Ayushman Bharat-Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) is an entitlement-based scheme. No enrolment or registration of beneficiary is required to avail benefits under the scheme. The beneficiary may directly visit the empanelled hospital (public or private) to avail cashless treatment benefits. The beneficiary may use the following channels to check his/her eligibility under the scheme:
NHA has a 24x7 call centre 14555, which is widelypublished.
Beneficiary can reach out to any government hospital (CHC and above) for beneficiary verification. After verification at the government hospital, the beneficiary can be provided necessary treatment or can be directed to nearby other public or empanelled private hospital.
Beneficiary can check his/her eligibility and also search for the nearest empanelled hospital through PMJAY Mobile App.
The beneficiary can visit mera.PMJAY.gov.in to check his/her eligibility under AB-PMJAY.
All empanelled hospitals (public orprivate) have Pradhan Mantri Arogya Mitra who facilitate the beneficiary with all the necessary steps required to avail scheme benefits.
National Health Authority -the implementing agency of AB-PMJAY has issued a comprehensive set of anti-fraud guidelines. Regular anti-fraud advisories are issued to States/UTs. National Anti-Fraud Unit (NAFU) is created at NHA for overall monitoring and implementation of anti-fraud framework supported by State Anti-Fraud Units (SAFUs) at State level. All claims require mandatory supporting documents along-with on-bed patient photo before approval and payment. The feature of Aadhar-based biometric verification of beneficiary at the time of admission and discharge is launched at all private hospitals. Use of artificial intelligence and machine learning is made for a comprehensive fraud analytics solution to detect fraud pro-actively, develop algorithms that can be used on large volume of data to identify suspect transactions and entities and risk scoring of hospitals and claims on continuously.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
The five-fold strategy of Test-Track-Treat-Vaccinate-adherence to COVID Appropriate Behaviour (CAB) was highlighted as the mainstay of public health response strategy for effective and timely control and management of COVID19 and its variants by Union Health Secretary Shri Rajesh Bhushan as he reviewed the public health preparedness to the Omicron variant of COVID-19 and progress of vaccination with Health Secretaries and NHM MDs of States and UTs, through video conference (VC) today. Dr Balram Bhargava, DG (ICMR) and Secy (DHR) as well as Dr Rajesh Gokhale, Secretary, DBT were also present.
States and UTs were urged to enhance testing and focus on surveillance to ensure early identification of the suspected cases for their prompt isolation and further clinical management. They were advised to ensure availability of PT-PCR testing in all districts. Districts reporting higher case positivity were alerted to monitor the case trajectory on a regular basis, and to promptly dispatch positive samples to the mapped INSACOG (Indian SARS-CoV-2 Genomics Consortium) labs for whole genome sequencing. Close monitoring of new emerging hotspots/clusters, breakthrough and re-infection cases, and prompt investigation of such events by the Rapid Response Teams was discussed. It was re-emphasised that all contacts for positive patients are to be traced and tested as per protocol as quickly as possible.
Based on the information of international travelers from ‘at risk’ countries from Air Suvidha portal, district-wise monitoring of international passengers who have tested negative on arrival with focus on symptomatic cases, was highlighted. In view of the forthcoming winter season, they were advised that prevalence of influenza-like illness (ILI) / severe acute respiratory infection (SARI) and respiratory distress symptoms needs to be closely monitored. Review of monitoring mechanism for effective home isolation was stressed.
In order to ensure that all hospital infrastructure is ready for any possible surge in cases, States/UTs were advised to review their readiness for providing quality medical care. They need to ensure that funds released by Government of India under ECRP-II are being utilised for creating and strengthening health infrastructure, and 100% matching funds are promptly released by States to the State Health Societies. As the progress of the funds utilisation at the field level is being reviewed by the Health Ministry, information on progress of funds utilisation needs to be filled in the portal created for the purpose. It needs to be ensured that al health facilities at the field level have functional ventilators, PSA plants, oxygen concentrators etc. States were informed that many ventilators supplied by the Centre are still lying unpacked and unused at some field hospitals, with consumables not being procured & made available by the States. This needs to be reviewed immediately to ensure that all PSA Oxygen plants, Oxygen concentrators and ventilators are installed and functioning.
