“The management of the COVID19 pandemic, and especially the recent Omicron surge, has strongly demonstrated to the world the power of strong political will, self-reliance through Atma Nirbharta, innovation powered by technology, shared goals and collaborative efforts through a “Whole of Government” and “Whole of Society” approach”. This was stated by Dr Mansukh Mandaviya, Union Minister of Health and Family Welfare while delivering the keynote address at a webinar with NITI Aayog and more than 200 NGOs and Civil Society Organizations that have worked at the grassroots during the pandemic. The webinar was titled “India’s Public Health Response to manage COVID-19”.
At the outset, the Union Minister stated that “India’s strategies of public health response have resulted in better management of Omicron surge compared to many other countries”. In a presentation, steps taken by the country demonstrating various timely public health measures were highlighted. At a time when many countries are reporting an increase in number of daily COVID cases, India’s daily cases have reported a substantial decrease, combined with increased recovery rate, and high levels of vaccination. Timely measures including test, track and treat approach with focused genome sequencing, containment through containment zones, community surveillance, protocols for home isolation and effective clinical treatment have in tandem contributed to India’s COVID management.
Applauding collective spirit of the citizens, he said that “India’s vaccination drive is testament to India’s capabilities and power of the people without whom this journey, and this high level of vaccine coverage, would not have been possible.” Despite the large population combined with geographical and sociological diversities, India has set global standards of COVID vaccination, he added. “One of the key achievements has been our capacity to deliver over 1.8 billion Covid vaccine doses across a multitude of regions and terrains.”

Speaking about the challenges India faced during the pandemic, the country has demonstrated that ‘We can do it’, he stated. Timely and prompt decisions for containment, management, treatment and vaccination were taken by India under the leadership and guidance of Hon’ble Prime Minister, he stated. “We have shown the world what a decisive and powerful political will can achieve”, he noted.
He highlighted the crucial role played by technology and how it was leveraged for increased accessibility of healthcare services during the pandemic through eSanjeevani, CoWIN portal for vaccination, Arogya Setu app etc. India’s ttechnological innovations have lent speed and efficiency to the country’s vaccination drive, he stated.
Emphasising Indian Philosophy of ‘Vasudhaiva Kutumbakam’ and ‘Shubh Laabh’, he said, “India has not only produced quality and affordable vaccines, we have exported medicines to more than 150 countries on humanitarian grounds. The Vaccine Maitri program of the Government has been lauded globally.”
Dr Mandaviya expressed deep gratitude to all the grassroots NGOs, stakeholders and CSOs who have worked tirelessly among the communities during the pandemic to support them in various ways. “I seek your continued partnership for connecting with the communities to take healthcare services to them. Your work will be pivotal in enhancing awareness and accelerating uptake of e-health services (such as eSanjeevani, and tele-health) among the masses”, he stated.
Dr Mark Esposito, Harvard University stated that the commendable work done by the Government of India lays the foundation that will be able to develop a Government System, which is committed to prompt and science-based response. He also noted that this governance approach of “Whole of Government and Whole of Society” adopted by the Government of India, in terms of dealing with the pandemic and ensuring the success of the vaccination drive, would be a model for dealing with a crisis or similar challenges in the future.
Reaffirming India’s success being embedded in science, Dr. Chris Elias, President, Bill and Melinda Gates Foundation appreciated India’s exemplary response to the COVID Pandemic. He asserted, “However, in dealing with these challenges and ensuring the success of vaccination drive, crucial policy decisions that were taken swiftly, at various junctures, under the leadership and guidance of Prime Minister, for indigenous vaccine manufacturing, rigorous clinical trials and protocols for approvals to ensure the safety of millions of people, meticulous distribution through training of healthcare and frontline workers, AEFI management, to ensure that millions were safely administered COVID-19 vaccines in India played an instrumental role. India’s Pandemic Response and the strategies developed by the country has now become the textbook approach to deal with future pandemic.”
