National Health Mission (NHM) envisages achievement of universal access to equitable, affordable & quality health care services that are accountable and responsive to people’s needs. NHM encompasses its two Sub-Missions, the National Rural Health Mission (NRHM) and the National Urban Health Mission (NUHM). The main programmatic components include Health System Strengthening in rural and urban areas, Reproductive-Maternal- Neonatal-Child and Adolescent Health (RMNCH+A) and Communicable and Non-Communicable Diseases.
Public Health & Hospital is a State subject, hence, the responsibility of strengthening public healthcare system, especially to rural & vulnerable population, including strengthening of existing health/ medical facilities lies with the respective State/ UT Governments. Under National Health Mission (NHM), Ministry of Health & Family Welfare, Government of India provides financial and technical support to States/UTs including support for health/ medical facilities, upto District Hospital level, based on the proposals submitted by States/UTs in their Program Implementation Plans (PIPs). Government of India provides approval for the proposals in the form of Record of Proceedings (RoPs) as per available resources.
Government of India extends supports to states to expedite the process of creating health infrastructure as per Indian Public Health Standards. These standards include norms for services, infrastructure, human resource, diagnostics, equipment, medicines etc.
Under Ayushman Bharat, the existing Sub-health Centres (SHCs) and Primary Health Centres (PHCs) are transformed into AB-HWCs to deliver twelve packages of Comprehensive Primary Health Care (CPHC) that includes preventive, promotive, curative, palliative and rehabilitative services which is universal, free and close to the community. Another essential component of Ayushman Bharat Health & Wellness Centres (AB-HWCs) is the roll out of tele-consultation services through ‘eSanjeevani’. This platform provides telemedicine services for the communities through a hub and spoke model which connects AB-HWCs (spokes) to District Hospitals/Medical colleges (Hubs) for specialist consultation services. Teleconsultation services are aimed to improve access of specialist services to the communities, especially in the rural areas.
Further, following steps have been taken for strengthening healthcare systems to cater to future healthcare needs:
“Emergency Response and Health Systems Preparedness Package – II (ECRP-II)” for Rs. 23,123 Crore, including Central Component, was approved by Cabinet to extend support to the States/UTs for establishing District Paediatric Units (including Oxygen Supported beds and ICU beds) in all the Districts of the Country. Besides, support is also provided to increase the availability of ICU beds in Government health facilities. Maintaining buffer stock of essential medicines required for effective COVID management, in addition to provision for required drugs and diagnostics, is also supported. Support is also available for establishing field hospitals (100 bedded or 50 bedded units), wherever required.
PM Ayushman Bharat Health Infrastructure Mission (PM-ABHIM) with an outlay of Rs. 64,180 crores till 2025-26 envisages increased investments in public health and other health reforms to provide better access to health in rural areas by i) Strengthening of Health and Wellness Centres in villages and cities for early detection of diseases; ii) Addition of new critical care-related beds at district level hospitals; iii) Support for Block Public Health Units (BPHU) in 11 high focus States; and iv) Integrated district public health laboratories in all districts.
The recommendations of 15th Finance Commission (FC-XV) w.r.t Health Grants through Local Governments announced in Union Budget 2021-22, addresses the gaps in Primary Healthcare facilities.
The Pradhan Mantri Swasthya Suraksha Yojana (PMSSY) aims at correcting regional imbalances in the availability of affordable tertiary healthcare services and to augment facilities for quality medical education in the country. The Scheme has two components, namely, (i) Setting up of All India Institute of Medical Sciences (AIIMS); and (ii) Upgradation of existing Government Medical Colleges/ Institutions (GMCIs). So far setting up of 22 new AIIMS and 75 projects of upgradation of GMCIs have been approved under the Scheme in various phases. The setting up of AIIMS contains Medical College, Nursing College, Hospital, Trauma Centre, Emergency, Blood Bank, ICU, Diagnostic & Pathology, Research etc. Upgradation of GMCIs under PMSSY broadly involves construction of Super Speciality Block (SSB) and/ or Trauma Centre/ or other facilities and/ or procurement of medical equipment. Under the Centrally Sponsored Scheme (CSS) , ‘Establishment of new medical colleges attached with existing district/referral hospitals’, establishment of 157 medical colleges has been sanctioned.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
With an objective to further enhance India’s existing manufacturing prowess in the Pharmaceuticals Industry, Department of Pharmaceuticals, Ministry of Chemicals & Fertilizers, Govt of India is planning to roll out a series of initiatives under the banner of Schemes for ‘Strengthening Pharmaceuticals Industry’ (SPI). Keeping in view the strategic role of MSMEs, who provide important forward and backward linkages to the industry, and also keeping in view that MSMEs tend to grow in clusters, these schemes shall address the issues of technology upgradation at both unit level and cluster level.