The Health Secretary also urged the States to ensure to maintain adequate buffer stock for the eight critical drugs identified in clinical treatment of COVID-19; the Guidelines for these were shared with States in July 2021.
Underscoring the critical importance of vaccination drive, States were advised to continue increasing the pace and coverage of COVID19 nationwide vaccination drive with focus on the ongoing ‘Har GharDastak’ campaign for ensuring full vaccination of all eligible population, with regular monitoring at the village and district levels. In order to keep rumours at bay and address issues of vaccine hesitancy, States/UTs suggested to organise evidence-informed awareness campaigns with regular media briefings.
The Medical Device Sector in India suffers from a considerable cost of manufacturing disability vis-à-vis competing economies, inter alia, on account of lack of adequate infrastructure, domestic supply chain and logistics, high cost of finance, inadequate availability of power, limited design capabilities, low focus on research & development (R&D) and skill development, etc.
With an objective to boost domestic manufacturing, attract large investment in the Medical Device Sector, the Department of Pharmaceuticals had launched a Production Linked Incentive (PLI) Scheme for Promotion of Domestic Manufacturing of Medical Devices to ensure a level playing field for the domestic manufacturers of medical devices with a total financial outlay of Rs.3,420 cr. for the period 2020-21 to 2027-28.
In the first round, 28 applications were received from 23 applicants under the Scheme. Out of which, 13 applications with committed investment of Rs.798.93 crore by the Companies were approved with expected employment generation of around 3,812 persons.
Since few slots remained unfilled for certain products, applications were invited again with the last date of submission as 31.08.2021. Total 14 applications were received.
In the 9th Meeting of the Empowered Committee held on 25.11.2021, after considering all the applications as per the scheme guidelines, applications of the following applicants have been approved:
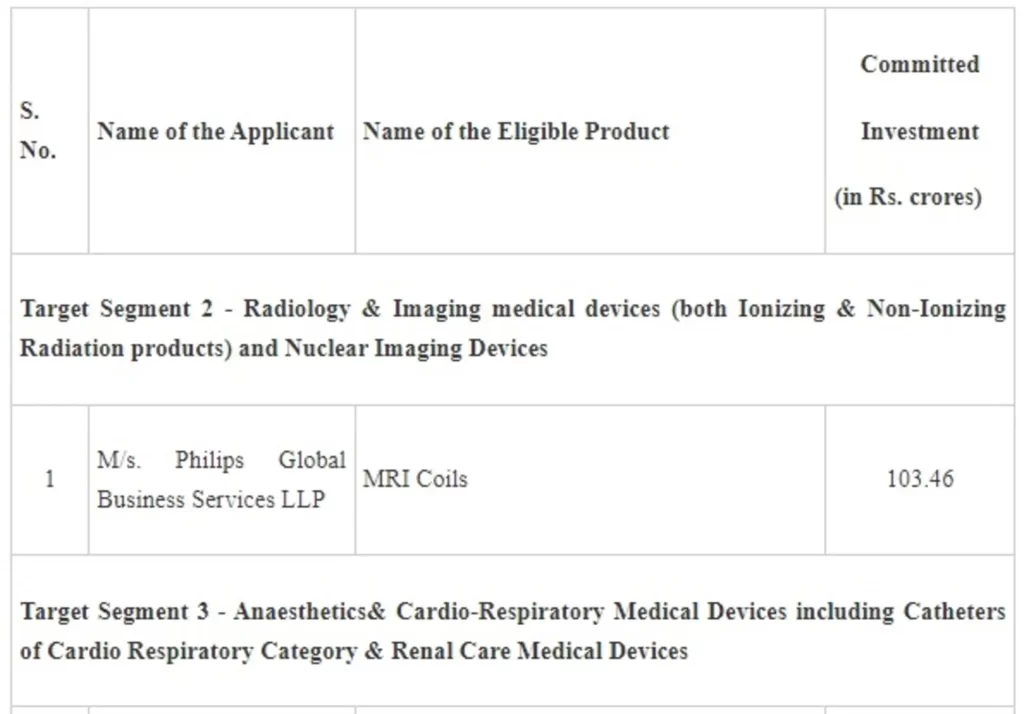
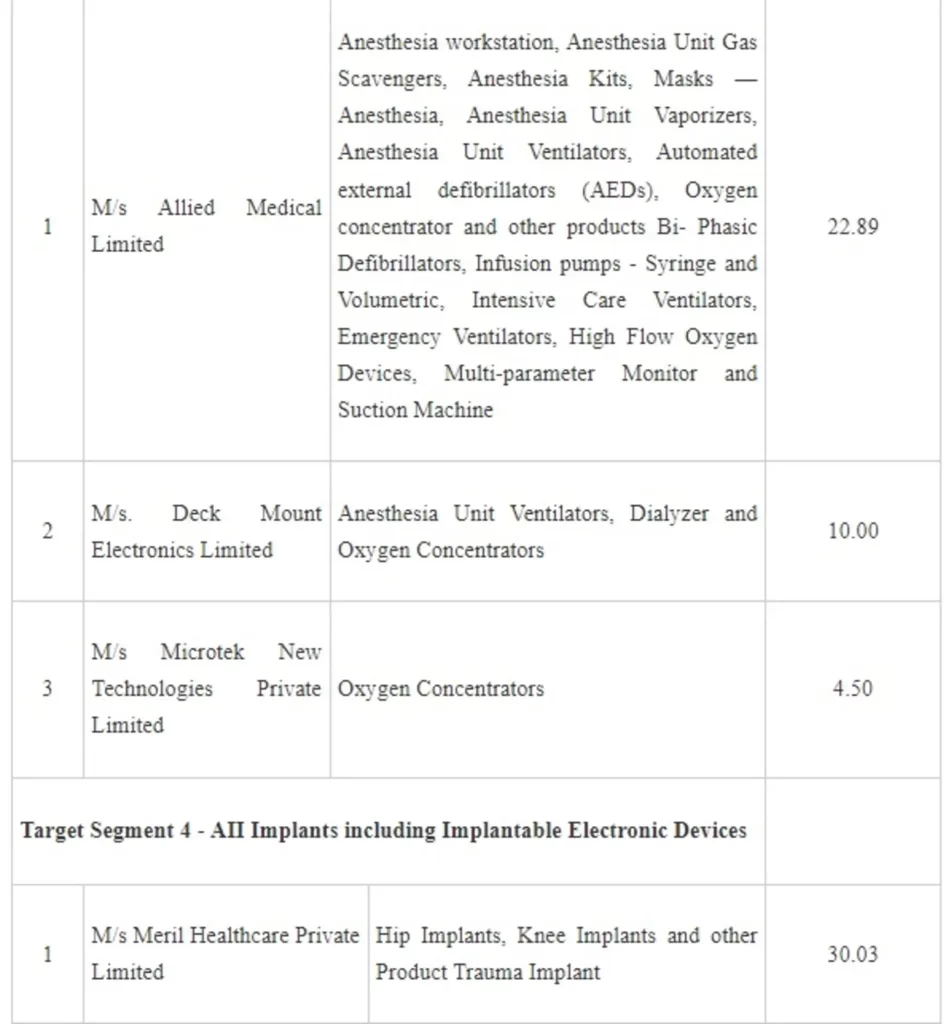
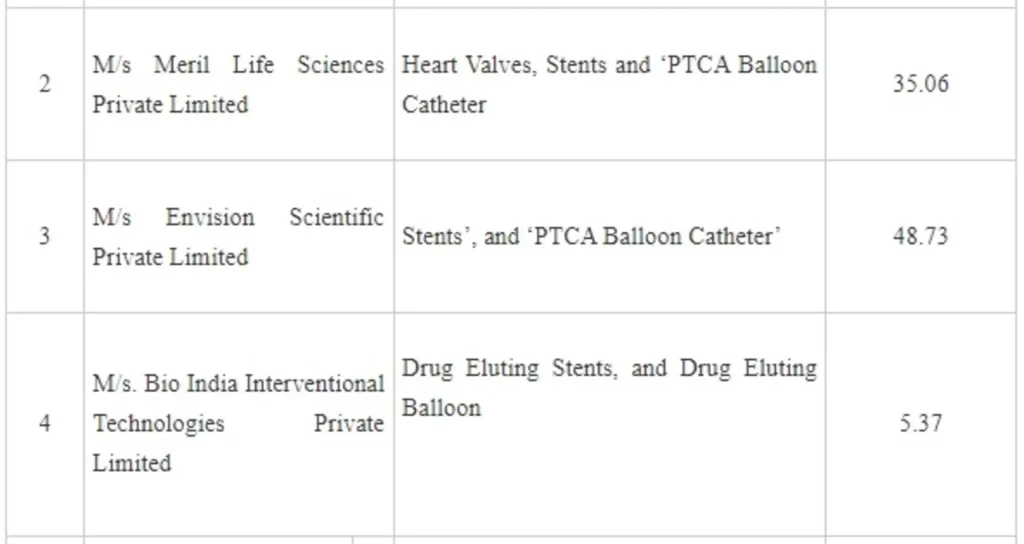
The setting up of these 8 plants will lead to a total committed investment of Rs. 260.40 crore by the companies and employment generation of about 2,599. The commercial production is projected to commence from 1st April, 2023 onwards. With this the total applications approved under both rounds aggregate to 21 applicants with committed investment of Rs 1,059.33 crore and employment generation of about 6,411. The setting of these plants will make the country self-reliant to a large extent in the specified target segments in the Medical Devices Sector.
