The extant National Pharmaceutical Pricing Policy (NPPP), notified on 7th December, 2012, has been formulated with an objective to put in place a regulatory framework for pricing of drugs so as to ensure availability of essential medicines at reasonable prices while providing sufficient opportunity for innovation and competition to support the growth of pharma industry. The policy made a shift from earlier ‘cost based’ pricing under the Drug Policy, 1994 to ‘market based’ pricing.
In pursuance of NPPP, 2012, the Government notified the Drugs (Prices Control) Order, 2013 (DPCO-2013). As per the provisions of DPCO, 2013, the ceiling price of all scheduled formulations appearing in National List of Essential Medicines (NLEM), are fixed by National Pharmaceutical Pricing Authority (NPPA) and are uniform throughout the country. All the manufacturers of these drugs are required to sell their product equal to or lower than the ceiling price. Further, NPPA monitors the prices of non-scheduled drugs so as to ensure that the increase in their Maximum Retail Price (MRP) is not more than 10% of what was prevalent during preceding twelve months.
The information was given by the Union Minister of Chemicals and Fertilizers, Shri Mansukh Mandaviya in a written reply in the Lok Sabha today.
The National Digital Health Mission (now known as Ayushman Bharat Digital Mission) was launched on 15th August 2020 as a pilot in six Union Territories - Andaman & Nicobar, Chandigarh, Dadra & Nagar Haveli and Daman & Diu, Ladakh, Lakshadweep and Puducherry.
The pilot phase of ABDM was successfully conducted between 15th August 2020 to 27th September 2021.Three key registries of NDHM namely Health ID, Health Professional Registry (HPR), Health Facility Registry (HFR) and digital infrastructure for data exchange have been developed and implemented in these UTs. On 27th September 2021, the national rollout of the ABDM has been announced.
Till 25th January 2022, 15,05,92,811 Health IDs (now known as ABHA – Ayushman Bharat Health Accounts) have been created in the country. A total of 15,016 health facilities and 8,378 doctors have been registered under ABDM ecosystem.
Creation of Health ID is voluntary. National Health Authority is creating awareness among the citizens about the usages and advantages of Health ID to maximize participation from citizen.
ABHA numbers (earlier known as Health IDs) have been created for those beneficiaries enrolled through CoWIN platform who have provided Aadhaar as the ID proof at the vaccination site only after taking their consent through the vaccinator.
The Information Technology Act 2000 along with policies and guidelines including but not limited to Health Data Management Policy, Sandbox Guidelines etc. published by ABDM from time to time lay down the regulatory and policy framework for the regulation of ABDM.
Besides a part of the funds available at the level of hospitals, Union Health Ministry is also supporting States and UTs in implementation and strengthening of health IT initiatives such as Hospital Management Information Systems, Telemedicine services etc. in public health facilities through National Health Mission.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the LokSabha today.
Private hospitals under Ayushman Bharat – Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) are empanelled by States/UTs based on well-defined criteria laid down by National Health Authority (NHA). The criteria include, among others, minimum in-patient bed strength and speciality-wise infrastructure facilities including equipment. The average bed strength of hospitals empanelled under the scheme is 45.
As of 28th January 2022, approximately 8.5 Lakh hospital admissions are authorized for the treatment of COVID-19 under the scheme. State/UT-wise details are at Annexure-I.
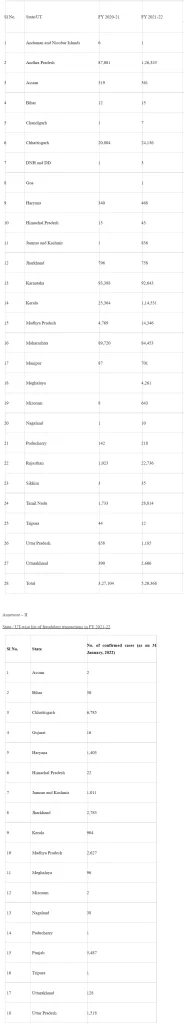
As of 31st January 2022, the number of fraudulent transactions recorded at Empanelled Healthcare Providers in the year 2021-22 is 22,874. State/UT-wise details thereof are at Annexure-II.
