The Government of India implements Anaemia Mukt Bharat (AMB) strategy under POSHAN Abhiyaan with the target for reducing anaemia in the six population groups - Children (6-59 months), Children (5-9 years), Adolescents girls and boys (10-19 years), Pregnant women, Lactating women and Women of Reproductive Age (WRA) group (15-49 years) in life cycle approach. Some of the major interventions by Ministry of Health and Family Welfare (MoHFW) to address anaemia under AMB include:
Prophylactic Iron and Folic Acid Supplementation. Deworming.
Intensified year-round Behaviour Change Communication (BCC) Campaign and delayed cord clamping.
Testing of anaemia using digital methods and point of care treatment.
Addressing non-nutritional causes of anaemia in endemic pockets with special focus on malaria, hemoglobinopathies and fluorosis.
Convergence and coordination with line department and other ministries.
Engaging National Centre of Excellence and Advanced research on Anaemia Control for capacity building of health care providers.
Monitoring progress in States/UTs using Anaemia Mukt Bharat Dashboard.
As per the fifth round of National Family Health Survey (NFHS), conducted by MoHFW during the year 2019-21, the prevalence of anaemia among women age 15-49 is 57.0 percent as compared to 53.1 percent in NFHS-4, (2015-16). However, thirteen (13) States/UTs namely Andhra Pradesh, Andaman and Nicobar Island, Arunachal Pradesh, Chandigarh, Dadra and Nagar Haveli and Daman and Diu, Haryana, Himachal Pradesh, Lakshadweep, Meghalaya, NCT of Delhi, Tamil Nadu, Uttar Pradesh and Uttarakhand have reported decline in prevalence of anaemia among women age 15-49 in NFHS-5 as compared to NFHS-4.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
It is primarily the responsibility of the State/UT Government to take concrete steps to prevent illegal activities taking place in blood banks and during blood transfusion. Government has made registration of blood banks mandatory as per the provisions of Drugs and Cosmetic Act and Rules thereunder. Further, Government has notified Drugs and Cosmetics (Second Amendment) Rules, 2020 which pertain to functioning of Blood Banks/centres, Blood processing and related matters. The implementation & monitoring of aforesaid Act and Rules is within the ambit of the States/UTs. A letter has been issued to States/UTs to take concrete steps for prevention of such illegal activities.
Further, the e-RaktKosh web portal of Government of India, provides the interface for Blood Banks data management and integration. For this, all blood banks have been advised to register on e-RaktKosh web-portal.
It is also the primary responsibility of the State/UT Government to ensure the establishment of blood banks as per their need. The policy of the Government advocates for a hub and spoke approach towards transfusion services, wherein blood is collected and processed in the hubs, which are high volume blood banks and distributed through spokes, which are smaller blood banks and blood storage centers. The requirement of blood banks depends upon the health infrastructure of the area. However, blood storage centres can be established in remote areas to meet the emergency requirement of blood, which may be attached to blood banks.
Support is provided to States/UTs to strengthen their healthcare system including strengthening existing blood banks/supporting new blood banks and blood storage units based on the proposals submitted by the States through National Health Mission. Government of India through Blood Transfusion Services also supports 1131 Blood Banks in the public and charitable sector in respect of manpower, procurement of Blood Bags and testing kits, conducting Voluntary Blood Donation (VBD) camps and Information Education Communication (IEC) activities etc. In addition, Mobile vans for Blood collection and blood transfusion vans are provided to the States/UTs.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
Ayushman Bharat –Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) is governed on a zero-tolerance approach to any kind of fraud viz. suspect/non-genuine medical treatment claims, impersonation and up-coding of treatment packages/procedures etc.
The Government of India adopts a pro-active approach towards fraud and abuse. It goes on to underscore that several countervailing strategies have been employed under PM-JAY to address various types of fraud and it acknowledges the success of the AI-based technologies deployed under the scheme.