Outlining the distinctive initiatives that set India apart from many other countries in terms of COVID management, Shri Amitabh Kant, CEO, NITI Aayog said, “India's approach was technology-led which helped in achieving this whole of society approach. CoWIN app turned out to be the heart of the vaccination drive. Many challenges like delivery and fair equitable distribution of vaccines were addressed through timely transparent technological and physical interventions. Vaccine hesitancy was prevented through mass communication. Procurement, logistics were solved through robust cold chain ecosystem development. Also, we trained and provided clear information which helped in creating momentum for phase-wise vaccination of vulnerable groups.” He appreciated the role played by CSOs, NGOs in working on the ground. He also emphasized that we were able to achieve this stupendous feat only due to the political will and commitment, clarity of policies and taking all stakeholders in our approach, which shows the good governance model India has adopted.
Representatives from various Community Radio Stations (CRS) and CSOs shared their experience of working on the ground during pandemic. Various CRS stations helped to bust myths by providing authentic and accurate information, simplifying information in local dialects to the masses, providing various services to the communities during lockdown.
Ministry of Health and Family Welfare (MoHFW) has issued guidance notes to all the States and UTs regarding “Enabling Delivery of Essential Health Services during the COVID 19 Outbreak” on 14th April, 2020 and “Provision of Reproductive, Maternal, Newborn, Child, Adolescent Health plus Nutrition services during and post COVID-19 pandemic” on 24th May, 2020 for continuation of service delivery amid COVID-19 pandemic.
As per these guidance notes, the States and UTs have been advised to ensure home distribution of Iron Folic Acid supplementation to the target age groups i.e. Pre-school Children 6-59 months, Children 5-9 years, adolescents 10-19 years, Pregnant and lactating Women.
They have also been advised to undertake distribution of IFA supplements through the Village Health Sanitation Nutrition Days (VHSNDs) to be held in the staggered manner while ensuring all the personal protective measures and physical distancing norms by the frontline workers i.e. ASHAs/ANMs/AWWs.
The Government of India has also developed and launched Anaemia Mukt Bharat (AMB) e-Training Modules to facilitate training of the health care providers through virtual platform amid COVID 19 pandemic.
Under National Health Mission, financial support is provided to the States and UTs for effective implementation of interventions under AMB strategy based on proposals submitted through their respective annual Programme Implementation Plan (PIP). Sufficient funds are made available to the States/UTs for carrying out activities pertaining to addressing anaemia. The details on budgetary allocation for activities towards the reduction of anemia are placed at Annexure.
Annexure
State/UT wise Approvals towards Anaemia under NHM for the F.Y. 2021-22

The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
National Pharmaceutical Pricing Authority (NPPA) under the aegis of the Department of Pharmaceuticals fixes the ceiling price of scheduled medicines as per provisions of the Drugs (Prices Control) Order, 2013. In case of non-scheduled medicines, a manufacturer is at liberty to fix the Maximum Retail Price (MRP) launched by it, but is not allowed to increase the MRP of such formulations by more than 10% per annum. NPPA has fixed ceiling prices of 886 scheduled formulations & 4 scheduled medical devices and fixed retail prices of 1,817 new drugs. In addition, it has fixed the ceiling price of Orthopedic Knee Implants under Para 19 of DPCO, 2013 in public interest. Moreover, under “Trade Margin Rationalisation” Approach, the NPPA has fixed Trade Margin of non-scheduled formulations of 42 select Anti-cancer medicines and recently during COVID-19 pandemic, regulated the prices of Oxygen Concentrators, Pulse Oximeter, Blood Pressure Monitoring Machine, Nebulizer, Digital Thermometer and Glucometer.
With an objective of making quality generic medicines available at affordable prices to all, Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP) has been launched by the Government wherein dedicated outlets are opened to provide quality generic medicines at cheaper rates to the citizens. Under the Scheme, till 28.02.2022, about 8,689 Pradhan Mantri Bhartiya Janaushadhi Kendras (PMBJKs) have been opened across the country, covering all districts of the country. Jan Aushadhi Medicines are cheaper by 50 -90% of market prices of branded medicines. During the current financial year (till 28.02.2022), total sales of Rs. 814.21 cr. has been achieved, which has led to estimated savings of approximately Rs. 4,800 cr. to the citizens.