Annexure – I
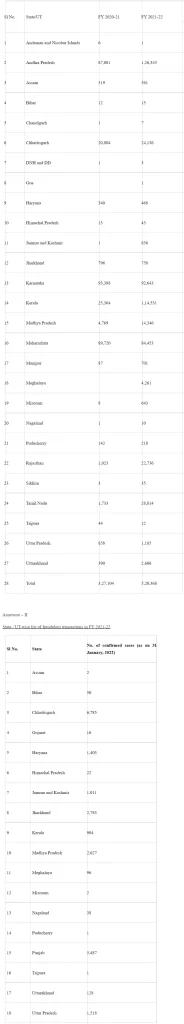
Authorized hospital admissions for COVID-19 treatment FY 2020/21 to FY 2021/22
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
India is the third largest player globally in Pharmaceuticals in terms of volume and is the largest supplier of low cost generics and vaccines to the world. The Government is also taking steps in the form of various schemes to attract investments in pharmaceuticals and medical devices sectors, including for the Active Pharma Ingredients (API) manufacturing.
The details of the scheme bolstering the Active Pharma Ingredients (API) manufacturers in India isgiven below:
(i) Production Linked Incentive Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates (DIs)/ Active Pharmaceutical Ingredients (APIs) in India.
The Union Cabinet approved the scheme on 20.03.2020 with the objective to boost domestic manufacturing of identified KSMs, DIs and APIs.The tenure of the scheme is from FY 2020-21 to 2029-30, with the total financial outlay of Rs. 6,940 crores.
The Financial incentive under the sub-scheme is provided on sales of 41 identified products categorized into four Target Segments.
PLI Scheme for Pharmaceuticals: -
Eligible drugs under this schemeapproved on 24.02.2021 include Active Pharmaceutical Ingredients among other categories of pharmaceutical products. The Operational Guidelines have been issued on 1.6.2021.
The total financial outlay of the scheme is Rs. 15,000 crore and the tenure of the scheme is from FY 2020-2021 to 2028-29.
Scheme for Promotion of Bulk drug parks
The scheme of “Promotion of Bulk Drug Parks” approved on 20th March, 2020. The scheme provides for support to establish three (03) Bulk Drug Parks for creation of world class Common Infrastructure Facilities (CIF). Guidelines of the scheme were notified on 27th July 2020.
The total financial outlay of the scheme is Rs. 3000 crores. The tenure of the scheme is from FY 2020-2021 to FY 2024-2025.
This information was given by the Minister of State in the Ministry of Commerce and Industry, Smt. Anupriya Patel, in a written reply in the Lok Sabha today.
Observing the 3rd World Neglected Tropical Diseases (NTDs) Day yesterday, as a key moment to highlight the global community’s commitment to ending NTDs, India joined close to 40 other nations to illuminate the iconic New Delhi Railway Station in purple and orange hues, which is one of the busiest railway stations in the country in terms of train frequency and passenger movement.
At the virtual event organized by National Centre for Vector Borne Diseases Control (NCVBDC) to observe World NTD Day, Dr Harmeet Singh Grewal, Joint Secretary, Union Ministry of Health and Family Welfare highlighted that the aim of illuminating the iconic New Delhi Railway Station was to generate awareness about NTDs in the visiting public and showcase progress and achievements towards their elimination. He also emphasized on creating a people’s movement to eliminate the NTDs. “There is a need for a behavioral change. We must fight it together and engage school children, social workers, activists etc. to create a mass movement to elimination NTDs.”
NTDs are caused mostly by a variety of pathogens including viruses, bacteria, parasites, fungi, and toxins. They affect over 1.7 billion people globally. They are “neglected” because they are almost absent from the global health agenda of the developed countries and are associated with stigma and social exclusion. Lauding India’s efforts towards eliminating these diseases, with a keen focus on Lymphatic Filariasis, Dengue and Visceral Leishmaniasis, Dr. Tanu Jain, Director, NCVBDC said, “Most people don’t know about NTDs because these diseases effect the poor and marginalized communities. We need to elevate messaging about NTDs and highlight the positive, ambitious, and winnable NTD goals by focusing on the NTD elimination target. This would happen through close coordination between all stakeholders.”
In Jharkhand, Gujarat and Karnataka too, iconic landmarks and monuments were illuminated as part of their commitment to eliminate NTDs. In Jharkhand, renowned Rajendra Circle in Ranchi and Clock Tower in Godda district were lit up in pink and orange colours in observance of the World NTD Day. The office building of the Department of Health and Family Welfare in Bangalore was also illuminated to mark the World NTD Day.