The Indian pharmaceutical industry is the 3rd largest in the world by volume. It has high market presence in several advanced economies such as the US and EU. The industry is well known for its production of affordable medicines, particularly in the generics space. However, the country is significantly dependent on the import of basic raw materials, viz., Bulk Drugs that are used to produce medicines. In some specific bulk drugs, the import dependence is 80 to 100%.
With an objective to attain self-reliance and reduce import dependence in these critical Bulk Drugs - Key Starting Materials (KSMs)/ Drug Intermediates and Active Pharmaceutical Ingredients (APIs) in the country, the Department of Pharmaceuticals had launched a Production Linked Incentive (PLI) Scheme for promotion of their domestic manufacturing by setting up greenfield plants with minimum domestic value addition in four different Target Segments (In Two Fermentation based - at least 90% and in the Two Chemical Synthesis based – at least 70% ) for 41 products with a total outlay of Rs. 6,940 cr. for the period 2020-21 to 2029-30.
In total, 215 applications were received for the 36 products spread across the 4 Target Segments. Out of which, total 42 applications were approved by the Government with a total Committed Investment of Rs. 4,347.26 Crore and expected Employment Generation of around 8,792 persons.
Since few slots remained unfilled for certain products, applications were invited again with the last date of submission as 31.08.2021. Total 24 applications were received.
In the 9th Meeting of the Empowered Committee held on 25.11.2021, after considering all the applications received, the applications of following companies, which fulfill the prescribed criteria laid down in scheme guidelines have been approved, as under:
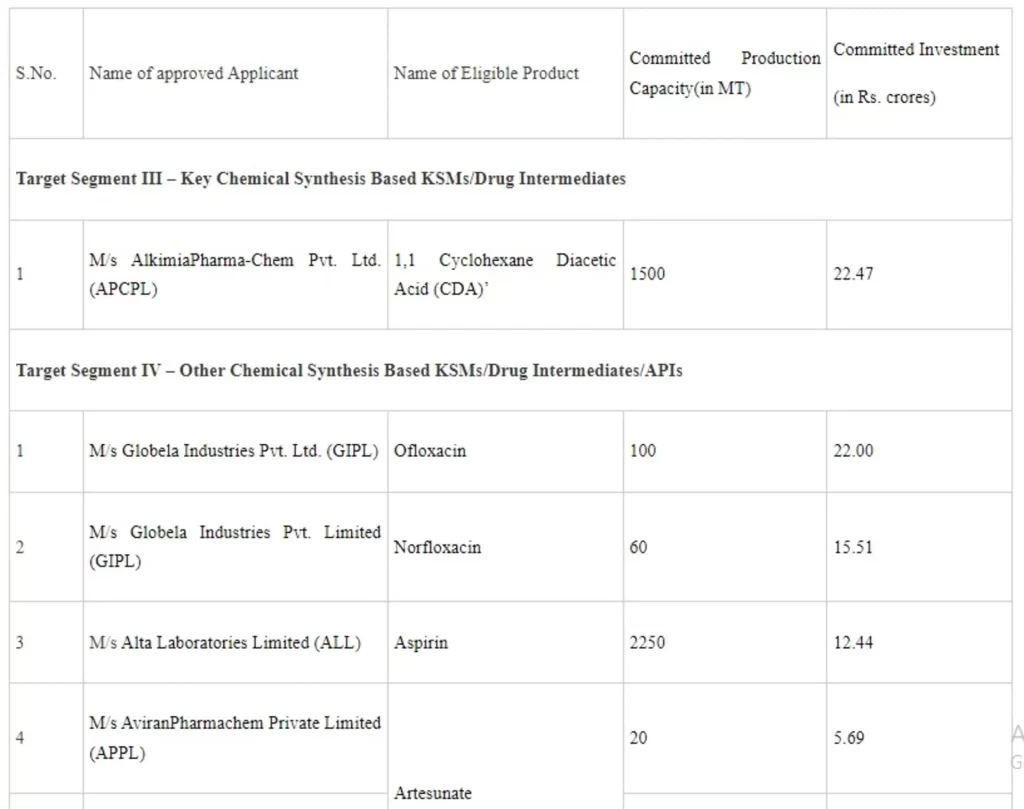

The setting up of these 8 plants will lead to total committed investment of about Rs.151.12 crore by the companies and employment generation of about 1,951. The commercial production of these plants is projected to commence from 1st April, 2023 onwards. With this, the total applications approved under both rounds aggregate to 50 applicants with committed investment of Rs 4,498.38 crore and employment generation of about 10,743. Setting up of these plants will make the country self-reliant to a large extent in respect of these Bulk drugs.
As per information provided by the Central Drugs Standard Control Organization (CDSCO) under the Ministry of Health & Family Welfare, the Organization regulates safety, efficacy and quality of the drugs, medical devices and Cosmetics as per the provisions of Drugs and Cosmetics Act 1940 & Rules made thereunder. CDSCO and Ministry of Health & Family Welfare from time to time have taken various measures for ensuring the quality of drugs in the country, such as:
