In continuation of updates on various utility features of Co-WIN for the beneficiaries, the following features are added in the self-registration portal of CoWIN.
a) Registration on Co-WIN –Instead of the existing limit of 4 members, now 6 members can be registered using one mobile number on Co-WIN.
b) Revoke Vaccination Status- A new utility feature has been introduced under raise an issue in Co-WIN account through which beneficiary can revoke its current vaccination status from fully vaccinated to partially vaccinated or unvaccinated status and also partially vaccinated to unvaccinated status. The vaccination status can be corrected by the beneficiaries, where in occasional isolated cases, the vaccination Certificates are generated due to inadvertent data entry errors by the vaccinator in updation of vaccination data of beneficiaries. The Changes may take 3-7 days after submitting the online request through raise an issue utility.Such beneficiaries can get their due vaccine dose, may be at nearest vaccination center, as per the existent standard guidelines once new vaccination status is successfully updated in the system.
151.94 cr vaccine doses have been administered so far under Nationwide Vaccination Drive
India's Active caseload currently stands at 7,23,619
Active cases stands at 2.03%
Recovery Rate currently at 96.62%
46,569 recoveries in the last 24 hours increases Total Recoveries to 3,45,00,172
1,79,723 new cases in the last 24 hours
Daily positivity rate (13.29%)
Weekly Positivity Rate (7.92%)
69.16 cr Total Tests conducted so far
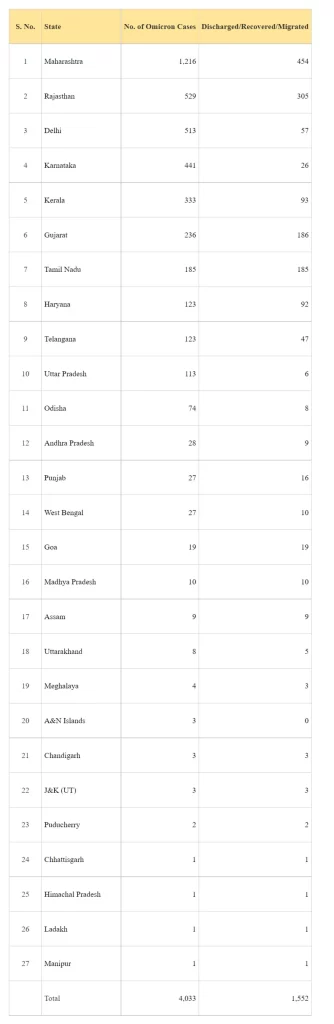
Statewise status of Omicron Variant
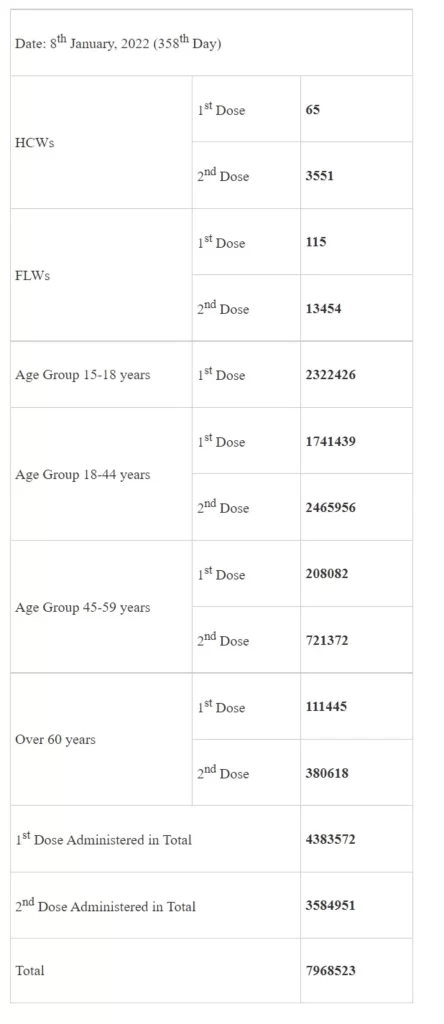
In a landmark achievement, more than 2 Cr (2,27,33,154) COVID-19 Vaccine Doses administered among adolescents for first dose between 15-18 age group. India’s COVID-19 vaccination coverage has crossed 151.47 Crore (1,51,47,41,090) today. More than 79 lakh (79,68,523) Vaccine Doses have been administered till 7 pm today. The daily vaccination tally is expected to increase with the compilation of the final reports for the day by late tonight.