“Mutual understanding, sharing best practices and collaborative spirit between the Centre and States have helped us in our fight against the pandemic”. This was stated by Dr. Mansukh Mandaviya, Union Minister of Health and Family Welfare as he applauded the remarkable synergy between the Centre and the States today during the Video Conference interaction with State Health Ministers and Principal Secretaries/ Additional Chief Secretaries, and Administrators of eight southern States/UTs (Karnataka, Kerala, Tamil Nadu, Andhra Pradesh, Telangana, Puducherry, Andaman & Nicobar Islands, and Lakshadweep) in the presence of Dr. Bharati Pravin Pawar, Union Minister of State for Health and Family Welfare. Chief Minister of Puducherry, Shri N Rangaswamy was also present at the meet.

The virtual meeting was held to review the public health preparedness for containment and management of COVID19 and progress of national COVID19 vaccination campaign.State Health Ministers who joined the high level review meeting included Dr. K Sudhakar (Karnataka), Dr. Veena George (Kerala), Shri Ma Subramaniam (Tamil Nadu) and Shri Thanneeru Harish Rao (Telangana). The State Health Ministers unanimously thanked the Union Health Minister for the continuous support of the Union Government and providing them the required doses of COVID vaccination.

Highlighting India’s remarkable first and second dose adult vaccination coverage of 95% and 74% respectively, the Union Health Minister stated that "India's COVID19 vaccination drive is a global success story, especially for such a populous country like ours". To give a further boost to the nationwide COVID19 vaccination drive, we have now started the Precaution Dose and the vaccination for the 15-17 yrs age group from this month, he added. He noted that more than the projected requirements of doses of both vaccines were provided to the States and UTs to ensure that there is no let-up in pace of the vaccination drive. He requested the states to accelerate the pace of vaccination of the 15-17 yrs age group and those whose second dose is due.
Stressing the role that tele-consultation and tele-medicine have played in the country to serve the far flung regions and those in home isolation, Dr Mandaviya advised the States to focus on ramping up opening of more tele-consultation centres as part of the Hub and Spoke Model for better public health management. “The tele-consultation centres will help us not only during COVID pandemic but also for non-COVID medical care”, he said. Kerala informed that non-COVID medical care for cancer, diabetes management, mental health etc., were also provided through tele-medicine centres to those in home isolation who could not access the institutional facilities. Andhra Pradesh and Karnataka which have shown good progress in eSanjeevani with very large number of tele-consultations were appreciated for their efforts.
The Union Health Minister underlined the importance of sturdy and resilient health infrastructure and the ECRP-II package under which the funds have been provided to states and UTS and need to be utilised before 31st March, 2022. “While some states have expedited effective utilisation of the approved funds for health infra creation, other states may also review the physical and financial progress under ECRP-II and expedite the progress”, he advised the states.
Dr. Mandaviya reiterated that the 5-fold strategy of ‘Test-Track-Treat-Vaccinate & Adherence to COVID Appropriate Behaviour’ along with effective surveillance of cases remains crucial for COVID management. States and UTs were advised to keep a close watch on the emerging clusters and hotspots. Those states which have reported lower share of RTPCR in COVID testing were advised to review the same. Adequate and timely testing will help in prompt identification of the infected cases and prevention of a sudden surge, they were advised.
Assuring states of all support from Centre in their efforts for COVID response and management, the Union Health Minister urged them to provide data on time as it will lead to more sturdy and efficient policy making.
Union Minister of State for Health, Dr. Bharati Pravin Pawar encouraged all the states to fully utilise the ECRP-II funds. She urged the states/UTs to strengthen laboratories, place timely purchase orders of medicines in case of shortages, expedite commissioning of PSA plants and open more teleconsultation centres. She highlighted that tele-consultation will help in treating patients in future and emphasized on the need to give special attention to co-morbid patients.
There was comprehensive and detailed discussion on various aspects of COVID management including regular monitoring of COVID tests, hospital bed occupancy, increasing positivity and active cases, ramping up of hospital infrastructure; increased testing; stringent restrictive measures for breaking the chain of transmission; and stress on COVID Appropriate Behaviour among the masses. Karnataka informed of the 8 war rooms established across 8 regions that were monitored by senior officers. The State Health Minister informed about the augmentation of the medical HR to handle the COVID pandemic. States like Telangana and Andhra Pradesh mentioned about conducting “fever survey” where healthcare workers went door-to-door to monitor residents for fever and SARI/ILI. Telangana highlighted that a hospital bed management system is installed in all public and private hospitals giving real time information on bed availability in hospitals. In addition, a multi-dept group has been created for monitoring households and supporting them with medicines, testing kits etc. These teams visit nearly 40-50 households every day.