Drugs and Cosmetics Act, 1940 was amended in the year 2008 to provide stringent penalties for manufacturer of spurious and adulterated drugs and certain offences have also been made cognizable and non-bailable.
States/UTs were requested to set up Special Courts for trial of offences under the Drugs and Cosmetics Act for speedy disposal. So far, 33 states have already set up designated Special Courts.
Guidelines for taking action on samples of drugs declared as spurious or ‘not of standard quality’ in the light of enhanced penalties under the Drugs & Cosmetics (Amendment) Act, 2008 were forwarded to the State Drugs Controllers for uniform implementation.
Number of sanctioned posts in CDSCO have been increased from 111 in 2008 to 492 in January, 2021.
Testing capacities of Central Drugs Testing Laboratories under CDSCO are being constantly strengthened to expedite testing of drug samples in the country.
In order to ensure efficacy of drugs, the Drugs and Cosmetics Rules 1945 have been amended in April, 2017 providing that applicant shall submit the result of bioequivalence study along with the application for grant of manufacturing License of oral dosage form of drugs falling under the Category II and Category IV of the Biopharmaceutical Classification System.
Drugs and Cosmetics Rules 1945 have been amended in October, 2017 making it mandatory that before the grant of manufacturing license, the manufacturing establishment is to be the inspected jointly by Drugs Inspectors of the Central Government and the State Government concerned. Further, the licensed manufacturing premises are to be inspected jointly by the Drugs Inspectors of Central Government and State Government concerned to verify the compliance with the conditions of license and the provisions of the Drugs & Cosmetics Act and Rules for not less than once in three years or as needed as per risk based approach.
Drugs and Cosmetics Rules 1945 have been amended in April, 2018 making it mandatory that the applicants for all drugs shall submit evidence of stability, safety of excipients etc. to the State Licensing Authority before grant of product manufacturing license by the Authority.
Government has approved a proposal for strengthening the drug regulatory system in the country, both at the level of Central and the State Governments at a total expenditure of Rs. 1,750 crores.
The information was given by the Union Minister of Chemicals and Fertilizers, Shri Mansukh Mandaviya in a written reply in the Lok Sabha today.
