The Indian Pharmaceutical industry is the 3rd largest in the world by volume. India exported pharmaceuticals worth Rs. 1,75,040 crore in the financial year 2021-22, including Bulk Drugs/ Drug Intermediates. India exported about Rs 33,321 crore and imported Rs 35,249 crore worth APIs and Bulk drugs in 2021-22.
In order to make the country Atmanirbhar in APIs and drug intermediates, the Department of Pharmaceuticals is implementing the following three schemes by attracting large investments in the sector to ensure their sustainable domestic supply and thereby reduce India’s import dependence on other countries: -
The Production Linked Incentive (PLI) Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) in India, with a financial outlay of Rs. 6,940 crores and the tenure from FY 2020-2021 to FY 2029-30, provides for financial incentive for 41 identified products. A total of 51 applicants have been selected under the scheme.
The Production Linked Incentive Scheme for Pharmaceuticals, with a financial outlay Rs. 15,000 crores and the tenure from FY 2020- 2021 to FY 2028-29, provides for financial incentive to 55 selected applicants for manufacturing of identified products under three categories for a period of six year. The eligible drugs under this scheme include APIs.
The Scheme for Promotion of Bulk Drug Parks, with a financial outlay of Rs. 3,000 crores and the tenure from FY 2020-2021 to FY 2024-25, provides for financial assistance to three States for establishing Bulk Drug Parks. The proposals received are under evaluation.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Rajya Sabha today.
Department of Pharmaceuticals implements the Scheme for Promotion of Bulk Drug Parks to facilitate setting up of Three (3) Bulk Drug Parks in the country with the objective to bring down the cost of manufacturing of bulk drugs by creation of world class common infrastructure facilities.
The financial assistance by the centre is subject to a maximum limit of Rs.1000 Crore per park or 70% of the project cost of CIF ( 90% in case of North Eastern States and Hilly States i.e. Himachal Pradesh, Uttarakhand, UT of Jammu & Kashmir and UT of Ladakh), whichever is less. The total financial outlay of the scheme is Rs. 3000 crore and the tenure of the Scheme is from 2020-21 to 2024-25. The proposals received from the States under the scheme are under evaluation.
Under the scheme, financial assistance would be provided for creation of Common Infrastructure Facilities (CIF) like (i) Central Effluent Treatment Plant(s) (CETP) (ii) Solid waste management (iii) Storm water drains network (iv) Common Solvent Storage System, Solvent recovery and distillation plant (v) Common Warehouse (vi) Dedicated power sub-station and distribution system with the necessary transformers at factory gate (vii) Raw, Potable and Demineralized Water (viii) Steam generation and distribution system (ix) Common cooling system and distribution network (x) Common logistics (xi) Advanced laboratory testing Centre, suitable for even complex testing/ research needs of APIs, including microbiology laboratory and stability chambers (xii) Emergency Response Centre (xiii) Safety/ Hazardous operations audits centre and (xiv) Centre of Excellence etc. in any upcoming Bulk Drug Park promoted by State Government/State Corporation.
The common infrastructure facilities created under the scheme will help reduce the manufacturing cost of bulk drugs and also enhance the competitiveness of the domestic industry.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Rajya Sabha today.
As per information provided by National Medical Commission (NMC), there are 13,08,009 allopathic doctors registered with the State Medical Councils and the National Medical Commission (NMC) as on June, 2022. Assuming 80% availability of registered allopathic doctors and 5.65 lakh AYUSH doctors, the doctor-population ratio in the country is 1:834 which is better than the WHO standard of 1:1000. Also, there are 34.33 lakh registered nursing personnel and 13 lakh Allied and Healthcare Professionals in the country.
The Government has taken following steps to further increase the availability of doctors in the country, particularly in underserved regions. The number of UG seats have increased from 51,348 before 2014 to 91,927 seats which is an increase of 79%. The numbers of PG seats have increased by 93% from 31,185 seats in 2014 to 60,202 seats. The steps to increase doctor-patients ratio include: -
Centrally Sponsored Scheme for establishment of new medical college by upgrading district/ referral hospital under which 157 new medical colleges have been approved and 72 are already functional.
Centrally Sponsored Scheme for strengthening/ upgradation of existing State Government/Central Government Medical Colleges to increase MBBS and PG seats.
Central Sector Scheme for upgradation of Government Medical Colleges by construction of Super Specialty Blocks. A total of 75 projects have been approved and 55 have been completed.
Under Central Sector Scheme for setting up of new AIIMS, 22 AIIMS have been approved. Undergraduate courses have started in 19 AIIMS.
Relaxation in the norms for setting up of Medical College in terms of requirement for faculty, staff, bed strength and other infrastructure.