In order to strengthen the supply chain of Pharmaceuticals industry where MSMEs are an integral part, Govt of India shall be incentivizing the intending MSME units going in for acquiring Schedule M certification or WHO GMP Certification through the sub-scheme Pharmaceuticals Industry Technology Upgradation Assistance Scheme (PTUAS). The MSME unit shall have the option to choose from either Capital subsidy or Interest subvention. At the cluster level, the sub-scheme ‘Assistance to Pharmaceuticals Industry for Common Facilities’ (APICF) envisages to support creation of common facilities like Testing Labs, Common Effluent Treatment plants and other such common facilities by providing Govt support in the form of capital grant to the extent of up to 70 per cent subject to a ceiling of a maximum of Rs. 20 crore. In order to plug the knowledge gap, the third sub-scheme proposes to conduct a series of awareness programs, conduct sectoral studies and organize similar such programs to generate soft inputs for policy advocacy.
These initiatives are formally getting unveiled by Union Minister of Chemicals & Fertilizers and Health & Family Welfare Dr. Mansukh Mandaviya, on July 21, 2022 at Dr. Bhimrao Ambedkar International Convention Centre. MoS for Chemicals & Fertilizers Shri Bhagwanth Khuba shall also be gracing the occasion. Senior officials from Department of Chemicals & Fertilizers, Ministry of MSME, SIDBI, NSIC, Banks besides representatives of Industry, entrepreneurs, Start Ups etc.
Government of India have been working towards boosting cancer research and treatment in the country. In this regard, National Cancer Institute at Jhajjar (Haryana) and Chittaranjan National Cancer Institute, Kolkata (First and Second Campus) have been set up by Ministry of Health & Family Welfare.
The Department of Health & Family Welfare, Government of India, provides technical and financial support to the States/UTs under the National Programme for Prevention and Control of Cancer, Diabetes, Cardiovascular Diseases and Stroke (NPCDCS), as part of National Health Mission (NHM), based on the proposals received from the States/UTs and subject to the resource envelope. Cancer is an integral part of NPCDCS. The programme focusses on strengthening infrastructure, human resource development, health promotion & awareness generation for Cancer prevention, early diagnosis, management and referral to an appropriate level of healthcare facility for treatment of the Non-Communicable Diseases (NCDs), including Cancer. Under NPCDCS, 685 District NCD Clinics, 266 District Day Care Centres, and 5451 Community Health Center NCD Clinics has been set up.
The Central Government has approved Tertiary Care Cancer Facilities Scheme in order to enhance the facilities for tertiary care of cancer. 19 State Cancer Institutes (SCIs) and 20 Tertiary Care Cancer Centres (TCCCs) have been approved under the said scheme.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The Government has taken various regulatory measures since 2019 to ensure the quality of medicines in the country. The key measures are as under:
The Drugs Rules, 1945 were amended providing that every Active Pharmaceutical Ingredient (bulk drug) manufactured or imported in India shall bear Quick Response Code on its label. The stored data or information shall include the minimum particulars including Unique product identification code, Batch No, Manufacturing date, Expiry Date.
The Drugs Rules, 1945 were amended providing that any marketer who sells or distributes any drug shall be responsible for quality of that drug as well as other regulatory compliances along with the manufacturer under these rules.
The Drugs Rules, 1945 were amended providing that “in case the applicant intends to market the drug under a brand name or trade name, the applicant shall furnish an undertaking in Form 51 to the Licensing Authority to the effect that to the best of his knowledge based on search in trademarks registry, central data base for brand name or trade name of drugs maintained by Central Drugs Standard Control Organisation, literature and reference books on details of drug formulations in India, and internet, such or similar brand name or trade name is not already in existence with respect to any drug in the country and the proposed brand name or trade name shall not lead to any confusion or deception in the market”.