Shri Rajesh Bhushan, Union Health Secretary, Dr Balram Bhargava, DG ICMR, Dr. Manohar Agnani, AS (Health Ministry), Shri Lav Agarwal, JS (Health Ministry), Dr. Sujeet Singh, Director, NCDC and senior officials from states and UTs were present in the meeting.
The National Regulator, Drugs Controller General of India (DCGI), has given nod to market authorization of two COVID19 vaccines, Covaxin and Covishield subject to certain conditions, here today. The Subject Expert Committee (SEC) of the Central Drugs Standard Control Organization (CDSCO) had recommended for upgradation of status for the vaccines from restricted use in emergency situations to grant of new drug permission with conditions in the adult population on 19th January 2022.
The market authorization of two COVID19 vaccines, Covaxin and Covishield, in the country by DCGI is subject to the following conditions:
1. Firm shall submit data of overseas ongoing clinical trials of the product with due analysis on six monthly basis or as and when available, whichever is earlier.
2. The vaccine shall be supplied for programmatic setting and all vaccinations done within the country to be recorded on CoWIN platform and Adverse Event Following Immunization [AEFI], Adverse Event of Special Interest [AESI] shall continue to be monitored. The firm shall submit the safety data including AEFI and AESI with due analysis on six monthly basis or as and when available, whichever is earlier as per NDCT Rules, 2019.
The proactive and agile approach followed by Government of India has been a hallmark of its strategy of management of COVID19. The latest approval accorded by DCGI for conditional market authorization to two COVID19 vaccines in the country indicates the promptness and timeliness with which the public response strategy and decision making apparatus of the country has responded to the emerging needs during the pandemic.
It may be noted that of the global Stringent Regulatory Authorities, only the United States Food and Drug Administration (USFDA) Medicines and Healthcare products Regulatory Agency (MHRA) of the UK have granted “conditional market authorization” to Pfizer and AstraZeneca, respectively, for their COVID19 Vaccines.
“Conditional Market Authorization” is a new category of market authorization that has emerged during the current global pandemic of COVID19. The approval pathways through this route are fast-tracked with certain conditions to enhance the access to certain pharmaceuticals for meeting the emerging needs of drugs or vaccines.
India’s nation-wide COVID19 vaccination program was launched on 16th January, 2021. As of today, more than 160 crore doses have been administered. The Union Government is committed to accelerating the pace and expanding the scope of COVID-19 vaccination throughout the country. New categories of population have been added to the national COVID19 vaccination drive starting from 3rd January 2022.
“In view of a substantial number of Active Cases across the country recuperating from COVID19 in home isolation, it is important to focus on expanding the reach of tele-consultation services for providing timely quality healthcare to the beneficiaries”. This was stated by Dr. Mansukh Mandaviya, Union Minister of Health and Family Welfare today as he interacted with State Health Ministers and Principal Secretaries/ Additional Chief Secretaries, and Administrators of nine states/UTs (Jammu & Kashmir, Himachal Pradesh, Punjab, Chandigarh, Uttarakhand, Haryana, Delhi, Ladakh and Uttar Pradesh) in the presence of Dr. Bharati Pravin Pawar, Union Minister of State for Health and Family Welfare and Dr V K Paul, Member (Health), NITI Aayog. He urged the States and UTs to adopt the hub and spoke model and ensure that more and more centre of tele-consultation are opened. This will enable beneficiaries to access expert advice from experts stationed at the district hubs. He added that eSanjeevani has been able to provide services to more than 2.6 crore beneficiaries where people can seek medical advice from the confines of their homes. “This will prove to be a game-changer and will be of immense value and importance for the hard-to-reach and far flung areas, and especially in the northern regions in the current winter season”, he highlighted. “States/UTs need to ensure that these centres work 24X7 and ensure convenience for both the common public and health experts. To ensure minimum hassle and travelling, such expert advice can be provided at the block levels, secondary or primary health centre levels too”, he stated. He advised states and UTs to ensure that those in home isolation are efficiently monitored in line with the National Guidelines. This will ensure that the vulnerable categories of active cases in home isolation get the required medical help in a timely manner.