DNB qualification has been recognized for appointment as faculty to take care of shortage of faculty.
Enhancement of age limit for appointment/ extension/ re-employment against posts of teachers/dean/principal/ director in medical colleges upto 70 years.
The tenure of Senior Residency for appointment of Assistant Professor have reduced from three year to one year.
Under the District Residency Programme (DRP), all the doctors admitted to PG courses from 2022 onwards, to serve in the district hospitals for three months mandatorily.
Further, with a view to augment the availability of qualified manpower in field of mental health, the Government, under National Mental Health Programme (NMHP), implements manpower development schemes for establishment of Centres of Excellence and strengthening/ establishment of Post Graduate (PG) Departments in mental health specialties. Support has been provided for establishment of 25 Centres of Excellence and strengthening/ establishment of 47 Post Graduate (PG) Departments in mental health specialties in the country.
The Government also augments the availability of manpower to deliver mental healthcare services in the underserved areas of the country by providing online training courses to various categories of general healthcare medical and para medical professionals through the Digital Academies established at the three Central Mental Health Institutes namely National Institute of Mental Health and Neuro Sciences, Bengaluru, Lokopriya Gopinath Bordoloi Regional Institute of Mental Health, Tezpur, Assam, and Central Institute of Psychiatry, Ranchi.
Besides the above, the Government has announced a “National Tele Mental Health Programme” in the Budget of 2022-23, to further improve access to quality mental health counselling and care services in the country.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The National Health Authority (NHA) will be hosting its first ever open-to-all hackathon series to drive ideation and innovations around the different building blocks in the Ayushman Bharat Digital Mission (ABDM) ecosystem. The series will begin with a hackathon - ‘Round 1: Kickstarting Unified Health Interface (UHI)’ slated for 14th to 17th July 2022. The hackathon shall be focussed on the Unified Health Interface (UHI) and would aim at mobilising the health start-up ecosystem in India and bringing together individuals and organisations to develop solutions through UHI.
The hackathon will provide an excellent opportunity for innovators, data experts and developers from all over the world to collaborate and build innovative solutions for providing access to digital health services through UHI.
UHI is designed as an open network to enable the interoperable transaction of digital health services such as teleconsultation. Through UHI, patients can discover, book, conduct and pay for services offered by a variety of participating providers from any application of their choosing. This stands in contrast to the current mode of digital health service delivery, where patients and doctors must transact using a common application or platform.
Announcing the Hackathon Series, Dr. R.S. Sharma, CEO, NHA said “India has enabled creation of some of the most efficient public delivery systems in the world, as has been witnessed through JAM and UPI, by breaking silos and unlocking network effects. ABDM and UHI will enable the same for the healthcare ecosystem and this Hackathon Series will enable players to collaborate and build solutions which define the future of healthcare. All are invited to participate in creating a better health ecosystem for every Indian”
‘ABDM Hackathon Series - Round 1: Kickstarting UHI’ will focus on two major themes:
Innovation Track: Challenge for innovative solutions to power digital health in an open network around various use cases such as teleconsultation, ambulance booking, lab tests, physical consultation booking, lab tests booking.
Integration Track: Challenge to accelerate development of applications that are compatible with UHI and integrate these applications with the applications of other such participants to enable digital health transactions on the UHI network.
The tentative prize pool for ‘Round 1 – Kickstarting UHI’ isRs. 60,00,000*. This shall be awarded to the top performers on each challenge track on the above-mentioned themes. The solutions shall be assessed by an independent jury. Interested participants are requested to register here https://abdm.gov.in/register
*The prizes are tentative; the jury might decide to award fewer or more prizes.
Further, as part of the UHI hackathon, NHA shall be organising masterclasses to further the participants' understanding of the ABDM ecosystem.The details about upcoming masterclasses, challenge statements, prizes under each categoryand other updates shall be posted on ABDM website - https://abdm.gov.in and UHI portal - https://uhi.abdm.gov.in.
The first round of ABDM Hackathon Series will be followed by a Health Claims Exchange (HCX) Hackathon; the details will be shared on the ABDM and UHI website.
For further details on UHI, the full text of the UHI Consultation Paper, as well as the recording of the UHI Public Webinar may be referred to. They are available for download on the ABDM website at https://abdm.gov.in/publications
In pursuance of provisions of the National Pharmaceuticals Pricing Policy, 2012 (NPPP, 2012) and the Essential Commodities Act, 1955, the Government notified the Drugs (Prices Control) Order, 2013 (DPCO-2013). The National Pharmaceutical Pricing Authority (NPPA), an attached office of the Department of Pharmaceuticals fixes the ceiling price of all scheduled formulations specified in the first schedule of the DPCO, 2013.All manufacturers of scheduled medicines have to sell their products within the ceiling price (plus applicable Goods and Service Tax) fixed by the NPPA. It also fixes retail price of new drugs under DPCO, 2013 for existing manufacturers of scheduled formulations. In respect of non-scheduled formulations, a manufacturer is at liberty to fix its Maximum Retail Price (MRP) but cannot increase the same by more than 10% of what was prevalent during the preceding 12 months.