The manufacture for sale, sale and distribution of 80 Fixed Dose Combinations (FDCs) drugs in India were prohibited in public interest after consultation with the Drugs Technical Advisory Board.
220 additional posts of various levels have been created including Joint Drugs Controller, Deputy Drugs Controller, Asst. Drugs Controller and Drugs Inspector in the year 2022-23.
Further, Department of Pharmaceuticals has launched a scheme, namely, Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP). Under the scheme, Generic Medicines are sold through dedicated outlets namely Pradhan Mantri Bhartiya Janaushadhi Kendras (PMBJKs). It is an initiative of Government of India towards making an impact on common masses to provide quality medicines at affordable prices. As on 30.06.2022, 8,742 Janaushadhi Kendras have been opened across the country and the product basket of PMBJP comprises 1,616 drugs and 250 surgical items.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The Transplantation of Human Organs & Tissues Act (THOTA), 1994 provides for regulation of removal, storage and transplantation of human organs & tissues for therapeutic purposes and for prevention of commercial dealings in human organs & tissues. The subject of artificial organs is not regulated under the aforesaid Act.
As reported by Directorate General of Health Services, there has been an overall increase in lifestyle diseases like Diabetes, Hypertension, Chronic Kidney Disease, Non-Alcoholic Fatty Liver disease in the past decade, which may cause organ damage. Once the end stage organ failure occurs, such cases may require organ transplants.
The Government of India has implemented National Organ Transplant Program (NOTP) to establish a network of organ and tissue transplant organizations at national, regional and state level and to link them with transplant & retrieval hospitals and tissue banks and to maintain a national registry of organ & tissue donors and recipients for the purpose to provide an efficient system for procurement and distribution of organs & tissues from deceased donors. Under the program, support is provided to establish new or to upgrade existing organ & tissue transplant and retrieval facilities and to establish tissue banks.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The Government has implemented the National TB Elimination Programme (NTEP) under the aegis of the National Health Mission (NHM). With the goal of achieving Sustainable Development Goals related to TB by 2025, five years ahead of the global targets, the programme has implemented a National Strategic Plan with the following objectives: -
Early diagnosis of TB patients, prompt treatment with quality assured drugs and treatment regimens.
To engage with the patients seeking care in the private sector.
Prevention strategies including active case finding and contact tracing in high risk /vulnerable population.
Airborne infection control.
Multi-sectoral response for addressing social determinants.
The Government has made steady progress towards achieving the SDG targets. As per the Global TB Report, 2021 by the World Health Organisation, the incidence of Tuberculosis in India has reduced from 217 / lakh population in 2015 to 188 / lakh population in 2020.
The other steps taken by the Government to ensure TB patients are diagnosed and complete treatment, are as under:
State and District Specific Strategic plan for targeted interventions in high burden areas.
Provision of free drugs and diagnostics to TB patients including for drug resistant TB.
Active TB case finding campaign in key vulnerable and co-morbid population.
Integration with Ayushman Bharat- Health & Wellness Centres to decentralize screening and treatment services closer to the community.
Private sector engagement including incentives for notification and management of TB cases.
Scale up of molecular diagnostic laboratories to sub-district levels.
Nikshay Poshan Yojana for nutritional support to TB patients.
Intensified IEC campaigns to reduce stigma, raise community awareness and improve health seeking behaviour.
Multi-sectoral response with involvement of line ministries.
Scale up TB preventive therapy to contacts of pulmonary TB.
Notified TB cases are tracked through web-based portal Ni-kshay.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The National Health Authority (NHA) is organising its first hackathon under the Ayushman Bharat Digital Mission Hackathon Series from 14th to 17th July, 2022 at the Smart City Operations Centre, Pune, Maharashtra in hybrid format. The hackathon is being organized under the Ayushman Bharat Digital Mission (ABDM) in association with Pune Municipal Corporation (PMC) and Pune Smart City Development Corporation Limited (PSCDCL). Various teams comprising of innovators, developers and data experts have joined physically and virtually to collaborate and build innovative solutions.