The Department of Health and Family Welfare has launcheda Digital Portal for Crowd funding and Voluntary donations for Patients of Rare Diseases in accordance with the mandate of the National Policy for Rare Diseases, 2021. The Digital Portal may be accessed through https://rarediseases.nhp.gov.in/
(c): ICMR has initiated a National Registry wherein epidemiological data is being collected for rare diseases and other inherited disorders. Data of a total of 4001 rare diseases cases (Storage disorders, Small meta bone inborn error of metabolism, primary immune deficiency disorders, skeletal dysplasia, neuro muscular disorders, hemoglobinopathies and bleeding disorders) have been collected till 31st October, 2021.
National Policy for Rare Diseases, 2021 (the Policy) has been finalized and put in public domain. The Policy can be accessed at website:-https://main.mohfw.gov.in/documents/policy
On the basis of provisions envisaged under National Policy for Rare Diseases, 2021, with regard to providing financial assistance to the patients suffering from rare diseases, it is submitted that for the diseases/ disorders amenable to one time curative treatment (listed under Group -1), financial support will be provided under umbrella scheme of RAN i.e. an amount up toRs. 20 lakh only for entitled beneficiaries shall be provided by the Central Government. The diseases requiring long term / lifelong treatment having relatively lower cost of treatment (listed under Group 2), the financial support is envisaged to be provided by the State Governments. The diseases for which definite treatment is available but involves very high cost and lifelong therapy (listed under Group 3), the policy provides assistance to the patients by creation of Digital Platform for inviting voluntary donations from individuals and corporate donors. Donors will have a choice to make donations to different Centres of Excellence (CoEs) and for the patients’ treatment by these CoEs. Funds are to be utilized in a decentralised manner, i.e. every Centre of Excellence (CoE) will have its own Rare Disease Fund which is to be utilized with the approval of in charge concerned.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Lok Sabha today.
Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar addressed International Symposium on Health Technology Assessment, was organised by the Department of Health Research, Ministry of Health and Family Welfare in collaboration with International Decision Support Initiative (iDSI). The theme of the event was “Translating Knowledge and Best Practices into Policy for Evidence Informed Decision making in Healthcare Sector for Universal Health Coverage”. The symposium was attended by about 500 participants, virtually or in person.
Dr. Bharati Pravin Pawar remarked in her key note address that the symposium has provided a platform to discuss the global best practices in Health Technology Assessment, development of a sustainable model of evidence-based decision making through HTA institutionalization in India in order to achieve universal health coverage. Dr. Bharti Pravin Pawar said, “HTA is bridging the gap between the researchers and the real world.”
The Minister of State, Dr Bharati Pravin Pawar also released video on “The Power of HTA” and two books – “Policy Briefs” and “Development of Health-Related Quality of Life Value Sets (EQ-5D-5L) for India”. The Policy Brief book contains all the policy briefs of the HTA studies conducted by HTAIn and approved by the Board till date. The aim of EQ5D5L study was to generate the Indian Tariff Values of all the health states that is used in the HTA studies. It is the first and the largest study of its kind in South Asia conducted by HTAIn in collaboration with its Resource Centers.
Dr V. K. Paul, Member NITI Aayog quoted that HTA is the pillar for policy formation. Later on, Dr Balram Bhargava, Secretary-DHR and Director General-ICMR stressed on the need for institutionalizing HTA in the country. Ms. Anu Nagar Joint Secretary, DHR detailed about the journey of HTA in India.
Dr. Somsak Chunharas, Advisor to the Health Intervention and Technology Assessment Program (HITAP) and Former Deputy Minister, Public Health, Thailand; Dr. Javier Guzman, Director, Global Health Policy & Senior Policy Fellow, Centre for Global Development; Sir Andrew Dillon Chair, Life Sciences Advisory Board and Founding Chief Executive, NICE, UK; Prof. Carole Longson Life Science Adviser NICE, UK; Prof. Stephen Jan Co-Director, Health Systems Science, George Institute for Global Health, Dr. Tessa-Tan-Torres Edejer Coordinator, Unit on Costs, Effectiveness, Expenditure & Priority Setting, WHO Geneva were present among the eminent speakers.
Health being a State subject, it is the responsibility of the respective State/Union Territory (UT) Government to take cognizance of complaints against private hospitals including for charging excess fees for the same procedures from the patients paying in cash as compared to those who pay through cashless health insurance schemes and to take suitable action to prevent and control such practices. Therefore, complaints, if any, are forwarded to State/UT Governments for taking appropriate action.