The virtual meeting was held to review the public health preparedness for containment and management of COVID19 and progress of national COVID19 vaccination campaign. State Health Ministers who joined the high level review meeting included Shri Anil Vij (Haryana) and Dr. Dhan Singh Rawat (Uttarakhand). The State Health Ministers expressed their gratitude to the Union Health Minister for the continued support from the Centre in the fight against the COVID-19 pandemic.
Reiterating that the Centre is dedicated to supporting States/UTs in containment and management of COVID-19, Dr Mansukh Mandaviya requested the 9 States and UTs to review and expedite implementation of activities under ECRP-II package for strengthening the health infrastructure. He urged the Health Ministers and the State authorities to plug the existing gaps by efficiently utilizing the amount sanctioned for various infra projects. “With strengthened health infrastructure, we can meet any health emergency and public health crisis with better preparedness”, he said. States were advised to ensure regular updation of availability, occupancy and use of infrastructure like hospital beds, PSA plants, oxygen equipments on the COVID-19 portal- https://covid19.nhp.gov.in/. It was noted that this updation would help in obtaining a bigger picture and taking data-driven decisions at several levels for efficient public health response.
The Union Health Minister re-emphasised the need for ramping up testing in the states and UTs. Those states/UTs that are showing a lower share of RTPCR testing were requested to ramp up tests through RTPCR. States and UTs were also reminded to keep a close watch on the emerging clusters and hotspots and monitor the trend of hospitalised cases along with the deaths in the state.
Dr Mandaviya stressed that “Vaccination remains a potent tool to fight COVID-19. Vaccinated people are less prone to severity and hospitalisation as seen in India and globally. Higher hospitalisation is being seen in unvaccinated people and hence it becomes crucial to vaccinate the unvaccinated.” He also requested States to promote vaccination among 15-18 yrs age group and ensure full coverage in the districts that have shown a lower coverage of the first and second doses.
Dr. Mandaviya said that with our past experience, ‘Test-Track-Treat-Vaccinate & Adherence to COVID Appropriate Behaviour’ along with monitoring of cases remains crucial for COVID management.
Union Minister of State for Health, Dr. Bharati Pravin Pawar stressed on the need for vigilance and alertness during the ongoing upsurge. She urged the states/UTs to ensure availability of essential medicines for any evolving situation and ensure timely purchase orders in case of shortages.
The States and UTs shared a snapshot of activities being undertaken for COVID management and best practices for COVID management in addition to trend of COVID trajectory, hospitalisation, bed occupancy, testing and vaccination data. States like Uttar Pradesh mentioned about the Nigrani Samitis for door-to-door monitoring of those in home isolation. UT of Ladakh is providing medicines, nutrition supplements, and immunity boosters to patients in home isolation.
Shri Rajesh Bhushan, Union Health Secretary, Dr Balram Bhargava, DG ICMR, Dr. Manohar Agnani, AS (Health Ministry), Smt. Arti Ahuja, AS (Health Ministry), Shri Lav Agarwal, JS (Health Ministry), Dr. Sujeet Kumar Singh, Director (NCDC), Dr Randeep Guleria, Director, AIIMS New Delhi, and senior officials from states were present in the meeting.
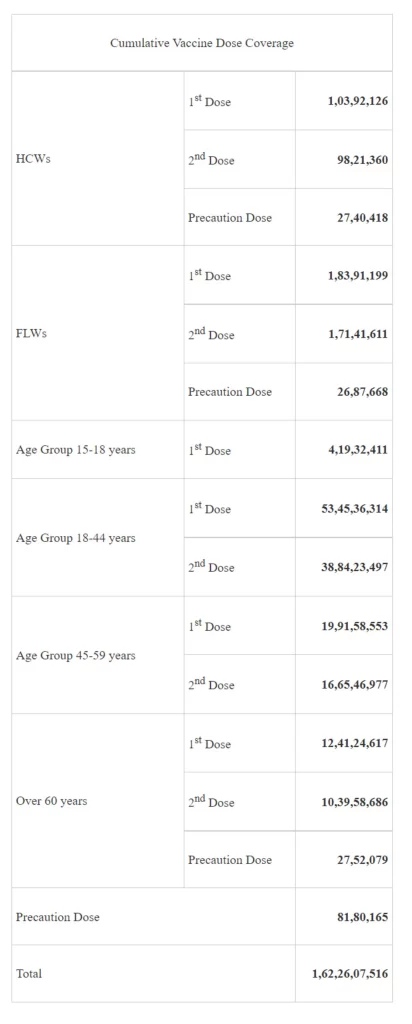
With the administration of more than 27 lakh Doses (27,56,364) vaccine doses in the last 24 hours, India’s COVID-19 vaccination coverage has exceeded 162.26 Cr (1,62,26,07,516) as per provisional reports till 7 am today.