The hackathon was inaugurated by Dr. R.S. Sharma, Chief Executive Officer, NHA. Shri. Saurabh Vijay, Secretary, Medical Education and Drugs Department, Government of Maharashtra, Dr. Praveen Gedam, Additional CEO, NHA, Shri. Vikram Kumar, Commissioner, PMC, Dr. N. Ramaswamy, Commissioner (Health Services) and Mission Director, Shri Kiran Gopal Vaska, Director, NHA, Dr. Sanjay Kolte, CEO, PSCDCL, Dr. Gaur Sunder, Associate Director, C-DAC Pune, Shri. Ravindra Binwade, Addl. Commissioner, PMC, and Dr. Kunal Khemnar, Addl. Commissioner PMC were present during the inaugural event.
Speaking at the inauguration, Dr. R.S. Sharma, CEO, NHA said – “Similar to the role played by UPI, the Unified Health Interface will act as a market enabler and will provide multiple health services. It is a unique attempt to build interoperability between healthcare providers and facilitate exchange of healthcare data - which can ultimately make quality healthcare accessible & affordable to all.”
Adding further on the hackathon, Dr. Sharma said – “This hackathon is the first in the series of hackathons we are planning to hold. This will encourage young talent in this country to contribute to the enhancement of the building blocks of our digital healthcare ecosystem and create innovative solutions for the country and the world.”
In addition to the Unified Health Interface (UHI), the hackathon is also focussed at mobilising the health start-up ecosystem in India by bringing together individuals and organisations to develop innovative solutions. The tentative prize pool for ‘Round 1 – Kickstarting UHI’ is Rs. 60,00,000. The solutions shall be assessed by an independent jury. The prize shall be awarded to the top performers on each challenge track on two major themes:
Innovation Track: Challenge for innovative solutions to power digital health in an open network around various use cases such as teleconsultation, ambulance booking, lab tests, physical consultation booking, lab tests booking. Integration Track: Challenge to accelerate development of applications that are compatible with UHI and integrate these applications with the applications of other such participants to enable digital health transactions on the UHI network.
More details on the ABDM Hackathon are available at: https://abdm.gov.in/register
More information on Unified Health Interface (UHI) is available at: https://uhi.abdm.gov.in/
“COVID is not over yet. There are reports of rising cases of COVID in some states. It is important at this time to be alert and not forget COVID Appropriate behaviour (CAB) such as wearing masks and maintaining physical distance to prevent spread of the infection”. This was stressed by Union Minister for Health and Family Welfare, Dr Mansukh Mandaviya as he chaired a meeting through Video Conference (VC) today with Health Ministers and senior officials of States and Union Territories (UTs) to review the progress of vaccination exercise HarGharDastak 2.0 campaign.
Highlighting increased case positivity in some districts and States and reduced COVID-19 testing, Dr Mandaviya stated that increased and timely testing will enable early identification of COVID cases and help to curb spread of the infection among the community. He urged States/UTs to continue and strengthen the surveillance and focus on genome sequencing for identifying new mutants/variants in the country. He stated that the five-fold strategy of Test, Track, Treat, Vaccination & Adherence to Covid Appropriate Behavior (CAB) needs to be continued and monitored by States/UTs. States were also urged to focus on implementing the Operational Guidelines for Revised Surveillance Strategy for COVID-19 which focuses on surveillance of incoming international travellers and surveillance through health facilities, labs, community etc.
Stressing on the importance of COVID vaccination among the vulnerable age groups, he urged the State Health Ministers to personally review the status and progress of the special month-long drive Har Ghar Dastak 2.0 campaign which has started since 1st June. He said “Let us accelerate our efforts to identify all beneficiaries in the 12-17 age group for the 1st and 2nd doses, so they can attend schools with the protection of the vaccine”. He urged States for focussed coverage of 12-17 age groups through school-based campaigns (Govt/Private/ informal Schools like Madrasas, Day Care Schools), along with targeted coverage of non-school going children during the summer vacation.