National Health Authority -the implementing agency of AB-PMJAY has issued a comprehensive set of anti-fraud guidelines. Anti-fraud advisories are issued to States/UTs. National Anti-Fraud Unit (NAFU) is created at NHA for overall monitoring and implementation of anti-fraud framework supported by State Anti-Fraud Units (SAFUs) at State level. All claims require mandatory supporting documents along-with on-bed patient photo before approval and payment. The feature of Aadhar-based biometric verification of beneficiary at the time of admission and discharge is launched at all private hospitals. Use of artificial intelligence and machine learning is made for a comprehensive fraud analytics solution to detect fraud pro-actively, develop algorithms that can be used on large volume of data to identify suspect transactions and entities and risk scoring of hospitals and claims. Around 0.18% of the total authorized hospital admissions under the scheme are confirmed as fraud since its inception.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The manufacturing licenses for generic drugs and new drugs are granted by the various State Drug Controllers and the Drug Controller General of India (under Ministry of Health and Family Welfare) respectively. A common list of all such manufacturers of generic drugs in the private sector is not maintained in the Department of Pharmaceuticals. However, three PSUs under Department of Pharmaceuticals viz. Bengal Chemicals and Pharmaceuticals Limited (BCPL) in West Bengal, Hindustan Antibiotics Limited (HAL) in Maharashtra and Karnataka Antibiotics & Pharmaceuticals Limited (KAPL) in Karnataka manufacturers generic medicines.
The Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP) was launched by the Department with an objective of making quality generic medicines available at affordable prices to citizens of the country. Under the Scheme, dedicated outlets known as Pradhan Mantri Bhartiyta Janaushadhi Kendras (PMBJPs) are opened to provide quality generic medicines at affordable prices. Under the PMBJP, till 30.06.2022, 8742 PMBJKs have been opened across the country including Government Hospitals and Government premises. The Scheme has a product basket of 1,616 medicines and 250 surgical supplies. Prices of medicines sold through these outlets are 50-90% less than that of branded medicines prices in the open market.
Public Health and Hospitals, being the State subject, the responsibility of providing medicines lies with the respective State Governments and the details of medicines prescribed and provided to the patients are not available with the Department of Pharmaceuticals. Under National Health Mission (NHM), support is provided for provision of essential generic drugs free of cost in public health facilities. Further, CGHS procures generic medicine through Medical Store Organisation of the Ministry of Health and Family Welfare.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Rajya Sabha today.
The Indian Pharmaceutical industry is the 3rd largest in the world by volume. India is one of the major producers of Active Pharma Ingredients (API) or bulk drugs in the world. India exported Bulk Drugs/Drug Intermediates worth Rs. 33,320 crore in financial year 2021-22. However, the country also imports various Bulk Drugs/APIs for producing medicines from various countries and most of the imports of the Bulk Drugs/APIs being done in the country are because of economic considerations. India imported Rs. 35,249 crore worth APIs and Bulk drugs in 2021-22.
In order to make the country Atmanirbhar in APIs and drug intermediates, the Department of Pharmaceuticals is implementing the following three schemes by attracting large investments in the sector to ensure their sustainable domestic supply and thereby reduce India’s import dependence on other countries: -
The Production Linked Incentive (PLI) Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) in India, with a financial outlay of Rs. 6,940 crores and the tenure from FY 2020-2021 to FY 2029-30, provides for financial incentive for 41 identified products. A total of 51 applicants have been selected under the scheme.
The Production Linked Incentive Scheme for Pharmaceuticals, with a financial outlay Rs. 15,000 crores and the tenure from FY 2020- 2021 to FY 2028-29, provides for financial incentive to 55 selected applicants for manufacturing of identified products under three categories for a period of six years. The eligible drugs under this scheme include APIs.
The Scheme for Promotion of Bulk Drug Parks, with a financial outlay of Rs. 3,000crores and the tenure from FY 2020-2021 to FY 2024-25, provides for financial assistance to three States for establishing Bulk Drug Parks.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Rajya Sabha today.
As per the District Residency Program notified by the Board of Governors, all the post graduate students pursuing MD/MS in broad specialties in all Medical Colleges/Institutes under the Indian Medical Council Act, 1956 shall undergo a compulsory residential rotation of three months in District Hospitals/ District Health System as a part of the course curriculum.
All the administrative and personnel matters related to health human resource including specialist doctors of various streams of medicines in rural and remote hilly regions lies with the respective State/UT Governments. However, under National Health Mission, Ministry of Health & Family Welfare provides financial and technical support to States/UTs to strengthen their healthcare systems based on the requirements posed by them in their Programme Implementation Plans (PIPs) within their overall resource envelope.