The details of drugs, including medical devices, brought under price control/ regulation by NPPA are as under:
(i) Ceiling prices of 890 scheduled formulations across various therapeutic categories under NLEM 2015 have been fixed by NPPA.
(ii) Retail price of approx. 2,023 new drugs under DPCO, 2013 have been fixed.
(iii) MRP of 106 non-scheduled drug formulations capped, including that for 22 diabetic and 84 cardiovascular drugs.
(iv) Ceiling price of Orthopaedic Knee Implants fixed under Para 19 of DPCO, 2013.
(v) Trade Margin of non-scheduled formulations of 42 select Anti-cancer medicines capped under ‘Trade Margin Rationalization’ approach as a pilot for proof of concept.
(vi) Regulated the price of Oxygen Concentrators, Pulse Oximeter, Blood Pressure Monitoring Machine, Nebulizer, Digital Thermometer and Glucometer under ‘Trade Margin Rationalisation’ Approach.
National List of Essential Medicines (NLEM) notified by the Ministry of Health & Family Welfare consists of essential medicines falling under various therapeutic categories. These medicines are incorporated in the First Schedule of the DPCO, 2013 and their ceiling prices is fixed by NPPA as per provisions of DPCO, 2013. The details of ceiling prices and retail prices of various formulations fixed by NPPA are available at its website at nppaindia.nic.in.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Lok Sabha today.
The Ministry of Health and Family Welfare has released the National Report of the fifth round of National Family Health Survey (NFHS), which is available on the public domain (http://rchiips.org/nfhs/).The major findings evident from NFHS-5 (2019-21) as compared to NFHS-4 (2015-16) is given at Annexure I.
The information on various important indicators captured in NFHS-5 are used for monitoring the SDG related health goals. India has made significant progress in various SDG health indicators like Total Fertility Rate (TFR), the proportion of pregnant women who had an ANC visit in the first trimester, Institutional Births, Child immunization, etc. List of SDG indicators, where NFHS-5 is the data source is given at Annexure II.
The Ministry of Health and Family Welfare has released the Rural Health Statistics (RHS) publication for the year 2020-21. It is a vital source of information on health infrastructure and human resources available in the public health institutions of all States/UTs.
As per RHS 2020-21, there are 1,57,819 Sub Centres (SC), 30,579 Primary Health Centres (PHCs) and 5,951 Community Health Centres (CHCs) respectively functioning in rural and urban areas of the country. Besides this, a total of 1,224 Sub Divisional/Sub District Hospital and 764 District Hospitals (DHs) are operational throughout the country.
The primary responsibility for ensuring the better health services and facilities lies with the State Governments/UT Administrations. Accordingly, the Ministry has conducted National, Regional and State level workshops for dissemination of the key findings of the NFHS-5. During these workshops the key findings of NFHS-5 have been shared with the State Governments/UT Administration, Central Ministries and other Stakeholders for policy/research purpose and for effective monitoring and implementation of the various Schemes/programmes.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
The major key findings evident from NFHS-5 (2019-21) compared to NFHS-4 (2015-16)
India has made striking progress in population control measures in recent times. The Total Fertility Rate (TFR), i.e. average number of children per women, has further declined from 2.2 to 2.0 at the National level between NFHS-4 & 5.
Overall Contraceptive Prevalence Rate (CPR) has increased substantially from 54% to 67% in the country. Use of modern methods of contraceptives has also increased in almost all States/UTs. Unmet needs of family Planning have witnessed a significant decline from13 percent to 9 percent. The unmet need for spacing, which remained a major issue in India in the past has come down to less than 4 percent.
The proportion of pregnant women who had an ANC visit in the first trimester has increased from 59 to 70 percent between NFHS-4 and NFHS-5. A considerable progress is noticed in percentage of mothers who had at least 4 ANC visits at the National level from 51 percent in 2015-16 to 58 percent in 2019-21.
Institutional Births have increased substantially from 79 percent to 89 percent in India. Even in rural areas around 87 percent births are delivered in health facilities and same is 94 percent in urban areas.
In NFHS-5, more than three-fourths (76.4%) children age 12-23 months were fully immunized, compared with 62 percent in NFHS-4.