In a tweet, Union Health Minister Dr. Mansukh Mandaviya praised the enthusiasm among Adolescents for Vaccination and congratulated them on this achievement.

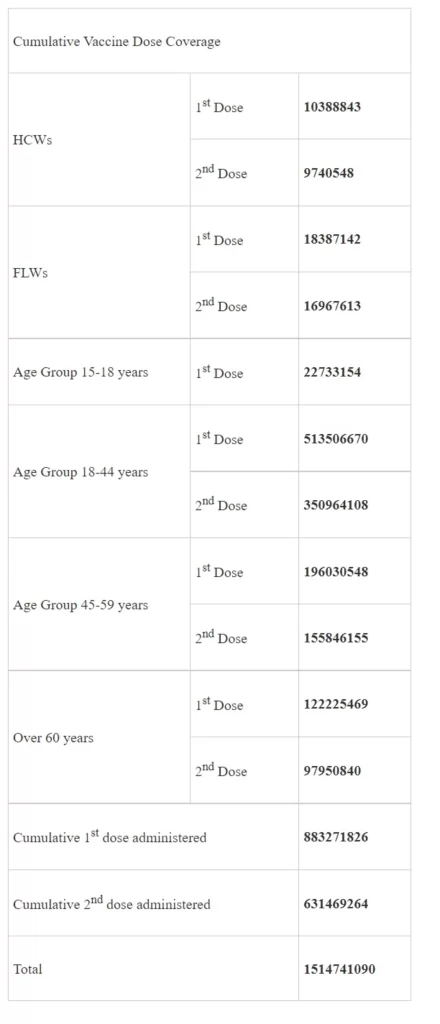
The cumulative coverage of vaccine doses, segregated based on population priority groups, is as follows:
Today’s achievement in the vaccination exercise, segregated by population priority groups, is as follows:
The vaccination exercise as a tool to protect the most vulnerable population groups in the country from COVID-19 continues to be regularly reviewed and monitored at the highest level.
Union Ministry of Health & Family Welfare has revised the Guidelines for International Arrival on 6th January 2022 in view of the changing nature of COVID -19 virus and evolution of SARS-CoV-2 variant of concern (VOCs) i.e. Omicron variant and reported increase in the cases worldwide. These guidelines shall be valid from 11th January 2022 (00.01 Hrs IST)
These were last updated on 30th November 2021.
The Revised Guidelines are posted on the Health Ministry’s website and can be accessed at-
https://www.mohfw.gov.in/pdf/RevisedGuidelinesforInternationalArrivalsdated7thJanuary2022.pdf
The following changes have been made in the Revised Guidelines of 6th January 2022 as compared to previous guidelines (of 30th November 2021):
All travellers (including those 2% who were selected for random testing on arrival and were found negative) will undergo home quarantine for 7 days and shall undertake RT-PCR test on the 8th day of arrival in India.
Travelers shall also be required to upload results of RT-PCR test for COVID-19 done on 8th day on Air Suvidha portal (to be monitored by the respective States/UTs).
All travellers who need to undertake testing on arrival, should preferably pre-book the test online on Air Suvidha Portal, to facilitate timely testing and avoid inconvenience.
Some media reports are suggesting that Union Ministry of Health & Family Welfare has in a meeting with the Election Commission of India (ECI) yesterday suggested that the “COVID situation in the country is nothing to be worried about” and “there is no cause of alarm or concern in the view of very few cases of Omicron in the poll bound states”. Such reports are highly ill- informed, misleading and far from the truth. These reports have a very high tendency to start a mis-information campaign in the midst of a pandemic.
The Union Health Secretary in his meeting with ECI presented the overall global and domestic status of spread of COVID, as well as Omicron, in the country. Details were also presented on status of preparedness of the public health response within the States for control and management of the increasing numbers of COVID cases. The focus of the presentation was on the 5 poll bound States and their neighbouring States.