Under NHM, following incentives and honorarium are provided for encouraging doctors to practice in rural and remote hilly regions of the country:
• Hard area allowance to specialist doctors for serving in rural and remote areas and for their residential quarters so that they find it attractive to serve in public health facilities in such areas.
• Honorarium to Gynecologists/ Emergency Obstetric Care (EmoC) trained, Pediatricians & Anesthetist/ Life Saving Anaesthesia Skills (LSAS) trained doctors is also provided to increase availability of specialists for conducting Cesarean Sections in rural & remote area.
• States are also allowed to offer negotiable salary to attract specialist including flexibility in strategies such as “You Quote We Pay”.
• Non-Monetary incentives such as preferential admission in post graduate courses for staff serving in difficult areas and improving accommodation arrangement in rural areas have also been introduced under NHM.
2 years Diploma course is conducted for graduate doctors which can be pursued through NEET exam. Further, the said course is not limited to graduate doctors working in district hospitals only.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The Department has recently issued the guidelines of the scheme “Strengthening of Pharmaceutical Industry (SPI) on 11th March 2022. The scheme is applicable to all parts of the country. The objectives of the scheme are as follows:
To strengthen the existing infrastructure facilities in order to make India a global leader in the Pharma Sector by providing financial assistance to pharma clusters for creation of Common Facilities.
To upgrade the production facilities of SMEs and MSMEs, to meet national and international regulatory standards, by providing interest subvention or capital subsidy on their capital loans.
To promote knowledge and awareness about the Pharmaceutical and Medical Devices Industry by taking up studies, building databases and brining industry leaders, academia and policy makers together to share their knowledge and experience.
The total financial outlay of the scheme is Rs.500 crore for a period of five years from 2021-22 to 2025-26. The scheme has three sub-components Viz, Assistance to Pharmaceuticals Industry for Common Facilities (API-CF), Pharmaceutical Technology Upgradation Assistance Scheme (PTUAS) and Pharmaceutical & Medical Devices Promotion and Development Scheme (PMPDS).
Under PTUAS, support is provided to MSME units in pharmaceutical sector for quality & technology upgradation. It is planned to provide support to about 420 MSME units, during the scheme period of SPI.
Under API-CF, support is provided to the pharma clusters for creating common infrastructure facilities. It is planned to provide support to 10 projects during the scheme period of SPI. A project of Inducare Pharmaceuticals and Research Foundation (IPRF) for creation of Common Facility Centre at Pune, Maharashtra with grant-in-aid support of Rs 20.00 Crore was approved in FY 20-21 under then API-CF. A total grant-in-aid of Rs. 18.00 Crore has been released till date.
Department of Pharmaceuticals strives to improve the infrastructural facilities of the pharma sector in the country towards making India, a global leader in the sector.
In order to make the country Atmanirbhar in pharmaceuticals, the Department of Pharmaceuticals has launched the Production Linked Incentives (PLI) Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/Drug Intermediates (DIs), and Active Pharmaceuticals Ingredients (APIs) in India. The Total financial outlay of the scheme is Rs. 6,940 crore and the tenure from FY 2020-2021 to FY-2029-30. A total of 51 applicants have been selected under the scheme.
Another ‘Production Link Incentive (PLI) scheme for Pharmaceuticals has been launched with total financial outlay of Rs. 15,000 crore and tenure from FY 2020-2021 to FY 2028-29. The scheme intends to enhance India’s manufacturing capabilities by increasing investment and production in the sector and contributing to product diversification to high value goods in pharmaceuticals sector. The eligible drugs under this scheme include APIs among other categories of pharmaceuticals products.
The Scheme for Promotion of Bulk Drug Parks, with a financial outlay of Rs. 3,000 crores and the tenure from FY 2020-2021 to FY 2024-25, provides for financial assistance to three States for establishing Bulk Drug Parks.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Rajya Sabha today.
“Let us change our attitude as we step out of our homes for work. A positive attitude and healthy mental framework along with dedication and commitment are pivotal building blocks for a healthy and progressive nation”. Union Health Minister Dr Mansukh Mandaviya stated this as he addressed the inaugural ceremony of training programme for senior administrative medical officers of CGHS along with Dr Bharati Pravin Pawar, Union Minister of State here today.