He added that the population group of above 60 years of age is a vulnerable category and needs to be protected with the Precaution Dose. “Our healthcare workers are going door-to-door to ensure that the vulnerable population is administered the Precaution Dose”, he said. The States Health Ministers were urged to also review administration of precaution dose to 18-59 years age-group with the private hospitals on a regular basis. He said that we can utilize the learning from the first Har Ghar Dastak campaign to achieve 100% coverage among the eligible population for ensuring extended protection against COVID. “Adequate vaccine doses are available across the country. Let us ensure accelerated coverage of COVID vaccination during the second phase of the Har Ghar Dastak campaign”, he stressed.
They were also strongly advised to ensure that there is no wastage of COVID-19 vaccines at any cost. This must be ensured through active monitoring and on the basis of “First Expiry First Out '' principle, where doses that expire earlier should be used for vaccination first, the Union Health Minister stated.
Dr Bharati Pravin Pawar, MoS(HFW) also stressed on accelerated COVID vaccination coverage in the States through Har Ghar Dastak2.0 campaign.
State Health Ministers Dr Sapam Ranjan Singh (Manipur), Shri Alo Libang (Arunachal Pradesh), Shri Thanneeru Harish Rao (Telangana), Shri Anil Vij (Haryana), Shri Rushikesh Ganeshbhai Patel (Gujarat), Shri Banna Gupta (Jharkhand), Shri Mangal Pandey (Bihar), Dr. Rajesh Tope (Maharashtra), Dr. Prabhuram Choudhary (Madhya Pradesh), Dr K Sudhakar (Karnataka) were present in the meeting.
Dr. Manohar Agnani, Addl. Secy, Ms Roli Singh, Addl. Secy, Shri Lav Agrawal, Jt. Secy, and other senior officials of the Union Health Ministry were present in the virtual review meeting along with NHM Mission Directors and other officials from States/UTs.
“Let us move from ‘volume’ to ‘value’ leadership to capture the global pharmaceutical market. It is time to accumulate knowledge from global best practices in research, manufacturing and innovation and develop our own models focused on accelerating production to meet the domestic demand while increasing our global footprint”. Union Minister for Chemicals & Fertilizers and Health & Family Welfare, Dr. Mansukh Mandaviya exhorted the champions and leaders of Pharmaceutical Industry at the interactive session with Indian Pharmaceutical Alliance. The objective of the meeting was to discuss India’s Pharma Vision 2047 and roadmap for the Indian pharmaceutical sector. The session discussed the current position of the pharma industry in India, key initiatives taken by the Government in the last few years and collaborative steps that will help India to realize this vision.

Reiterating the prime minister’s vision of attaining Aatmanirbharta in all fields, Dr. Mansukh Mandaviya pushed for making the pharmaceutical sector leapfrog in its growth trajectory in the upcoming years. He said, "We already have the required ‘manpower and brand power,’ and Indian companies today are at an inflection point to capture top global positions.” India has been acknowledged as the “pharmacy of the world” based on its generic medicine production and the volume share in the global market. It is time to move ahead and capture the top global positions based on the value too, he stressed.
While reiterating India’s philosophy of “Vasudaiva Kutumbakam” he said that “we have always believed in supporting the world while also balancing our domestic demands. It is during the pandemic crisis when world looked up to India, we delivered. This has led to global appreciation of India’s strengths and we must now use this opportunity to take it to the next level of “Discover and Make in India”.
Dr. Mandaviya underscored the importance of long-term policies which provide stability to the industry. Emphasising that the government is committed to supporting the Pharma Companies with Industry-Friendly policies and investor promoting ecosystem, he said “This Government believes in taking a holistic approach. Our policies are based on extensive and comprehensive stakeholder consultation which provide the basis for comprehensive, long -term and vibrant policy ecosystem”.
Simultaneously, he added that “we need to move ahead with the Pharma Industry being at the forefront in taking lead and proposing their own models and initiatives in expanding manufacturing capacity, invest in innovative technologies and focus on research and development. The government will strengthen the sector through streamlining policies and effective schemes such as the PLI which support cutting edge research”. Apart from policy front, Dr. Mandaviya also focused on the investments in research and development, efficient manufacturing capacities along with creating ample opportunities for innovation that is the need of the hour now. He was hopeful that through these steps we will be able to create a vibrant ecosystem for this sector.