Dr. Mansukh Mandaviya, Union Minister of Health and Family Welfare today virtually interacted with State Health Ministers and Principal Secretaries/ Additional Chief Secretaries of States/UTs to review the Public Health Preparedness to COVID19 and progress of National COVID19 vaccination campaign. The meeting was held in view of the rising cases of the Omicron variant and the recent decisions to roll-out vaccination for the age group 15-18 years and precautionary dose for identified vulnerable categories. The meeting was officiated by the Union Health Secretary Shri Rajesh Bhushan.
State Health Ministers who joined the meet included Shri S Pangnyu Phom (Nagaland), Shri N K Das (Odisha), Dr. Prabhuram Choudhary (MP), Shri Ma Subramanian (Tamil Nadu), Shri Keshab Mahanta (Assam), Shri Anil Vij (Haryana), Satyendar Jain (Delhi), Shri Alo Libang (Arunachal Pradesh), Banna Gupta (Jharkhand), Shri Mangal Pandey (Bihar), Shri T.S. Singh Deo (Chhattisgarh). Ms. Chandrima Bhattacharjee, MoS Health (West Bengal) and others.
The Union Health Minister noted at the very outset that globally, countries are experiencing 3-4 times the surge in Covid-19 cases in comparison to their earlier peaks. The Omicron variant being highly transmissible, a high surge in case can overwhelm the medical system. He therefore advised the States to leave no stone unturned in ramping up infrastructure to manage a high surge so that India escapes unscathed from this episode of COVID-19.
Dr. Mandaviya said in this regard that irrespective of the COVID variants, measures for preparedness and protection remain the same. He urged the states to re-invigorate their teams to work at the ground level and strengthen monitoring and containment mechanisms. This was followed by a comprehensive and detailed discussion on various aspects of COVID management including ramping up of hospital infrastructure; increased testing; stringent restrictive measures for breaking the chain of transmission; and stress on COVID Appropriate Behaviour among the masses. Critical bottlenecks in medical infrastructure were also discussed.
Applauding the dedication and patience shown by state administrations in their fight against the pandemic and in simultaneously ensuring the welfare of the people, Dr. Mansukh Mandaviya said, “We have put up a strong fight against Covid earlier and this learning must be used to re-focus on efforts against the Omicron variant.” He stressed on the continued need to follow COVID Appropriate Behaviour along with renewed and stringent focus on containment measures for addressing the present surge.

Underscoring the critical importance of vaccination drive, the Minister observed, “We must focus on planning with regard to the 15-18 age group vaccination and precautionary doses for eligible beneficiaries”. Dr Mansukh Mandaviya also urged states to ensure that the healthcare workers and frontline workers are fully vaccinated, as they form the vulnerable categories.
While appreciating the efforts of States/UTs in achieving the national average of 90% coverage of first dose vaccination of all eligible adults, he urged those states, whose progress of vaccination is below the national average, to ramp up their vaccination campaign. States were also advised to prepare a weekly plan to catch up & exceed the average national vaccination coverage and review the implementation of this plan on a daily basis at the level of Secretary/ACS Health [with special emphasis in 5 poll-bound States of Punjab, Goa, Uttarakhand, Uttar Pradesh and Manipur]
Stressing on the need to ensure smooth implementation of new vaccination guidelines, States/UTs were advised to ensure the orientation of vaccinators and vaccination team members for vaccination of 15-18 years age-group and the identification of dedicated session sites for vaccination of 15-18 years age-group. To avoid the mixing-up of vaccines during administration, separate CVCs, separate session sites, separate queue (if at same session where adult vaccination is ongoing) and separate vaccination team (if at same session site) are to be strived for. States/UTs were also advised to share their requirement of vaccine doses through district wise estimation of beneficiaries using Co-WIN. They were urged to plan for distribution of Covaxin to identified session sites well in advance and publish sessions for at least 15 days to provide sufficient visibility.

Dr Mansukh Mandaviya also pointed out that various measures are being taken by the Union Government to ramp up infrastructure development and vaccine production in the country. He requested states/UTs to share their best practices so that the entire country can benefit.
The Union Health Minister brought attention of the States/UTs to the fact that collectively, they have only used just over 17% of the available approved funds under Emergency COVID Response Package (ECRP-II). States/UTs were exhorted to expedite the physical progress under ECRP-II in terms of ICU beds, oxygen beds, Paediatric ICU/HDU beds etc. States/UTs were also urged to effectively use IT tools for tele-medicine and tele-consultation, including training and capacity building of human resources, timely availability of ambulances, readiness of states to operationalise COVID facilities for institutional quarantining, and effective and supervised monitoring of those in home isolation. At the same time, he also urged states/UTs to better utilize the approved funds under ECRP-II and invited any suggestions regarding the same.