Government of India has enacted the Clinical Establishments (Registration and Regulation) Act, 2010 (CE Act, 2010) and notified Clinical Establishments (Central Government) Rules, 2012, as amended in 2018 and 2020thereunder, which prescribe conditions for registration and regulation of clinical establishments (both Government and Private) in the country. As per the Act, among other conditions the clinical establishments in the States/UTs which have adopted the Act, are required to display the rates charged for each type of service provided and facilities available in the local as well as in English languageat a conspicuous placefor the benefit of the patients. Further, as per the Act, the clinical establishment shall charge the rates for each type of procedure and service within the range of rates determined and issued by the Central Government in consultation with the State Government from time to time.
Towards this the Central Government has shared a standard list of medical procedures and standard template for costing with the States/UTs where the Act is applicable, for necessary action by them. The Act further provides for a registering authority in each district, which is empowered to conduct inquiry in case of violation of the provisions of the Act and take necessary action including imposing penalties and cancellation of registration, if applicable.
Further as per the Act, the Central/State Government issues Standard Treatment Guidelines (STG) from time to time which also facilitate rationalisation of cost of treatment. So, far STG for 227 medical conditions belonging to 21 clinical specialties/super specialties in allopathy and 18 medical conditions in Ayurveda have been issued by Ministry of Health and Family Welfare, Government of India.
The implementation and monitoring of the CE Act is within the remit of the respective State Government/UT Administration. As on date the CE Act has been adopted by 11 States and 6 UTs. Another 17 States and 1 UT have their own Act.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Lok Sabha.
The Government has taken several measures to enable training and capacity building in COVID-19. Training, including virtual training, has been provided to doctors, nurses and allied healthcare professionals and volunteers regarding the relevant aspects of COVID-19. The Government of India has utilized the iGOT (Integrated Government Online Training) platform to train various personnel. Since the beginning of COVID-19, close to 14 lakh unique users across the country have registered on this platform, recording enrollment of 29.29 lakh for different course. Additionally, more than 80 lakh health workforces have been trained in COVID related topics through the State Governments. Webinars and tutorials on various COVID related subjects were also uploaded by the Ministry of Health and Family Welfare and reputed institutes like AIIMS (New Delhi), NIMHANS, PGIMER, JIPMER, etc. that recorded a viewership of 2.23 crore.
As informed by National Medical Commission (NMC), a competency-based module was introduced by the erstwhile Board of Governors,Medical Council of India (BoG-MCI) on management of pandemics in the MBBS curriculum in August, 2020.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
National Expert Group on Vaccine Administration for Covid (NEGVAC) and National Technical Advisory Group on Immunization (NTAGI) are deliberating and considering scientific evidences related to vaccination of beneficiaries aged less than 18 years.
Government of India has been supplying Covid-19 vaccines free of cost to the States/UTs for administration to prioritized beneficiaries as recommended by NEGVAC.
ZyCoV-D vaccine manufactured by M/s Cadila Healthcare has received the approval for Restricted Use in Emergency Setting by the National Regulator i.e., Drug Controller General of India (DCGI) for the age group of 12 years and above based on the interim clinical data of Phase II &Phase III clinical trial conducted in the country.
Following COVID-19 vaccines are under clinical trials in the country for age-group of less than 18 years:
i. M/s Bharat Biotech is conducting Phase II/III clinical trial of COVAXIN on Healthy Volunteers aged 2 to 18 years & firm has submitted interim safety & immunogenicity data to the National Regulator.
ii. M/s Serum institute of India is conducting Phase II/III clinical trial of Nanoparticle Vaccine (Liquid) (COVOVAX) in 920 subjects of >2 to 17 years age group.
iii. M/s Biological E Ltd., is conducting Phase II/III clinical trial of RBD of SARS-CoV-2 gene in 624 subjects of ≥5 to less than 18years age group.
iv. M/s Johnson & Johnson Pvt. Ltd., is conducting Phase II/III clinical trial of Ad.26COV.2S vaccine in age group of 12-17 years (Global Clinical trial wherein India is one of the clinical trial sites).
The approval of aforementioned COVID-19 vaccines is dependent on the successful completion of clinical trials and submission of requisite data to the National Regulator i.e., Drug Controller General of India as per the requirements of New Drugs and Clinical Trials Rules, 2019 under Drugs and Cosmetics Act, 1940.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha today.