List of SDG indicators, where NFHS-5 is the data source
1 Percentage of households with any usual member covered by a health scheme or health insurance
2 Percentage of children aged under 5 years who are underweight
3 Percentage of children under age 5 years who are stunted
4 Prevalence of malnutrition among children under 5 years of age, by type (wasting and overweight)
5 Prevalence of anaemia in women aged 15 to 49 years, by pregnancy status (percentage)
6 Percentage of women (15-49 years) whose Body Mass Index (BMI) is below normal
7 Percentage of Children age 6-59 months who are anaemic (<11.0g/dl)
8 Percentage of births attended by skilled health personnel (Period 5 years)
9 Percentage of births attended by skilled health personnel (Period 1 year)
10 Percentage of women aged 15-49 years with a live birth, for last birth, who received antenatal care, four times or more (Period 5 years/1 year)
11 Percentage of population (men (15-49 years) & women (15 - 49 years)) who drink alcohol about once a week out of total population (men (15-49 years) & women (15 - 49 years)) who drink alcohol
12 Percentage of population (15 years and above) who consume alcohol, by sex
13 Percentage of currently married women aged 15-49 years who have their need for family planning satisfied with modern methods
14 Percentage of Institutional Births (5 years)
15 Percentage of currently married women (15-49 years) who use any modern family planning methods
16 Percentage of women aged 15-19 years who were already mothers or pregnant
17 Percentage of currently married women (15-49 years) who use any modern family planning methods
18 Prevalence of hypertension among men and women age 15 years and above (in percentage)
19 Percentage of population in age group 15-49 who reported sought treatment out of total population in that age group having diabetes
20 Percentage of women aged 30- 49 years who have ever undergone a screening test for cervical cancer
21 Proportion of men and women reporting Asthma in the age group 15-49 years
22 Percentage of children age 12-23 months fully vaccinated with BCG, measles and three doses each of polio and DPT or Penta vaccine (excluding polio vaccine given at birth)
23 Proportion of ever-married women age 18-49 who have experienced physical, sexual or emotional violence by husband/partner in the past 12 months
24 Proportion of women and girls aged 18-49 years subjected to sexual violence by persons other than an intimate partner in the previous 12 months
25 Percentage of women aged 20-24 years who were married by exact age 18 years
26 Unmet need for family planning for currently married women aged 15-49 years (in percentage)
27 Percentage of population aged 15-24 years with comprehensive knowledge of HIV/ AIDS
Government of India has several initiatives to provide financial and medical support to all pregnant women including women belonging to economically weaker sections during their pregnancy and delivery in Government hospitals. These include:
Janani Suraksha Yojana (JSY), a demand promotion and conditional cash transfer scheme for promoting institutional delivery. The details of the scheme can be seen at the link given below: https://nhm.gov.in/index1.php?lang=1&level=3&lid=309&sublinkid=84
Janani Shishu Suraksha Karyakaram (JSSK) entitles every pregnant woman to free delivery, including for caesarean section, in public health institutions along with the provision of free transport, diagnostics, medicines, other consumables, diet and blood. The Government of India has taken various steps to ensure assured availability of all facilities to pregnant women near their homes, especially at the time of delivery in the remote areas of the country including rural areas of Uttarakhand and Bundelkhand region as given in the Annexure.
Government of India has not received any complaints for cases of negligence in pregnant women during their delivery in Government hospitals.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
Steps taken by Govt. of India to ensure assured availability of all facilities to pregnant women near their homes, especially at the time of delivery in the remote areas of the country are as follows:
Over 25,000 ‘Delivery Points’ across the country have been strengthened in terms of infrastructure, equipment, and trained manpower for provision of comprehensive RMNCAH+A services Setting up of Maternal and Child Health (MCH) Wings at high caseload facilities to improve the quality of care provided to mothers and children.
Functionalization of First Referral Units (FRUs) by ensuring manpower, blood storage units, referral linkages etc.
Operationalisation of 102 services for pregnant women for providing free transport from home to facility, inter facility transfer in case of referral and drop back to home.
Birth Waiting Homes (BWH) have been established in remote and tribal areas to promote institutional delivery and improve access to healthcare facilities
Surakshit Matratva Ashwasan (SUMAN) for providing assured, dignified, respectful and quality healthcare at no cost and zero tolerance for denial of services for every woman and newborn visiting the public health facility to end all preventable maternal and newborn deaths.
Pradhan Mantri Surakshit Matritva Abhiyan (PMSMA) provides pregnant women a fixed day, free of cost assured and quality Antenatal Care on the 9th day of every month.