Shri Vikas Sheel, Addl. Secretary and MD, NHM spoke on the status of infrastructure development and utilisation of ECRP II funds by states through a detailed presentation. Dr. Manohar Agnani, Addl. Secretary (Health) presented the update on the status of vaccination in the country. Shri Lav Agarwal, Joint Secretary, Health Ministry presented a granular analysis of the COVID trajectory in the States and suggested the ramping up of testing, surveillance and containment mechanisms to address the recent surge in cases. Principal Secretary (Health), Additional Chief Secretary (Health) and State Surveillance Officer of the concerned states/UTs offered their feedback and suggestions while discussing their points of concern.
With a view to attain self-reliance and reduce import dependence in critical APIs, a scheme called “Production Linked Incentive (PLI) Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) In India” was approved by the Government. The total outlay of the scheme is Rs.6,940 crore and financial incentive under the scheme is provided to the selected participants on incremental sales of 41 identified products in four different Target Segments for a period of six years.
Another Scheme called “Production Linked Incentive Scheme for Pharmaceuticals” was approved by the Government of India in March, 2021 to enhance India’s manufacturing capabilities by increasing investment and production in the sector and contributing to product diversification to high value goods in the pharmaceutical sector. The total outlay of the scheme is Rs. 15,000 croreand financial incentives is provided to the selected participants on incremental sales for a period of six years.
Under the PLI Scheme for Bulk Drugs, 50 applicants have been approved. Whereas under the PLI Scheme for Pharmaceuticals, 55 applicants have been approved. The details of approved applicants under both the Schemes are available at the website of the Department, viz., pharmaceuticals.gov.in.
The information was given by the Union Minister of Chemicals and Fertilizers, Dr. Mansukh Mandaviya in a written reply in the Rajya Sabha today.
Under National COVID-19 Vaccination Programme, COVID-19 vaccine is available free of cost for all citizens aged 18 years and above irrespective of their socio-economic status at all Government COVID-19 Vaccination Centres (CVCs). There is provision to facilitate vaccination of persons without prescribed identity cards such as migrants, seers, nomads, refugees, destitute, homeless people, beggars etc.through special sessions where 100% vaccination is done through on-site registration by the vaccinator under a prescribed procedure.
Government of India has issued advisory to all States/UTs for Near-to-Home COVID-19 Vaccination Centre (NHCVC) for elderly and differently abled citizens and for vaccination at the place of residence of persons who might be bed-ridden or have extremely restricted mobility or disability and/or special needs that may hamper their accessibility even to NHCVCs. From 3rd November 2021 ‘Har Ghar Dastak’ Teekakaran Abhiyan has been launched wherein missed beneficiaries for 1st dose and due beneficiaries for 2nd dose are identified and vaccinated through house-to-house activity.
Union Ministry of Health and Family Welfare has issued the national policy for admission of COVID patients to various categories of COVID facilities with the aim to ensure prompt, effective and comprehensive treatment of patients suffering from COVID-19.
Union Ministry of Health & Family Welfare has been in regular interaction with the States/UTs to seek continued support in terms of strict and persistent implementation of 5-fold strategy (Test-Track-Treat-Covid Appropriate Behaviour-Vaccinate) against COVID-19.
Government of India has also significantly increased the testing capacity for Covid-19 related tests. As on 14th December 2021, more than 3,000 operational laboratories are conducting Covid tests and reporting to Indian Council of Medical Research (ICMR) on RT-PCR, TruNat, CBNAAT and Other Molecular Nucleic Acid Tests for COVID-19.
Under PM-Ayushman Bharat Health Infrastructure Mission (PM-ABHIM) setting up of integratedpublic health laboratories in all districts and strengthening block public health unitsin 11 high focus states has been provisioned.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
Under the National Programme for Prevention and Control of Cancer, Diabetes, Cardiovascular Diseases and Stroke (NPCDCS) launched in 2010 as part of National Health Mission (NHM), the Union Ministry of Health & Family Welfare provides technical and financial support to the States/UTs based on the proposals received from the States/UTs and subject to the resource envelope. The programme focuses on strengthening infrastructure, human resource development, health promotion & awareness generation for prevention, early diagnosis, management and referral to an appropriate level of healthcare facility for treatment of the Non-Communicable Diseases (NCDs).
