakeholders including regulators.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Lok Sabha today.
Government is aware about the challenges posed by anti-microbial resistance (AMR) in India and has taken following measures to address the issue:
AMR surveillance network has been strengthened by establishing labs in State Medical College. 36 sites in 26 States/UTs have been included in this network so far.
National action plan on containment of Antimicrobial Resistance (NAP-AMR) focusing on One Health approach was launched in April 2017 with the aim of involving various stakeholder ministries/departments. Delhi Declaration on AMR– an inter-ministerial consensus was signed by the ministers of the concerned ministries pledging their support in AMR containment.
AMR Surveillance Network: ICMR has established AMR surveillance and research network (AMRSN) comprising 30 tertiary care hospitals, both private and government to generate evidence and capture trends and patterns of drug resistant infections in the country.
AMR Research & International Collaboration: ICMR has taken initiatives to develop new drugs /medicines through international collaborations in order to strengthen medical research in AMR.
ICMR along with Research Council of Norway (RCN) initiated a joint call for research in antimicrobial resistance in 2017.
ICMR along with Federal Ministry of Education and Research (BMBF), Germany has a joint Indo-German collaboration for research on AMR.
Government of India has taken following initiatives to increase awareness about antibiotics misuse:
ICMR has initiated antibiotic stewardship program (AMSP) on a pilot project basis in 20 tertiary care hospitals across India to control misuse and overuse of antibiotics in hospital wards and ICUs. DCGI has banned 40 fixed dose combinations (FDCs) which were found inappropriate.
ICMR worked in collaboration with Indian Council of Agriculture Research, Department of Animal Husbandry, Dairy and Fisheries and the DCGI to ban use of Colistin as growth promoter in animal feed in poultry.
Various IEC activities like public conclave, poster and quiz competitions have been conducted by National Centre for Disease Control (NCDC) in schools, colleges and Health melas to create awareness about AMR, its containment & prevention and judicial use of antibiotics among the common public.
To raise awareness among the community and the health care providers, communication material includes posters, videos and radio jingles has been developed with emphasis on prevention of irrational use of antibiotics during viral-illnesses and also on infection prevention through hand hygiene to prevent spread of infections.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
The healthcare system of the country involves a three-tier system with Sub Health Centre (Urban and Rural), Primary Health Centre (Urban and Rural) and Community Health Centre (Urban and Rural) as the three pillars of Primary Health Care System in India. Rural Health Statistics (RHS) is an annual publication, based on Health care administrative data reported by States/UTs.
Similarly, District Hospital (DH), Sub-District Hospital (SDH) and First Referral Unit - Community Health Centres provide secondary care services whereas Medical Colleges and State-level Institutions provide tertiary care services.
Ministry of Health and Family Welfare, Government of India provides technical and financial support to the States/UTs to strengthen the public healthcare facilities. To address the healthcare challenges, particularly in rural areas, the National Rural Health Mission (NRHM) was launched in 2005 to supplement the efforts of the State/UT governments to provide accessible, affordable and quality healthcare to all those who access public health facilities. Currently, NRHM is a sub-mission of National Health Mission (NHM).
Support is provided to states/UTs under the National Health Mission (NHM) for strengthening of health care facilities as per norms based on the requirements posed by them in their Programme Implementation Plans (PIPs) and within their overall resource envelope. Under NHM, States are supported for several interventions towards strengthening the primary healthcare services in the country.
XV-Finance Commission Health Grants through Local Government announced as part of Union Budget 2021-22, for strengthening the healthcare system at primary healthcare level, has recommended grants aggregating to Rs. 70,051 Crores over the period of five years (2021-2026) through local government to strengthen the healthcare system in states.
Pradhan Mantri Ayushman Bharat Health Infrastructure Mission (PM-ABHIM) with an outlay of Rs. 64,180 Crores aims to fill critical gaps in public health infrastructure especially in critical care facilities and primary care in both urban and rural areas. It is a Centrally Sponsored Scheme with some Central Sector components. The Centrally Sponsored Scheme (CSS) Components includes support for infrastructure development for Sub-Health Centres, Urban Health and Wellness Centres, Support for Block Public Health Units, Integrated District Public Health Laboratories and Critical Care Hospital Blocks.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
Ministry of Health and Family Welfare (MoHFW) conducts one integrated survey namely National Family Health Survey (NFHS) at an interval of about 3 years and so far, completed five rounds of survey. NFHS provides high quality, reliable and comparable data on population dynamics and health indicators as well as data on emerging issues in health and family welfare and associated domains, so as to assist the policy-makers and programme implementing agencies for setting the benchmarks. NFHS also provides evidence on the effectiveness of ongoing programmes, which are implemented by the Government of India.