Obstetric High Dependency Unit (HDU) & Intensive care unit (ICU)- Setting up of Obstetric HDU/ICU in the high case load tertiary care facilities across country to handle complicated pregnancies.
LaQshya (Labour room Quality improvement Initiative) to improve the quality of care in Labour room and Maternity operation theatres to ensure that pregnant women receive respectful and quality care during delivery and immediate post-partum.
Monthly Village Health, Sanitation and Nutrition Day (VHSND) is an outreach activity for provision of maternal and child care including nutrition.
Information, education and communication (IEC) to promote institutional deliveries.
Birth Microplanning and Birth Preparedness by Skilled birth attendance (SBA) trained ANMs.
MCP Card and Safe Motherhood Booklet are distributed to the pregnant women for educating them on diet, rest, danger signs of pregnancy, benefit schemes and institutional deliveries.
Outreach camps are provisioned for improving the reach of health care services especially in tribal and hard to reach areas. This platform is used to increase the awareness for the Maternal & Child health services, community mobilization as well as to track high risk pregnancies.
National Health Authority (NHA) under the Ministry of Health & Family Welfare is mandated with the implementation of Ayushman Bharat Pradhan Mantri – Jan ArogyaYojana (ABPM-JAY). The Scheme provides an annual health cover of Rs. 5 Lakhs per eligible beneficiary family for secondary and tertiary care hospitalizations corresponding to 1,949 treatment procedures across 27 specialties. The treatment packages under Ayushman Bharat PM-JAY cover for the cost of implants to certain procedures.
National Pharmaceutical Pricing Authority (NPPA) under the Department of Pharmaceuticals (DoP) had on 13th February 2017 fixed the ceiling price of Bare Metal Stents and Drug Eluting Stents as per provisions of the Drugs (Prices Control) Order, 2013 at ₹7,260 for Bare Metal Stents and ₹29,600 for Drug Eluting Stents (including BVS/ Biodegradable). The current ceiling price, based on revision of WPI, for the Coronary Stents with effect from 30th March 2022 is as under:

The hospitals cannot charge the patients more than the ceiling price fixed by NPPA. Further, to facilitate availability and ensure affordability, NPPA has capped the trade margin for medical equipment, viz., Pulse Oximeter, Glucometer, BP Monitor, Nebulizer and Digital Thermometer vide notification dated 13th July, 2021.
As informed by the Ministry of Health & Family Welfare, as on 21st July 2022, more than 3.63 Crore hospital admissions amounting to over Rs. 42,880 Crores have been authorized under Ayushman Bharat PM-JAY. The authorized hospital admissions for the previous three financial years from FY 2019/20 to FY 2021/22 are as follows:
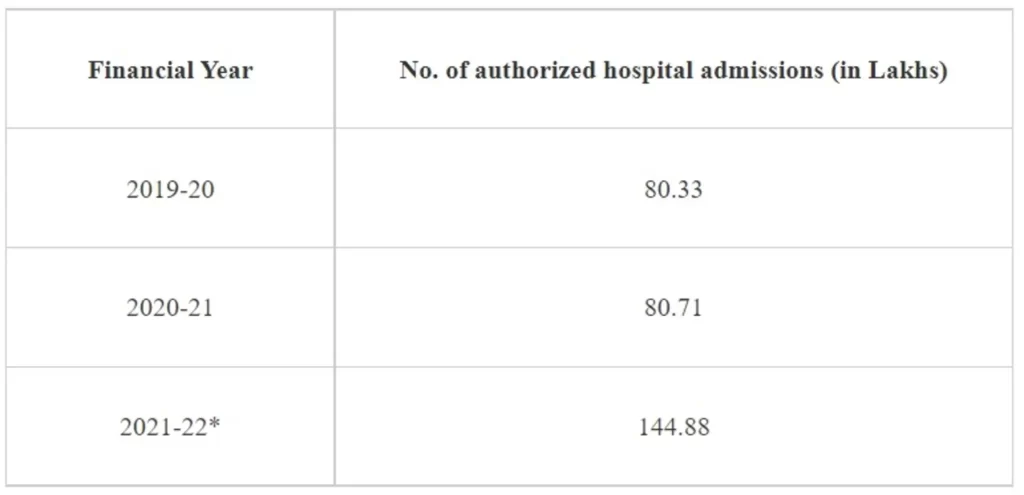
*Note: – Hospital admissions data for Rajasthan and Telangana for FY 2021-22 is sourced directly from the respective State Health Agencies.