The total fund provided to States/UTs, in NPCDCS Program under Flexible Pool for Non-Communicable Diseases programme (NCD) under NHM during 2021-22 is Rs. 56118.07 Lakhs.
Under NPCDCS, 677 NCD clinics at District level, 187 District Cardiac Care Units, 266 District Day Care Centres and 5392 NCD clinics at Community Health Centre level has been set up to ensure the treatment of common NCDs.
A population-based initiative for prevention, control and screening for common Non-Communicable Diseases (NCDs) i.e. diabetes, hypertension and common cancers has been rolled out in the country under NHM and also as a part of Comprehensive Primary Health Care. Under the initiative, persons more than 30 years of age are targeted for their screening for the common NCDs. Screening of these common NCDs is an integral part of service delivery under Ayushman Bharat – Health and Wellness Centres.
Preventive aspect of Cancer is strengthened under Comprehensive Primary Health Care through Ayushman Bharat Health Wellness Centre scheme, by promotion of wellness activities and targeted communication at the community level. Other initiatives for increasing public awareness about Cancer and for promotion of healthy lifestyle includes observation of National Cancer Awareness Day, World Cancer Day and use of print, electronic and social media for continued community awareness. Furthermore, healthy eating is also promoted through FSSAI. Fit India movement is implemented by Ministry of Youth Affairs and Sports, and various Yoga related activities are carried out by Ministry of AYUSH. In addition, NPCDCS gives financial support under NHM for awareness generation (IEC) activities for Cancer to be undertaken by the States/UTs as per their Programme Implementation Plans (PIPs).
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
ChAdOx1 nCoV- 19 Corona Virus Vaccine (Recombinant) (COVISHIELD) is manufactured by M/s Serum Institute of India Pvt., Ltd., Pune, while the Whole Virion Inactivated Corona Virus Vaccine (COVAXIN) is manufactured by M/s Bharat Biotech International Limited, Hyderabad.
As communicated by the M/s Serum Institute of India, the current monthly vaccine production capacity of Covishield is approx. 250-275 Million doses per month.
Further, as communicated by M/s Bharat Biotech International Limited, Hyderabad, the current monthly vaccine production capacity of Covaxin is approx. 50-60 Million doses/month. Both companies have achieved close to 90% of present production capacity.
Moreover, as per the provisions of New Drugs and Clinical Trials Rules, 2019 under Drugs and Cosmetics Act, 1940 and in light of urgent need due to COVID pandemic in the country, CDSCO has granted permissions to following COVID-19 vaccines other than COVAXIN & COVISHIELD for prevention of COVID-19 for restricted use in emergency situation:
Gam-COVID-Vac Combined vector vaccine [SPUTNIK-V] manufactured by M/s Ra (biologicals), Panacea Biotec Ltd., New Delhi using imported Ready to Fill (RTF) bulk from M/s Generium JSC, Russia on 02.07.2021.
Novel Corona Virus 2019-nCoV vaccine [ZyCoV-D] manufactured by M/s Cadila Healthcare Limited, Ahmedabad on 20.08.2021. Ad26.COV2-S (recombinant) COVID-19 Vaccine manufactured by M/s Biological E limited, Hyderabad using imported bulk of M/s Johnson & Johnson Pvt. Ltd on 18.08.2021.
Gam-COVID-Vac Combined vector vaccine [Sputnik–V] manufactured under technology transfer from M/s RDIF, Russia by M/s Hetero Biopharma Limited, Hyderabad on 07.10.2021.
Gam-COVID-Vac Combined vector vaccine [SPUTNIK-V] to M/s Dr. Reddy’s Laboratories Ltd, Hyderabad on 12.04.2021.
mRNA-1273 COVID-19 vaccine (Moderna) to M/s Cipla Limited, Mumbai on 29.06.2021. Ad26.COV2-S (recombinant) COVID-19 Vaccine to M/s Johnson & Johnson Pvt. Ltd., Mumbai on 07.08.2021.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