Ms S Aparna, Secretary, Department of Pharmaceuticals, said that the Government of India continues to work towards realizing our goals for the pharmaceutical sector, from various initiatives aligning with the industry to sustaining focus on access, innovation, quality and affordability to help achieve India Pharma Vision 2047.” She further added that they are addressing the issue of policy stability, facilitating reforms & regulatory ease to expand manufacturing and ensure quality assessments. Government is also encouraging investment in Research and Development(R&D),exports and digitalization, push to incubation of new startups and entrepreneurship initiatives. She also added that review processes are being expedited and accountability is being ensured through creation of proper channels. Focusing of these factors will help us achieve growth in pharmaceutical sector and will further build confidence in the sector.
The Indian Pharmaceutical Alliance discussed and highlighted various issues such as policy stability, innovation, regulatory framework reforms and ease including transparency and accountability, review process at CDSCO, regular monitoring of implementation of schemes, pricing and associated control, investment in infrastructure aimed to give impetus to research in innovation, expansion of manufacturing capacity of chemical and API for boosting exports, digitalization to harness its potential in expanding knowledge sharing, use in medical education and increasing access to quality healthcare, and how to enrich “Brand India” through investment in start-ups and collaboration with academia for tapping best manpower.
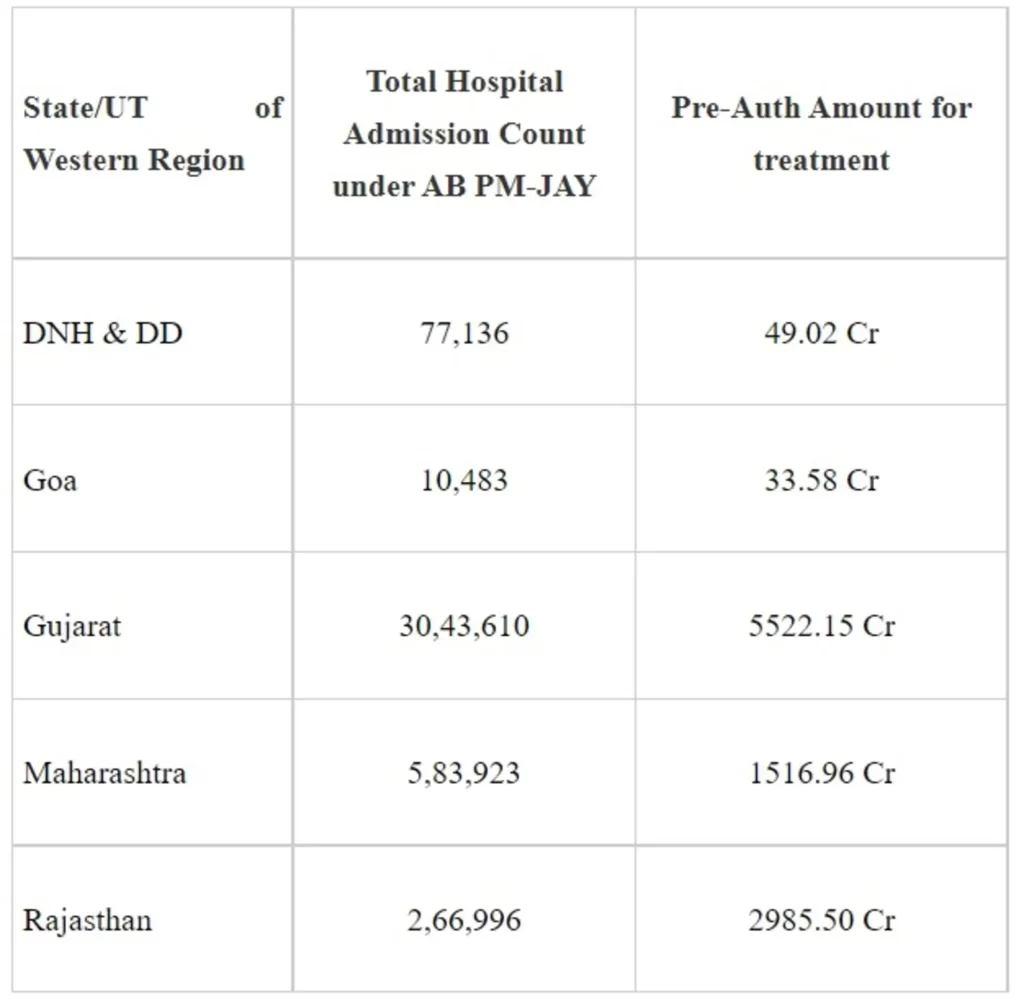
The fifth edition of Regional Review Meeting – ‘Ayushman Sangam’ was organised by National Health Authority (NHA) in Pune, Maharashtra for the Western region on 30th June and 1st July 2022. The event saw a gathering of senior government officials, policymakers and experts associated with the two schemes of Ayushman Bharat Pradhan Mantri Jan Arogya Yojana (AB PM-JAY) and Ayushman Bharat Digital Mission (ABDM) from the states/UT of Gujarat, Goa, Rajasthan, Maharashtra, Dadra and Nagar Haveli & Daman and Diu. The review meeting was chaired by Dr. R. S. Sharma, CEO, NHA. Addressing the event, Dr Sharma lauded the efforts and contribution made by the Western Zone towards the successful implementation of AB PM-JAY.