Further, the authorized hospital admissions for the previous three financial years from FY 2019/20 to FY 2021/22, involving the use of cardiac stents or implants, is as follows:
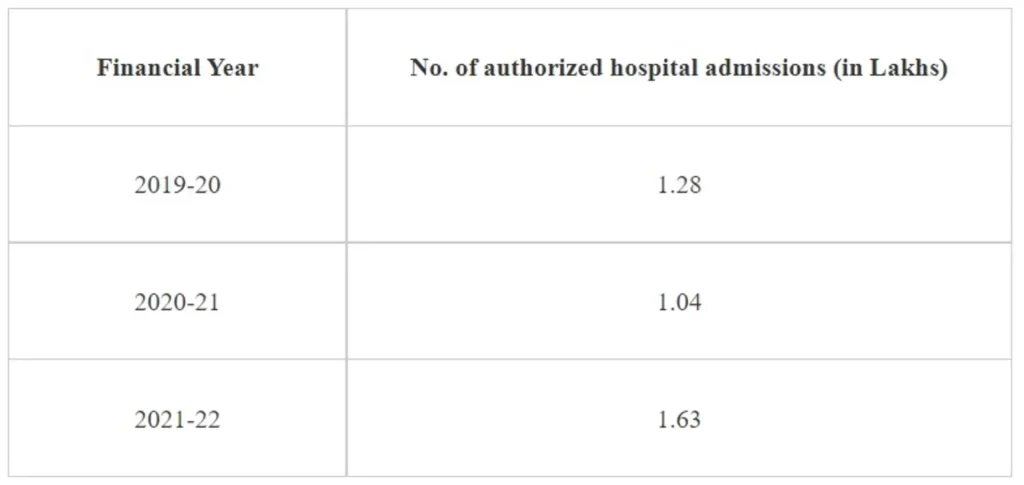
As per available records, no such suggestions/ complaints have been received by the Department of Pharmaceuticals, Ministry of Chemicals & Fertilizers.
This information was given by Minister of State for Chemicals and Fertilizers, Shri Bhagwanth Khuba in a written reply in Lok Sabha today.
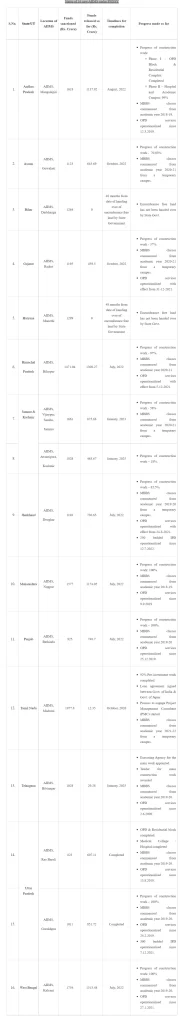
Setting up of 22 new AIIMS has been approved under Pradhan Mantri Swasthya Suraksha Yojana (PMSSY), 6 of which at Bhopal (Madhya Pradesh), Bhubaneswar (Odisha), Jodhpur (Rajasthan), Patna (Bihar), Raipur (Chhattisgarh) and Rishikesh (Uttarakhand) are fully functional. State/UT-wise details of remaining 16 AIIMS, including details of funds sanctioned, funds released, the present status and timelines for completion is at Annexure.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
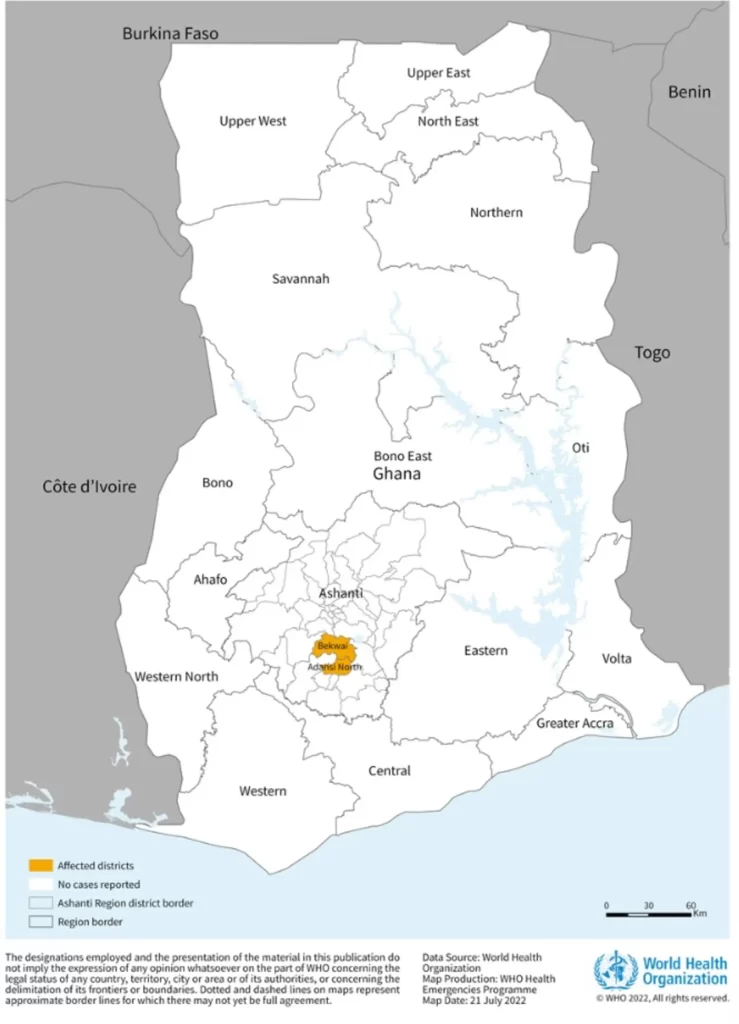
Two fatal cases of Marburg virus disease (MVD) were reported from Ashanti region, Ghana. On 28 June 2022, these cases were notified to health authorities as suspected viral hemorrhagic fever (VHF) cases and tested positive for Marburg virus on 1 July 2022. An outbreak of MVD has only been reported once previously in West Africa, and this is the first time MVD has been notified in Ghana. An outbreak of MVD may represent a serious public health threat as it is severe and often fatal.
On 28 June 2022, two suspected viral hemorrhagic fever (VHF) cases were notified to health authorities in the Ashanti region, Ghana (Figure 1). This region is located in the middle belt of Ghana and is the most populated region in the country.
The first case was a 26-year-old male who was a farm worker in Adansi North district, Ashanti region with a travel history to the Western region. Prior to symptom onset on 24 June, he arrived from the Western region to Ashanti region. He sought care at a hospital on 26 June and died on 27 June. The case was transported and buried in the Sawla-Tuna-Kalba district, Savannah region, which borders both Burkina Faso and Côte d’Ivoire. The burial was conducted before the results of laboratory tests for MVD were available.
The second case was a 51-year-old male who was a farm worker in Bekwai Municipal, Ashanti region. He sought care at the same hospital as the first case on 28 June but died the same day.