In order to attain self-reliance in the pharmaceutical sector, develop global champions and to retain the position of the country as ‘Pharmacy of the world’, the government has launched two Production Linked Incentive (PLI) schemes. PLI scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates (DIs)/ Active Pharmaceutical Ingredients (APIs) was launched with a total financial outlay of Rs. 6,940 crore. Under the scheme, 49 applicants have already been approved. The other PLI Scheme for Pharmaceuticals has a total financial outlay of Rs. 15,000 crore. Under this scheme, 55 applicants have been selected. In addition, the department has launched a Scheme for Promotion of Bulk Drug Parks with a total outlay of Rs. 3,000 cr wherein financial assistance is to be provided for creation of Common Infrastructure Facilities in 3 Bulk Drug Parks.
The information was given by the Union Minister of Chemicals and Fertilizers, Shri Mansukh Mandaviya in a written reply in the Rajya Sabha on 15/03/2022.
With an objective of making quality generic medicines available at affordable prices to all, Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP) was launched by the Department of Pharmaceuticals, Ministry of Chemicals & Fertilizers, Government of India. Under the scheme, dedicated outlets known as Pradhan Mantri Bhartiya Janaushadhi Kendra (PMBJKs) are opened to provide generic medicines at cheaper rates to the citizens of the country.
As on 28.02.2022, about 8689 PMBJKs have been opened up in the country covering all districts of the country. State/Union Territory-wise list of PMBJKs is enclosed as Annexure.
The Government has set a target to have about 10,500 PMBJKs by March 2025 across the country. The target for the year 2022-23 is to have about 9,300 PMBJKs across the country.
The details of the funds allocated and utilized under the Scheme for the financial years 2020-21 and 2021-22 is as under:

There is no State/UT-wise specific budget allocated under the Scheme.
Annexure
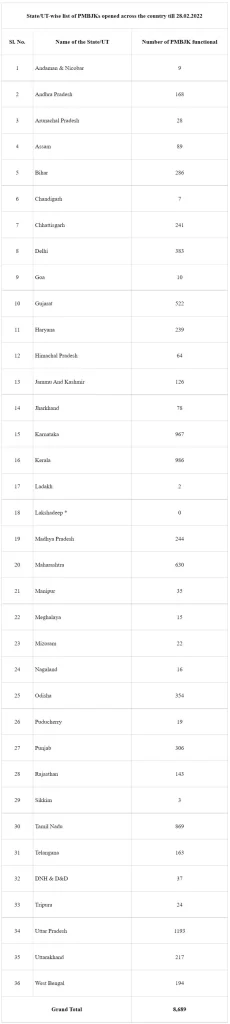
* Medicines are directly supplied to the administration of UT of Lakshadweep.
The information was given by the Union Minister of Chemicals and Fertilizers, Shri Mansukh Mandaviya in a written reply in the Rajya Sabha on 15/03/2022.
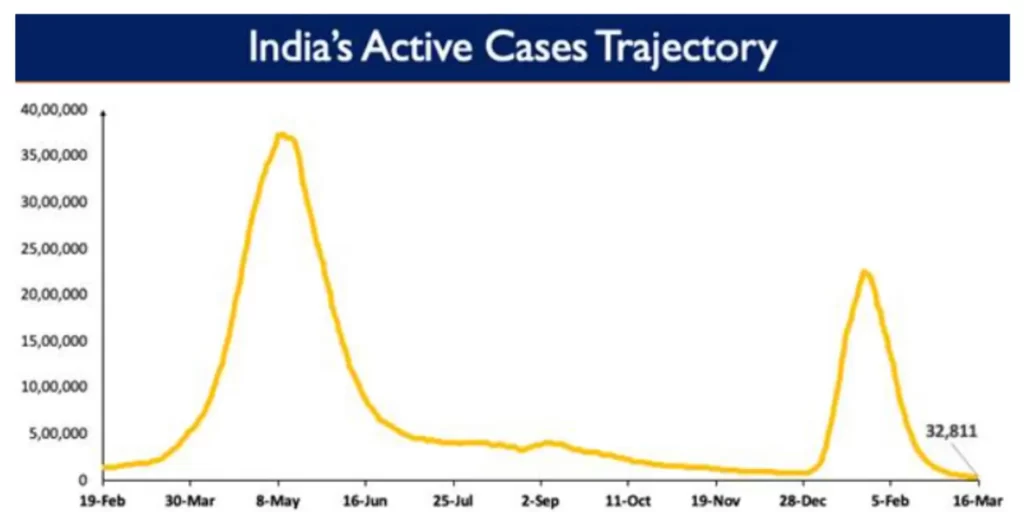
Following a continuous downward trend, India’s active caseload has declined to 32,811 today. Active cases constitute 0.08% of the country's total positive cases.