A week long training and orientation program has been organised at National Institute for Health and Family Welfare (NIHFW) for CGHS officers, under the guidance of the Union Health Minister to enhance and upgrade their skills at interpersonal communication, administration and use of technology.

Dr Mansukh Mandaviya stressed on importance of “Samvaad” as a strong tool for interpersonal communication and grievance redressal. “Those organisations and individuals that are in a learning mode will always progress. We shall always benefit from a “Vidyarthi bhaav” where we are open to imbibing knowledge, new insights and learn from each other”, he stated. The Union Minister stated that empathy, care and softer skills further refine and enhance our technical and clinical abilities. Many challenges are resolved with a positive attitude, he stressed. He shared experiences to highlight the importance of being open to suggestions to enhance one’s attitude towards work.

Highlighting the achievements of CGHS, Dr Bharati Pravin Pawar, Union Minister of State said that CGHS as an organization has widened its network and is now functional in 75 cities across the country with approximately 450 wellness centers.Over the period, it has undergone many changes to keep pace with the developments in the health sector such as digitization of services and inclusion of various new health modalities. Planning and implementation of these changes has been only possible due to consistent efforts and hard work of the entire workforce, she stated.
She further added, “It is commendable that CGHS has adopted good administration practices like the recently introduced exercise of ‘CGHS Panchayat’. Such practices help in assessing the different aspects of CGHS service system like the wellness centers, the empanelment procedures, bill reimbursement etc. Interactive meetings held with all stake holders of CGHS such as staff and the beneficiaries in various cities to address their grievances and receive feedbacks if any is a welcome initiative. Health resources – both human and material are precious assets to the society and a robust health care administration and management system ensures their effective utilization.” She also re-emphasized on Hon’ble Prime Minister’s mantra of skill, re-skill and up-skill.
Central Government Health Scheme (CGHS) is a unique Health Scheme providing comprehensive health care to its nearly 41.2 lakh beneficiaries who are mainly central government employees and pensioners, through its 460 wellness centres spread across the country in 75 cities. The wellness centres provide holistic health care services to around 55000-60000 beneficiaries daily besides there being a provision of number of empanelled Health Care Organizations including Super Speciality Hospitals.
CGHS in collaboration with NIHFW, a premiere training institute under the Ministry of Health and Family Welfare, has initiated training programmes for various categories of employees in CGHS. The training programme has been launched for Senior Administrative Medical Officers who are holding administrative posts in various CGHS offices/Wellness Centres or are likely to hold these positions near future. The training will be held from 1st August to 6th August 2022 and around 70 of these officers will be trained in two batches. The six-day comprehensive training programme aims at capacity building of Senior Administrative Medical Officers in CGHS for their holistic development, in alignment with the CGHS vision and mission, which include not only upgrading of administrative and managerial skills but also enhancing for creating a better and more humane personality.
Shri Alok Saxena, Additional Secretary, Central Government Health Scheme (CGHS), Dr.Nikhilesh Chandra, Director, CGHS, Ms. NidhiKesarwani, Director, NIHFWand other eminent dignitaries were also present at the event.
Under National Health Mission (NHM), technical and financial support to the States is given as per the proposals submitted by the States/UTs in the form of Programme Implementation Plans (PIPs) based on the requirements, including maternal and child health and state specific innovations, and the approvals are accorded as Record of Proceedings (RoP) to States/UTs.
Further, one of the components of the 12-packages to deliver Comprehensive Primary Healthcare under Ayushman Bharat – Health & Wellness Centres (AB-HWC) scheme is “Care in pregnancy and childbirth” and includes:
Early diagnosis and registration of pregnancy and issuing of ID number and Mother and Child protection card
Ensuring four antenatal care checks of high risk pregnancies including screening of Hypertension, Gestational Diabetes, Anaemia, HIV, Hepatitis b, Thyroid Profile for hype- or hyper- thyroidism, syphilis, etc.
Referral and follow up of the high risk pregnancy cases.
Counselling regarding care during pregnancy including information about nutritional requirements
Facilitating institutional delivery and supporting birth planning
The Government has formulated several schemes/ programmes for facilitating check-ups and screening of pregnant women, which are as follows:
Surakshit Matritva Aashwasan (SUMAN) provides assured, dignified, respectful and quality healthcare at no cost and zero tolerance for denial of services for every woman and newborn visiting public health facilities to end all preventable maternal and newborn deaths.