Both cases presented with fever, general malaise, bleeding from the nose and mouth, and subconjunctival bleeding (bleeding of the blood vessels in the eyes). Blood samples were collected on 27 June for the first case and 28 June for the second case and sent to Noguchi Memorial Institute of Medicine Research (NMIMR) for testing. On 1 July, both cases tested positive for Marburg virus by reverse transcriptase polymerase chain reaction (RT-PCR). On 12 July, samples collected from the two cases were sent to Institute Pasteur in Dakar, Senegal (IPD) which confirmed the results from NMIMR on 14 July 2022.
Figure 1. Region of the two confirmed cases of Marburg virus disease reported in Ghana, as of 20 July 2022
The source of infection is unknown. Preliminary investigations have shown that neither of these cases had a history of contact with dead animals, sick persons, or animals and had not attended any social gathering within three weeks of symptoms onset. Although both cases were farmers, they worked in different locations, and they have not been found to be epidemiologically linked. However, both cases came from communities living in a forest environment.
A cumulative number of 108 individuals (50 from Ashanti region, 48 from Savannah region, and 10 from the Western region) were identified as contacts of the two cases, all of whom were under self-quarantine and daily monitoring for 21 days. On 20 July, all contacts completed their follow up period. These contacts included health care workers and immediate family members of the cases. One contact reported some symptoms, but a blood sample that was collected tested negative at NMIMR on 7 July. All the other contacts reported to be in good health during the follow up period.
MVD is an epidemic-prone disease associated with high case fatality ratios (CFR; 24-88%). In the early course of the disease, the clinical diagnosis of MVD is difficult to distinguish from many other tropical febrile illnesses due to the similarities in the clinical symptoms. Other VHFs need to be excluded, particularly Ebola virus disease, as well as malaria, typhoid fever, leptospirosis, rickettsial infections, and plague. Human MVD infection can result from prolonged exposure to mines or caves inhabited by Rousettus bat colonies. Marburg virus spreads through human-to-human transmission via direct contact (through broken skin or mucous membranes) with the blood, secretions, organs or other bodily fluids of infected people, and with surfaces and materials (e.g., bedding, clothing) contaminated with these fluids.
Although there are no vaccines or antiviral treatments approved to treat the virus, supportive care – rehydration with oral or intravenous fluids – and treatment of specific symptoms, improve survival. A range of potential treatments, including blood products, immune therapies, and drug therapies, are being evaluated.