As per the fifth round of National Family Health Survey NFHS conducted by MoHFW during the year 2019-21, the Total Fertility Rate (TFR) has declined to 2.0 children per woman from 2.2 children per woman as per the fourth round of NFHS conducted during the year 2015-16, resulting in the achievement of the replacement level of fertility, which is 2.1 children per woman.
The progress of some of the key Health & Family Welfare Indicators as per NFHS-5 as compare to NFHS-4 are as follows:

The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The National Health Authority (NHA) under its flagship scheme Ayushman Bharat Digital Mission (ABDM) is building a comprehensive digital health ecosystem for the country. Since nationwide launch in September 2021, ABDM has achieved significant growth with over 23 crore ABHA numbers (earlier known as Health ID), 1.14 Lakh health facilities registered in the Health Facility Registry (HFR), 33 thousand healthcare professionals under the Healthcare Professionals Registry (HPR), 6.6 lakh ABHA app downloads and 3.4 Lakh health records linked to ABHA of individuals.
With more individuals, healthcare professionals like doctors, nurses, paramedics and health facilities like hospitals, nursing homes, wellness centres, clinics, diagnostic labs, pharmacies joining ABDM, digitization of health records at their point of creation is now possible. For digitization of old health records, individuals can use the ABHA app or any other personal health record (PHR) app to scan and save their records. By linking these digital records to their ABHA, individuals will be able to connect with professionals and facilities digitally and get quality and affordable health services irrespective of geographic distance.
Several states/ UTs which have made significant contribution to the progress of the scheme so far are given below:
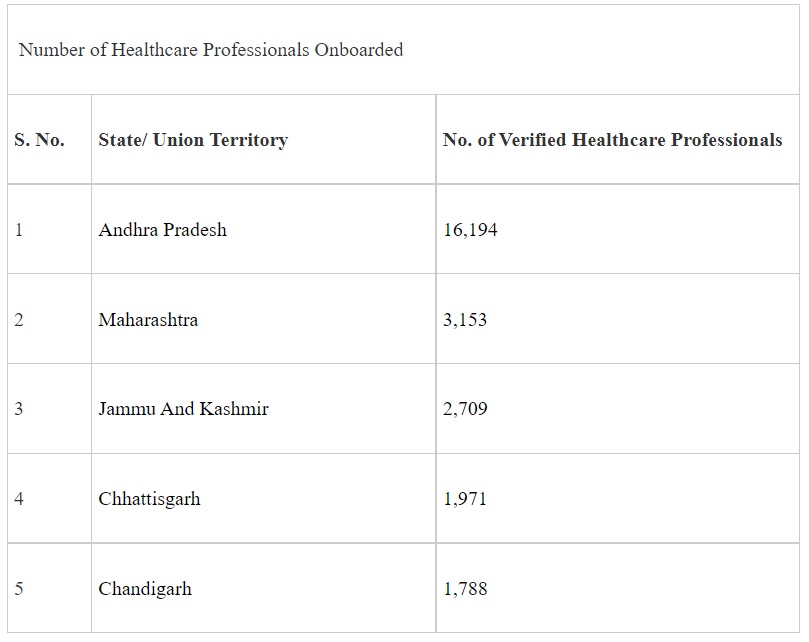
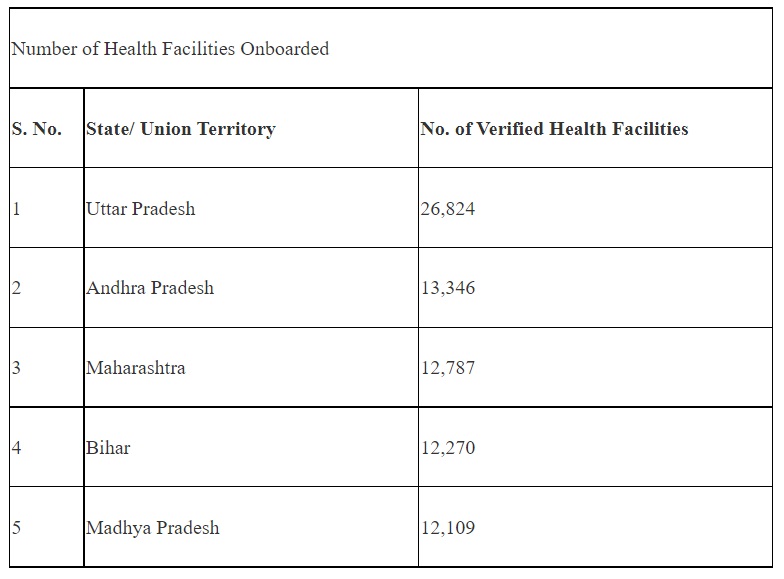
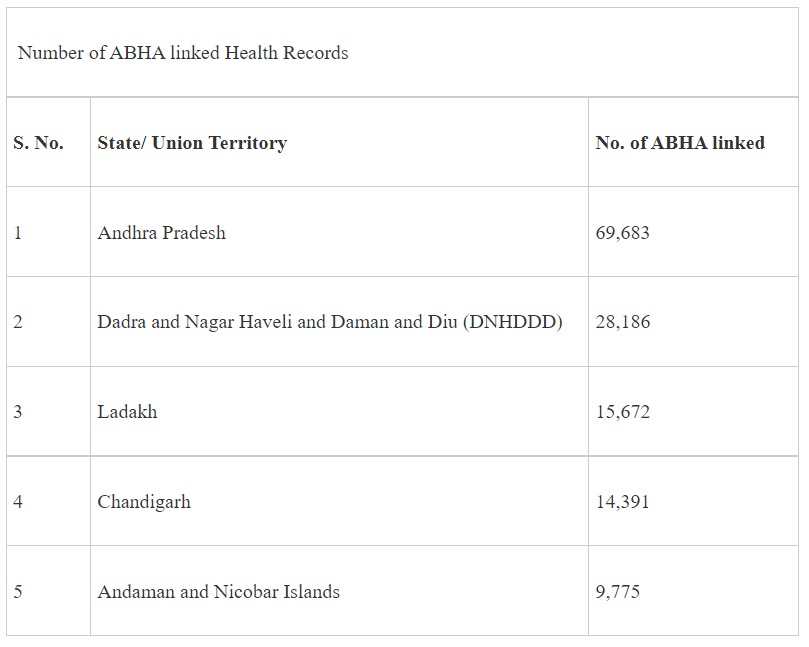