This has been achieved through 1,75,24,670 sessions. The break-up of the cumulative figure as per the provisional report till 7 am today include:
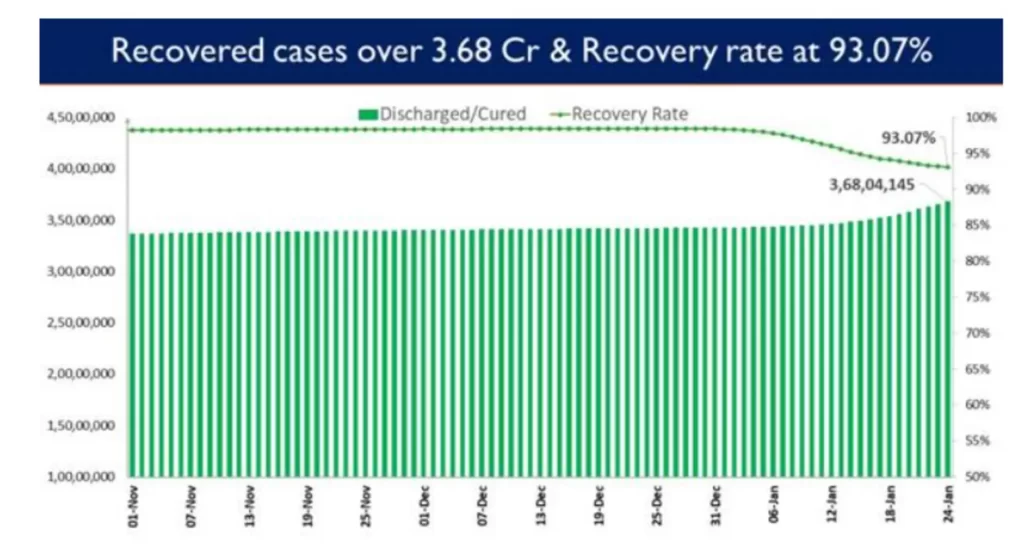
2,43,495 patients have recovered in the last 24 hours and the cumulative tally of recovered patients (since the beginning of the pandemic) is now at 3,68,04,145.
Consequently, India’s recovery rate stands at 93.07%.
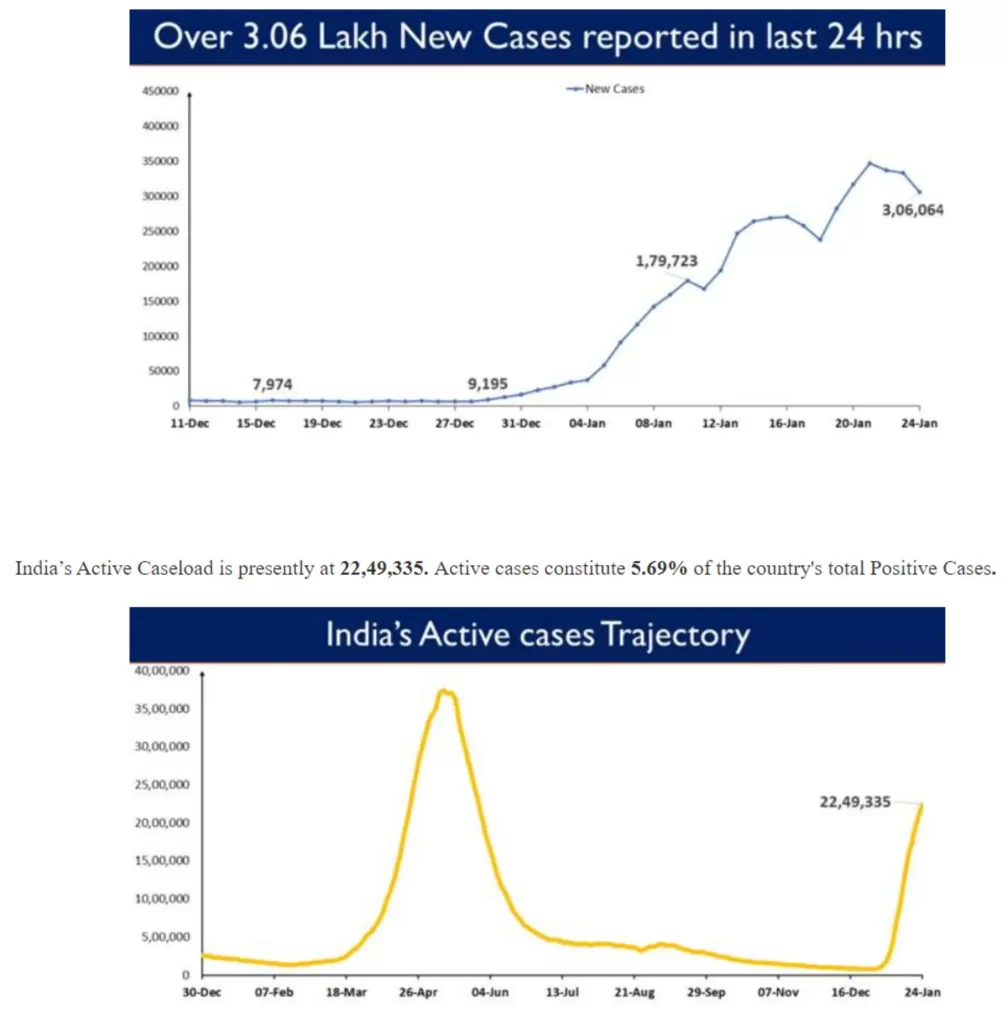
3,06,064 new cases were reported in the last 24 hours.
The testing capacity across the country continues to be expanded. The last 24 hours saw a total of 14,74,753 tests being conducted. India has so far conducted over 71.69 Cr (71,69,95,333) cumulative tests.
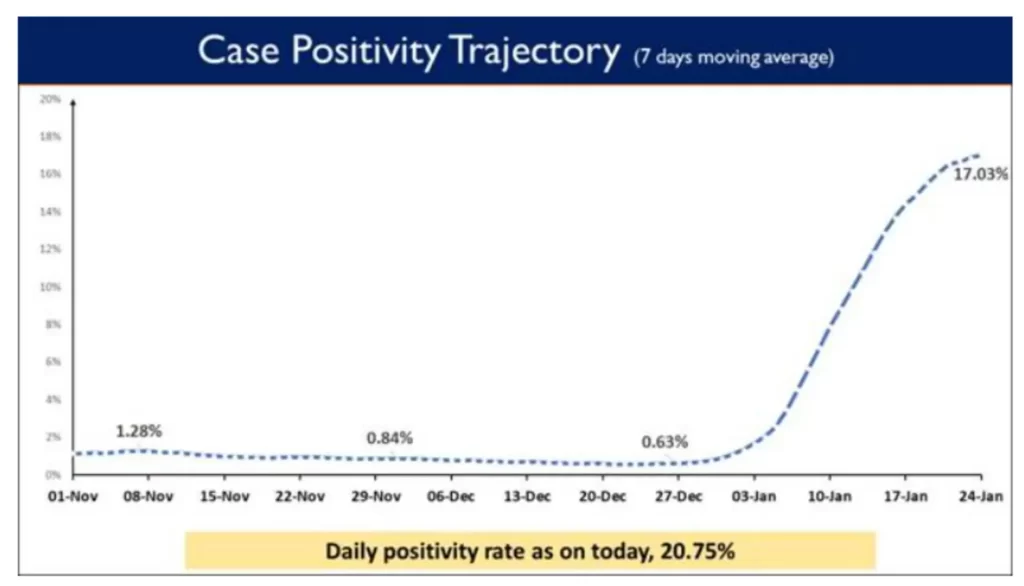
While testing capacity has been enhanced across the country, Weekly Positivity Rate in the country currently stands at 17.03% and the Daily Positivity rate also reported to be 20.75%.
160.43 cr vaccine doses have been administered so far under Nationwide Vaccination Drive
India's Active caseload currently stands at 20,18,825
Active cases stand at 5.23%
Recovery Rate currently at 93.50%
2,51,777 recoveries in the last 24 hours increases Total Recoveries to 3,60,58,806
3,47,254 new cases recorded in the last 24 hours
9,692 Total Omicron cases detected so far; an increase of 4.36% since yesterday
Daily positivity rate (17.94%)
Weekly Positivity Rate (16.56%)
71.15 cr Total Tests conducted so far; 19,35,912 tests conducted in the last 24 hours