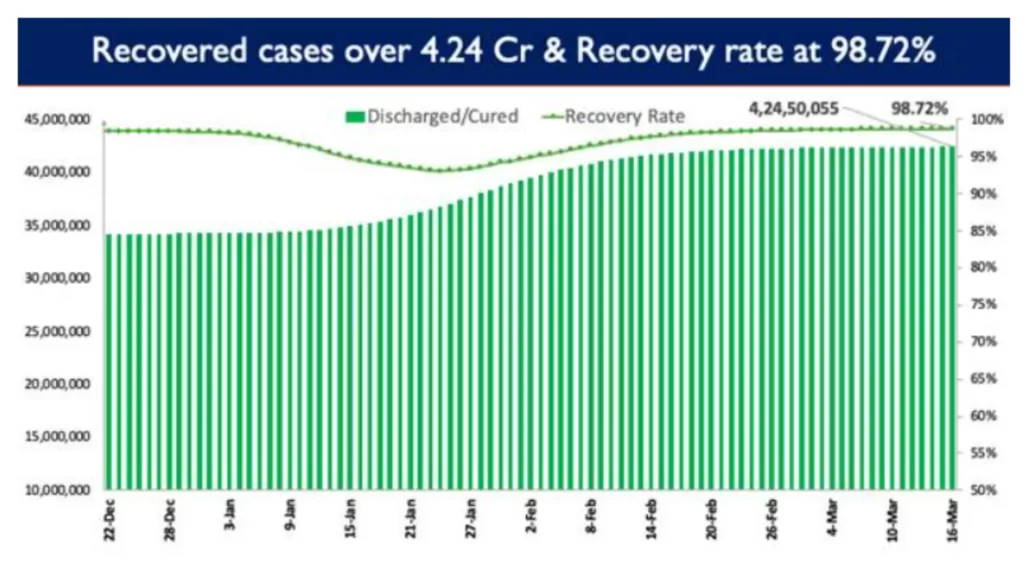
Consequently, India’s recovery rate stands at 98.72%. 3,884 patients have recovered in the last 24 hours, and the cumulative tally of recovered patients (since the beginning of the pandemic) is now at 42,450,055.
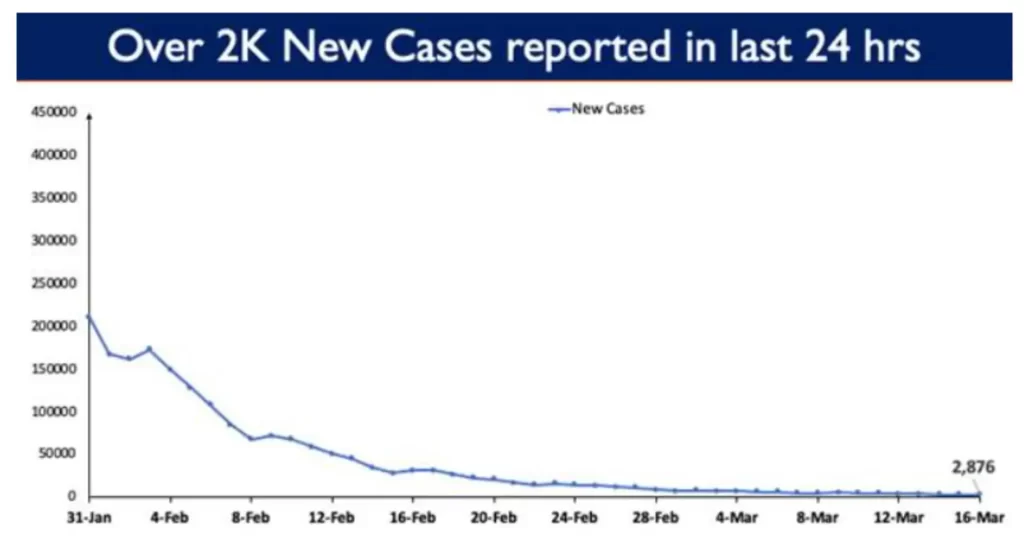
2,876 new cases were reported in the last 24 hours.