Acknowledging the cooperation and active involvement of the respective States & UTs in taking the mission forward and improving access and affordability of healthcare services, NHA will recognize the top performing states/ UTs, districts and health facilities (public as well as private) for their contribution under ABDM. The States/ UTs, districts and health facilities contributing the most in a 50-day period from 01.08.2022 to 19.09.2022 would be recognized during the first anniversary of the scheme under 4 categories as follows:
Category A: States/UTs with highest number of health records linked to ABHA(s) in the state/UT from 01.08.2022 to 19.09.2022 per one lakh population.
Category B: Districts with highest number of health records linked to ABHA(s) in the district from 01.08.2022 to 19.09.2022 per one lakh population.
Category C: States/UTs achieving highest percentage saturation in population of HFR and HPR registries in private and public Sector by 19.09.2022.
Category D: Facilities from government and private sector with highest number of health records linked to ABHA(s) in 50-day period from 01.08.2022 to 19.09.2022.
In addition to the above categories, NHA will recognize top performing ABDM integrators (digital health solutions integrated with the scheme) on the basis of number of health records linked to ABHA numbers within 50 days period from 01.08.2022 to 19.09.2022. The top performing integrators from government and public sector shall be recognized in separate categories.
An individual can create their Ayushman Bharat Health Account (number) (ABHA) which is a unique identifier for a citizen in the health ecosystem. Presently, ABHA can be created by an individual through their Aadhaar number or driving license.
ABHA will help the individual to link and share their digital health records with consent across multiple systems and stakeholders. This helps create their longitudinal health records across different healthcare providers which facilitates better delivery of healthcare to them.
Ministry has taken multiple measures to accelerate ABHA adoption in the country. National Health Authority (NHA) is collaborating with several government health programmes, government health solutions/applications, private integrators to accelerate creation of ABHA. For ABHA creation in remote locations with limited or no internet connectivity, ABDM has also rolled out the feature of offline ABHA creation.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