Pradhan Mantri Surakshit Matritva Abhiyan (PMSMA) provides pregnant women a fixed day, free of cost assured and quality antenatal check up by a Specialist/Medical Officer on the 9th day of every month.
Monthly Village Health, Sanitation and Nutrition Day (VHSND) is an outreach activity at Anganwadi centers for provision of maternal and child care including nutrition in convergence with the ICDS.
MCP Card and Safe Motherhood Booklet are distributed to the pregnant women for educating them on diet, rest, danger signs of pregnancy, benefit schemes and institutional deliveries. Reproductive and child health (RCH) portal is a name-based web-enabled tracking system for pregnant women and new born so as to ensure seamless provision of complete services to them including antenatal care, institutional delivery and post-natal care.
Outreach camps are provisioned for improving the reach of health care services especially in tribal and hard to reach areas. This platform is used to increase the awareness for the Maternal & Child health services, community mobilization as well as to track high risk pregnancies.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
Government of India has the National Organ Transplant Programme (NOTP) to promote organ donation and transplantation across the country. The provisions under the programme include financial support for:
Setting up of State Organ and Tissue Transplant Organizations (SOTTOs) in each State/UT.
Setting up of National/ Regional/State Bio-material centres;
Establishing new Organ Transplant/retrieval facilities and strengthening of existing Organ Transplant/retrieval facilities.
Provision of Transplant Coordinators to medical colleges and trauma centres.
Maintenance of deceased donor
Dignified funeral of deceased donor
Post transplant immune-suppressant drugs to BPL patients.
Under this programme, an apex level National Organ and Tissue Transplant Organisation (NOTTO) at New Delhi, five Regional Organ and Tissue Transplant Organizations (ROTTOs) and sixteen State Organ and Tissue Transplant Organizations (SOTTOs) have been established.
Organs like Heart, Kidney, Liver etc. need to be transplanted immediately after retrieval. The National Organ and Tissue Transplant Organisation (NOTTO) has a facility of National Level Tissue Bank (Biomaterial Centre) for storing tissues. Under the National Organ Transplant Programme (NOTP), a provision has been made for providing financial support to the States for setting up of Bio- material centre. A Regional Bio-material centre has been established at Regional Organ and Tissue Transplant Organisation (ROTTO), Chennai, Tamil Nadu. Further, funds have been released to the State of Bihar and Maharashtra for establishment of Bio-material Centre under NOTP.
The NOTTO, ROTTOs and SOTTOs disseminate relevant information about organ donation in the public. A website www.notto.gov.in and a 24x7 call centre with a toll free helpline number (1800114770) have been made operational. A number of activities, for generating awareness and for imparting training to all those associated with transplant including doctors and transplant coordinators, such as celebration of Indian Organ Donation Day, seminars, workshops, debates, sports events, walkathons, participation in marathons, nukkad natak, etc. are organized at different places in the Country. Awareness activities through advertisement in print media, audio-visual messages and expert talks on Doordarshan and other television channels are carried out in this regard. Jan Andolan for creating Organ donation awareness and pledging in the form of social media campaign has been carried out as part of Amrit Mahotsav to celebrate 75th anniversary of independence of country.
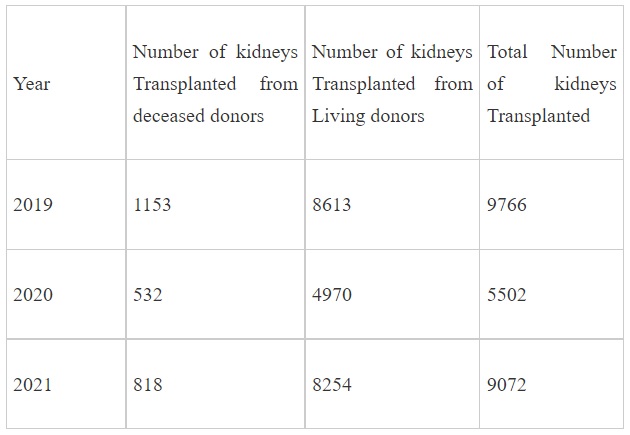
As per data made available by States/UTs to National Organ and Tissue Transplant Organisation (NOTTO), the number of patients for kidney transplant in the country along with the number of kidneys available for such transplants are as under:
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