The last 24 hours saw a total of 752,818 COVID-19 tests being conducted. India has so far conducted over 78.05 Cr (780,506,974) cumulative tests.
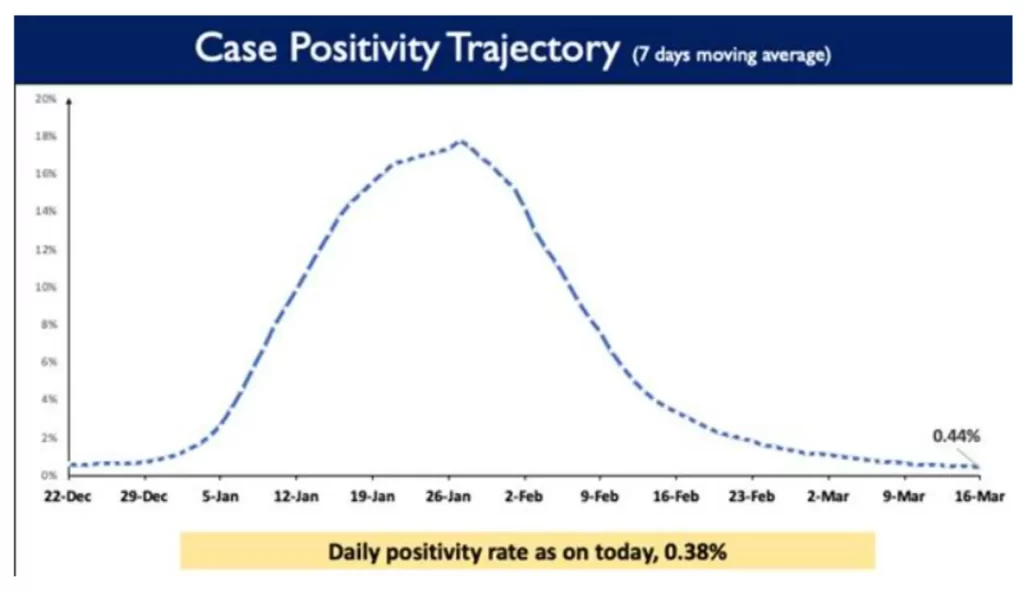
There has been a sustained fall in the weekly and daily positivity rates too. The weekly positivity rate in the country currently stands at 0.44%, and the daily positivity rate is reported to be 0.38%.
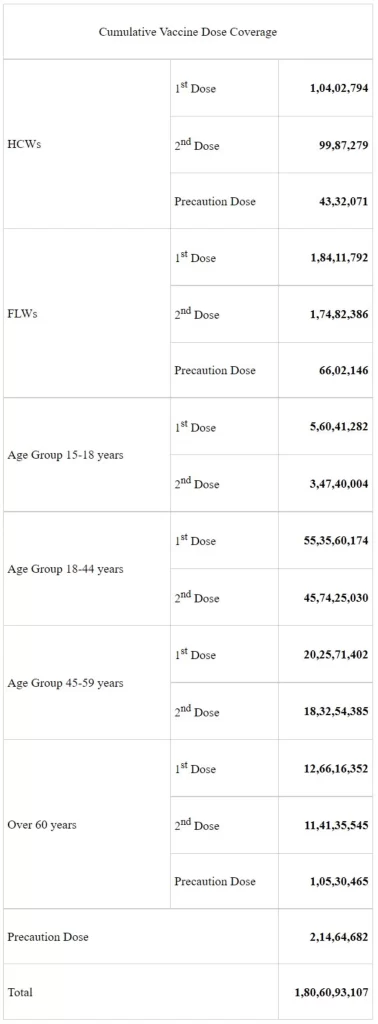
On the vaccination front, India’s COVID-19 vaccination coverage has exceeded 180.60 Cr (1,806,093,107) as per provisional reports till 7 am today. This has been achieved through 21,193,183 sessions. The break-up of the cumulative figure as per the provisional report till 7 am today includes
As per Constitutional provisions, ‘Health’ and ‘Law & Order’ are State subjects. State Governments are expected to set up mechanisms to prevent violence against doctors including imposition of penalties or setting up Helplines to extend immediate help to such Doctors who are victims of violence. Details of number of instances of attacks on doctors are not maintained centrally.
Further, violence against healthcare professionals is a criminal offence and needs to be dealt suitably by the State /UT Governments under provisions in Indian Penal Code (IPC)/ Code of Criminal Procedure (CrPC) so that doctors/clinical establishments discharge their professional pursuit without fear of violence.
In the wake of COVID-19 outbreak, Government of India has notified the Epidemic Diseases (Amendment) Act, 2000 on 28th September 2020. The Amended Act provides that acts of violence against healthcare personnel during any situation akin to current pandemic would be cognizable and non-bailable offences. Commission or abetment of such acts of violence shall be punished with imprisonment for a term of three months to five years, and with fine of Rs.50,000/- to Rs.2,00,000/-. In case of causing grievous hurt, imprisonment shall be for a term of six months to seven years and with fine of Rs.1,00,000/- to Rs.5,00,000/-. In addition, the offender shall also be liable to pay, compensation to the victim and twice the fair market value for damage of property.
The Union Ministry of Health & Family Welfare has, in order to prevent violence against doctors and for inculcating an effective sense of security among the doctors on duty, taken several measures including issuing advisories in this regard to all States/Union Territories (UTs) to consider the following:
Security of sensitive hospitals to be managed by a designated and trained force,
Installation of CCTV cameras and round the clock Quick Reaction Teams with effective communication / security gadgets particularly at Casualty, Emergency and areas having high footfalls,
Well-equipped centralized control room for monitoring and quick response,
Entry restriction for undesirable persons,
Institutional FIR against assaulters,
Display of legislation protecting doctors in every hospital and police station,
Appointment of Nodal Officer to monitor medical negligence,
Expeditious filling up of vacant posts of doctors and para-medical staff in hospitals / Primary Health Centres (PHCs) to avoid excessive burden / pressure on doctors and to maintain global doctor-patient ratio,
Better infrastructural facilities and medical equipment and provision of extra monetary incentive for the doctors and para medical staff serving in hard/remote areas as compared to major and metro cities with better career prospects, etc.
The Ministry of Health & family Welfare has on multiple occasions, through formal communications as well as more recently through video conferences with States and UTs, highlighted the need to ensure safety and security of healthcare workers at their living/working premises.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The CoWIN Portal is the digital platform to capture COVID-19 vaccination program details. CoWIN connects to various stakeholders, including vaccine manufacturers, administrators, and verifiers; public and private vaccination facilities; vaccine recipients, etc.
The CoWIN platform was developed in a record speed with ample consideration to its scalability, modularity, and interoperability. For quick scale, CoWIN has been integrated with other government mobile applications such as Aarogya Setu and UMANG. Users can access the platform via desktop, laptop, tablet, and mobile phones and can use it to meet a variety of needs. The platform has adapted quickly to the need for covering different beneficiary groups during each phase and adjusted the platform to accommodate an influx of new users, even those from remote areas and/or those without access to digital tools. Registered users receive automated text messages on their registered mobile numbers regarding vaccination appointments, second dose & precaution dose reminders, and links to digital vaccination certificates.
Nine countries have sought India’s cooperation in this endeavour.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar, stated this in a written reply in the Rajya Sabha today.
Free of cost COVID-19 vaccination for all beneficiaries of the 12-14 age group will begin tomorrow (16th March 2022), National Vaccination Day, at all government COVID vaccination centers. The COVID-19 vaccine to be administered would be Corbevax, manufactured by Biological E. Limited, Hyderabad. This can be through online registration (beginning 9 am on 16th March 2022) or through onsite walk-in. This was reiterated by Union Health Secretary Shri Rajesh Bhushan in a meeting through video conference (VC) today with all states and UTs.
This follows the decision of the Union Government to start COVID-19 vaccination for 12-13-year-olds and 13-14-year-olds (those born in 2008, 2009, and 2010). i.e., those who are already above 12 years of age) from 16th March 2022. Additionally, all above 60 years of age are now eligible for the precaution dose from tomorrow, as the condition of comorbidity for this age group has been removed. The precaution dose (same as the previous two doses) is to be administered 9 months (36 weeks) after the date of the second vaccination. Detailed instructions and operational guidelines have been sent to states and UTs in this regard.
States were advised to ensure that only those who have attained the age of 12 years on the date of vaccination are vaccinated against COVID-19; if the beneficiary is registered but has not attained the age of 12 years on the date of vaccination, the COVID-19 vaccine is not to be given. Vaccinators and vaccination teams need to be trained to ensure that, particularly for the 12-14-year-old age group, there is no mixing of vaccines. States were advised to organize dedicated vaccination sessions through earmarked COVID-19 Vaccination Centers for vaccination of the 12-14-year-' age group to avoid mixing with other vaccines.
Vaccines that can be used in different age groups
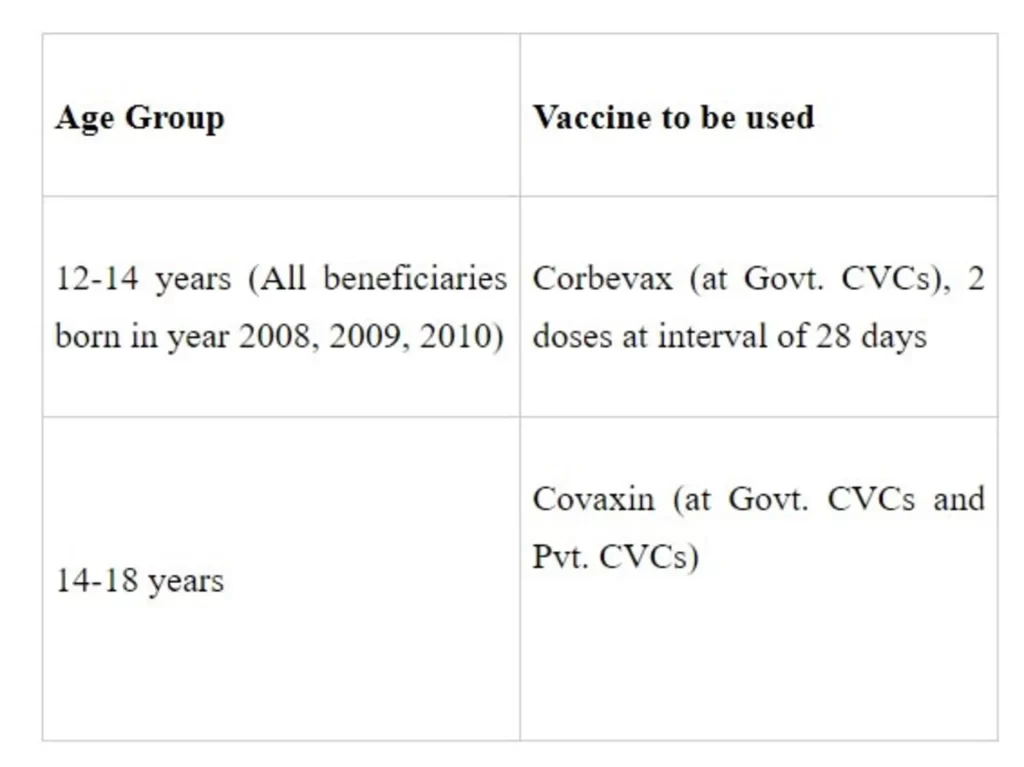
States/UTs were informed that at present the age of beneficiary in CoWIN is being filtered based on year of birth. The responsibility for verification of age (12 years) will lie with the vaccinator/verifier at the time of vaccination for the first few days, as the provision for recording the exact date of birth in the Co-WIN portal is under process. Once implemented, the system, by default, will not allow registration of beneficiaries who are not of the recommended age.
Underscoring the slow pace of vaccination among the vulnerable groups, states were urged to ensure that all those above 60 years of age are covered with both doses of the COVID-19 vaccine. Regular reviews are to be conducted at district & block levels to ensure coverage of eligible beneficiaries.
States were also advised to ensure judicious utilization of available COVID-19 vaccines. As per earlier guidelines, states can replace those that are due to expire and also replace them from one district within the state to another to ensure that vaccines are not wasted.
Dr. Manohar Agnani, Addl. Secy. (Health), and other senior officers of the Union Health Ministry were present in the virtual meeting along with the Health Secretary and NHM Mission Directors and other officials from states/UTs.
Studies at various ambient temperature and duration are required to finalize when the vaccine reaches end point beyond which it cannot be used to decide the type of Vaccine Vial Monitoring (VVM) to be used. VVM is manufactured by a single offshore manufacturer and in view of the global urgency to use COVID-19 vaccine upon development, requirement of very high volumes of VVM and limited production capacity, COVID-19 vaccines are used without VVM across the world.
The National Regulator i.e. Central Drugs Standard Control Organization, has approved the shelf life of 9 months for Covishield vaccine and 12 months for Covaxin & Sputnik vaccine from the date of manufacture. Government of India has issued suitable guidelines and training material highlighting appropriate storage, handling and administration of Covid-19 vaccines.
The storage and transportation of COVID-19 vaccines from the manufacturer up to the Covid-19 Vaccination Center (CVC) is ensured in a temperature-controlled environment at 2-8 degree Celsius. A robust Cold Chain Network of around 29,000 cold chain points along with insulated/refrigerated vaccine vans, Cold Boxes and Vaccine carries is used to ensure storage and transport of COVID-19 vaccines in a temperature-controlled environment. Monitoring of storage temperature is also done on real time basis so as to ensure maintenance of recommended storage temperature.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
Department of Pharmaceuticals, Ministry of Chemicals and Fertilizers has extended the date of receipt of applications for the vacant slots under “Production Linked Incentive (PLI) Scheme for Promotion of Domestic Manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates and Active Pharmaceutical Ingredients (APIs) in the Country” shortly called as PLI scheme for Bulk Drugs till the end of March 2022.
The Department has issued notice on 27th January 2022, inviting the applications for vacant slots (10 APIs) under PLI Scheme for Bulk Drug with the last date of application as 13.03.2022. Based on the representation received from the Industries/Associations, the Department has decided to extend the timeline for filling of application upto 31.03.2022.
Department is implementing three PLI schemes viz PLI scheme for Bulk Drugs (Rs.6,940 Cr), PLI scheme for Medical Devices (Rs.3,420 Cr) and PLI scheme for Pharmaceuticals (Rs.15,000 Cr)} to increase the domestic manufacturing of Bulk Drugs, Medical Devices and pharmaceuticals respectively and to create global champions in the field of Pharmaceuticals and Medical Devices (IVDs).
PLI Scheme for Bulk Drugs, approved by the Cabinet in the year 2020, is the flagship scheme, aiming to attain self-reliance and reduce import dependence of critical KSMs/DIs/APIs and attract large investments in the API Sector. To realise the vision of AtmaNirbhar Bharat, setting up of green field plants with minimum domestic value addition of 90% in four different target Segments (Two Fermentation based and Two Chemical Synthesis based) with a total outlay of Rs. 6,940 cr. for the period 2020-21 to 2028-29 will be undertaken.
Under two rounds of inviting applications, a total 49 projects have been approved for 33 critical APIs with a committed investment of ₹ 3,685 crore. It is to inform that against a notified quantity of about 44,000 MT of annual production capacity of these APIs, industry has responded very well with the committed production capacity of 83,270 MT. The Hon’ble Union Minister for C&F Dr. Mansukh Madaviya, while interacting with the commissioned projects under the PLI scheme on 25.2.2022 had complimented the industry representatives for this achievement towards attaining the vision of Prime Minister for self-reliance in critical drugs. He encouraged the industry to invest in the area of research and innovation for sustainable global competitiveness by allocating adequate resources.
The IFCI Ltd, a Statutory Non-Banking Finance Company in the public sector, is the Project Management Agency for this PLI scheme. Details of the schemes can be accessed at https://pharmaceuticals.gov.in/schemes and the Bulk Drug Industries can apply for the vacant slots at https://plibulkdrugs.ifciltd.com/