National and international coordination involving key partners are ongoing to respond to this outbreak.
The Ministry of Health of Ghana is coordinating response activities and is engaging with partners for support, as needed.
The Ashanti region health directorate established coordination mechanisms in the affected health districts and response activities have commenced.
Epidemiological investigations continue to be conducted, including enhanced surveillance using the Integrated disease surveillance and Response (IDSR) system and follow-up of contacts. Eight alerts have been reported from Ashanti, Oti and Western regions. Following investigations, seven alerts were discarded and one alert from Oti region tested positive for yellow fever.
Sensitization activities of health care workers on case definition and infection prevention and control measures are ongoing.
The NMIMR laboratory has the capacity to test for viral hemorrhagic fevers and is testing samples resulting from alerts. To date, 15 samples were tested for Marburg virus, and all samples tested negative. Sequencing of the virus is ongoing.
A designated hospital has been identified in Ashanti region to isolate and care for additional cases.
WHO is deploying technical experts to support the country in strengthening Infection Prevention and Control (IPC), coordination, surveillance, and conducting investigations assessing risks for further outbreaks.
An orientation has been held for community-based surveillance volunteers to enhance surveillance at the community level.
WHO is offering testing support in the form of supplying reagents to NMIMR which first tested the samples.
Côte d’Ivoire and Burkina Faso have been informed of the event and are initiating preparedness activities.
The risk of this outbreak is high at the national level, moderate at the regional level, and low at the global level. The notification of two confirmed cases of MVD with a CFR of 100% (2/2) raises concern. The epidemiological investigation has not yet identified the source of this outbreak, which highlights the need to intensify the community-based surveillance.
There is a risk for this outbreak to spread to neighboring countries because the first case travelled from Western region prior to symptoms onset. The Western region shares a border with Côte d’Ivoire, and this may present a risk of transborder transmission if more cases continue to be reported or affect other areas. In addition, the first case was not buried safely in Savannah region which also borders Côte d’Ivoire and Burkina Faso. Therefore, WHO has assessed the risk of this outbreak as high at the national level, moderate at the regional level, and low at the global level.
Community engagement activities need to be carried out in the three identified regions in Ghana (Ashanti, Savannah and Western regions) with a particular focus on providing adequate information and support for the population to implement measures to prevent further transmission, avoid stigmatization within the community, and encourage early presentation to treatment centers and other necessary outbreak containment measures, including safe burials.
It is also important to raise awareness of the risk factors for MVD infection and the protective measures that individuals can take to reduce human exposure to the virus. Key public health communication messages include:
Reducing the risk of human-to-human transmission in the community arising from direct or close contact with infected patients, particularly with their body fluids. Close physical contact with Marburg patients should be avoided. Gloves and appropriate personal protective equipment should be worn when taking care of ill patients at home. Regular hand washing should be performed after visiting sick relatives in hospital, as well as after taking care of ill patients at home.
Reducing the risk of bat-to-human transmission. Handle wildlife with gloves and other appropriate protective clothing. Cook animal products (blood and meat) thoroughly before consumption and avoid consuming raw meat. During work, research activities, or tourist visits in mines or caves inhabited by fruit bat colonies, wear gloves and other appropriate protective clothing including masks.
Outbreak containment measures should be continued and strengthened in the three regions of Ghana. These measures include:
Maintaining and building trust with communities
Scaling-up surveillance activities (active case search, contact monitoring, and investigation)
Testing all suspected cases of MVD in a timely manner
Ensuring that adequate infection prevention and control measures are implemented when caring for suspected, probable and confirmed patients
Isolating early, and caring for suspected and confirmed cases, if possible, in designated health facilities with trained health workers and adequate IPC measures.
Performing safe and dignified burial of deceased individuals who are probable or confirmed cases of MVD.
Marburg virus transmission has previously been reported in health care settings when appropriate infection prevention and control (IPC) measures were not implemented. IPC measures to reduce risk of transmission in health care include:
Establish an IPC task force in the health zone to ensure implementation of the Infection prevention and control activities
Ensure health workers are briefed on MVD (standard and transmission-based precautions) including an emphasis on safe injection practices
Ensure minimum WASH and isolation requirements and capacity are met in health facilities.
Based on the current risk assessment WHO advises against any restriction of travel and trade to Ghana.
The World Health Organization provides global leadership in public health within the United Nations system. Founded in 1948, WHO works with 194 Member States, across six regions and from more than 150 offices, to promote health, keep the world safe and serve the vulnerable. Our goal for 2019-2023 is to ensure that a billion more people have universal health coverage, to protect a billion more people from health emergencies, and provide a further billion people with better health and wellbeing.