The two-day interaction created avenues of cohesive thought exchange facilitating assessment of progress made by both Ayushman Bharat PM-JAY & ABDM schemes in the States/UTs of Western Region.
Along the side-lines of the programme, around 1000 Doctors were felicitated on 1st July, 2022 for their exemplary contribution provided under PM-JAY scheme.
Dr. R. S. Sharma, CEO NHA while giving the keynote address highlighted upon the role played by the doctors in India’s fight against the pandemic and the roll out of the vaccination drive. Dr. Sharma said, ‘On the face of it, the COWIN platform is the tech-backbone of India’s vaccination drive but in spirit it is the network of our doctors and medical professionals who have made the world’s largest vaccination drive successful. Our community of doctors have been instrumental not only in our fight against the pandemic but have also played a crucial part in ensuring the successful implementation of the key healthcare schemes like the Ayushman Bharat umbrella scheme PM-JAY.’
While addressing the gathering, Dr Pradeep Vyas, Additional Chief Secretary, Public Health Department, Government of Maharashtra expressed his gratitude to the doctors and the medical fraternity. He said ‘The doctors of our country who work selflessly need to be recognised for their exemplary service. Today, we are felicitating a few among them as a token of gratitude to the collective contribution of doctors to our society and I salute all of them for their dedication to provide quality healthcare. Let us all commit to continuously strive for excellence in our service to the nation.’
Speaking during the event Dr. Vipul Aggarwal, Deputy CEO, NHA said: ‘In my view it is not just a job, it’s a challenging commitment and service which our doctors manage to do without a single frown upon their face. As we move forward, we know that the pandemic would continue to evolve, and the health care community would also need to match the pace of this evolution to stay ahead of the game and deliver high-quality care to all patients.’
Dr. Neelima Kerkatta, Principal Secretary, Public Health Department, Government of Maharashtra and Dr. Sudhakar Shinde, CEO State Health Authority Assurance Society, Maharashtra were also present during the meeting.
With an objective to continue with its endeavour to reward and recognise the medical fraternity associated with the PM-JAY scheme on a regular basis, NHA announced the launch of following key initiatives:
Ayushman Bharat Utkrist Chikitsak Samman: An award to felicitate five best performing doctors working in the Ayushman Bharat PM-JAY ecosystem.
Ayushman Bharat Utkrist Chikitsalaya Samman: An award for one best performing hospital empanelled under Ayushman Bharat PMJAY in each state.
Ayushman Bharat PM-JAY Fellowship: Under this, as many as five healthcare professionals will be given a one-year fellowship grant to undertake research under Ayushman Bharat PM-JAY.
All these awards will be conferred during the anniversary celebration of AB PM-JAY in September 2022. Officials and doctors from State Health Agencies across various PM-JAY implementing States & UTs virtually participated in the felicitation event and it kicked off similar felicitation ceremonies across India to recognise the contribution of doctors.
The performance of States/UTs of Western Region under the AB PM-JAY scheme so far in providing treatment to eligible beneficiaries is as below: