The Department of Health and Family Welfare has launched a Digital Portal for Crowdfunding & Voluntary Donations for the treatment of patients of Rare Diseases in accordance with the mandate of the National Policy for Rare Diseases 2021. The Digital Portal may be accessed through https://rarediseases.nhp.gov.in/
The Department of Pharmaceuticals has initiated the implementation of Production Linked Incentive Scheme for Pharmaceuticals. The Scheme provides for financial incentives to manufacturers selected under the scheme for domestic manufacturing of various product categories, which also include Orphan drugs. The guidelines for the Scheme are available on the website of the Department of Pharmaceuticals under the tab ‘Schemes’.
The Minister of State (Health and Family Welfare), Dr Bharti Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
COVID-19 pandemic has re-affirmed the importance of adequate number of qualified and skilled human resources in health.
The number of MBBS Seats in the country has increased by 53.22% from 54,348 seats in 2014 to 83,275 seats in 2020 and the number of PG seats has increased by 80% from 30,191 seats in 2014 to 54,275 seats (including DNB & CPS seats) in 2020. The number of nursing seats in the country has increased as below:
ANM seats have increased by 5.73% from 52,479 in 2014 to 55,490 in 2020
GNM seats have increased by 12.38% from 1,15,844 in 2014 to 1,30,182 in 2020
iii. B.Sc. (N) seats have increased by 21.24% from 83,192 in 2014 to about 1,00,865 in 2020
iv. M.Sc. (N) seats have increased by 23.53% from 10,784 in 2014 to about 13,322 in 2020.
The Government has taken various steps to further increase the availability of doctors and nurses in the country which include:-
Centrally Sponsored Scheme for establishment of new medical college by upgrading district hospital in underserved districts of the country.
Centrally Sponsored Scheme for Strengthening/ upgradation of existing State Government/Central Government Medical Colleges to increase MBBS and PG seats.
iii. Viability Gap Funding Scheme for establishment of medical colleges in Public-Private Partnership mode.
iv. A Consortium (a group of 2 or up to 4 private organizations) has been allowed to establish a medical college.
Relaxation in the norms for setting up of Medical College in terms of requirement for faculty, staff, bed strength and other infrastructure.
vi. Enhancement of maximum intake capacity at MBBS level from 150 to 250.
DNB qualification has been recognized for appointment as faculty to take care of shortage of faculty.
Enhancement of age limit for appointment/ extension/ re-employment against posts of teachers/dean/principal/ director in medical colleges upto 70 years.
ix. By amending the regulations, it has been made mandatory for all medical colleges to start PG courses within 3 years from the date of their MBBS recognition /continuation of recognition.
Provision has been made in the Regulations to offer less number of seats to the applicant medical college, in case, it falls short of minimum prescribed requirements of applied intake to avoid wastage of human resources.
xi. Under the Central Sector Scheme - Development of Nursing Services, financial assistance to the tune of Rs. 7 crore for each institution is provided to schools of nursing to be upgraded to college of nursing.
The requirement of land to construct building for School/College of Nursing and Hostel has been relaxed.
The requirement of a 100 bedded parent hospital has been relaxed for hilly and tribal areas for School/College of Nursing and Hostel.
The student teacher ratio for M.Sc.(N) programme has been relaxed from 1:5 to 1:10.
Student patient ratio for Nursing Institutions has been relaxed from 1:5 to 1:3.
Distance from Nursing School to hospital has been relaxed from 15 km to 30 km. However, for hilly and tribal areas the maximum distance is 50 km. Superspeciality hospital can start M.Sc.(N) without having under graduate programme.
The Minister of State (Health and Family Welfare), Dr. BhartiPravin Pawar stated this in a written reply in the Rajya Sabha here today.
To address the health care challenges, in urban/rural areas, National Health Mission (NHM) and its two sub missions namely National Rural Health Mission (NRHM) and National Urban Health Mission (NUHM) provide technical and financial support to supplement the efforts of the States/UT Governments in order to meet the objective of equitable, accessible and affordable health care services to all. Such support includes, free medicines/ drugs, free diagnostics and patient transport, in order to lessen the burden of the out-of-pocket expenditure on health care.
Under Ayushman Bharat-Health and Wellness Centres (AB-HWC) initiative, launched by Government of India in the year 2018, 1.5 lakh Sub-Health Centres (SHCs) / Primary Health Centres, both in rural/urban areas, are being upgraded into HWCs for the betterment of local public health care. Accordingly, more than 77,406 HWCs have become functional (73,391 in rural areas & 4,015 in urban areas) and are delivering comprehensive primary health care closer to the homes of the people. Details of packages provided under AB-HWCs in rural/urban HWCs is at Annexure.
Also, the Fifteenth Finance Commission has recommended grants aggregating to Rs.70,051 Crores, over the period of five years (2021-2026) through local governments, for strengthening healthcare system at the primary health care level.
Care in Pregnancy and Child Birth.
Neonatal and Infant health care services.
Childhood and Adolescent health care services.
Family planning, contraceptive services and other reproductive health care services.
Management of communicable diseases: National Health programmes.
General out-patient care for acute simple illnesses and minor ailments.
Screening, prevention, control and management of non-communicable diseases and chronic communicable diseases like tuberculosis and leprosy.
Basic oral health care.
Screening and basic management of mental Health ailments.
Care for common ophthalmic and ENT problem.
Elderly and palliative health care services.
Emergency medical services including burns and trauma.
Expanded Service Delivery- At the AB-HWCs, the package of Comprehensive Primary Health Care services is being expanded to go beyond Reproductive and Child Health to include care for non-communicable diseases, Palliative and Rehabilitative care, Oral, Eye and Ear, Nose and Throat (ENT) care, Elderly care, Mental health and first level care for Emergencies and Trauma as mentioned in the list above.
Continuum of care/Telehealth –The AB-HWCs provide teleconsultation services, whereby every level of service provider from community health officer (CHO) to medical officer can access a higher level of consultation, including with specialists in secondary and tertiary centres, so that physical travel by patients can be minimized, reducing costs and any potential hardship. Currently, 67% of the AB-HWCs have teleconsultation facilities.
Expanding HR-MLHP & multiskilling- To improve the delivery mechanism, a new cadre of healthcare providers are introduced at the mid-level with educational backgrounds such as B.Sc in Community Health or a Nurse (GNM or B.SC) or an Ayurveda Practitioners with due certification in public health are added as Community Health Officer (CHO) or Mid-Level Health Care Provider. The CHO at SHC-HWC carry out public health functions, ambulatory care, and management and provide leadership at the Health and Wellness Centres.
Medicines & expanding diagnostics- The AB-HWCs serve as the hub for diagnostic tests and dispensing medicines based on treatment plans initiated by the medical officer at the PHC. Number of essential medicines at PHC –AB-HWCs have been increased to 172 and number of essential diagnostic services to 63. While at the SHC-AB-HWCs, these have been increased to 105 essential medicines and 14 essential diagnostic services. This ensures the uninterrupted availability of medicines to ensure adherence and continuation of care, but also reduces any patient hardship by providing medicines closer to their homes.
Community mobilisation and health promotion-
The AB-HWC team works closely with communities enables empowerment of individuals, families and communities with knowledge and skills to take responsibility for their own health. The AB-HWCs also focus on improving health literacy through interpersonal communication, and media including social media, for promotion of healthy lifestyles – diet, yoga, exercise, tobacco cessation, and self-care for those with chronic disease conditions. Institutional structures such as Jan Arogya Samitis (JAS), with representation from the local bodies and Panchayats, Self Help Groups and patients, have also been envisaged to enable community ownership and accountability of AB-HWC teams.
A Fit Health Worker Campaign was also launched at these centres to enable the screening and early detection of non-communicable diseases in the Frontline-Health Care Workers. Frontline Workers (FLWs) are not only involved in ensuring essential services at these centres but also played a crucial role in community-based surveillance and pandemic outbreak management related activities in the community.
GOI initiated training of ‘Eat Right’ and ‘Eat Safe’ module to the primary healthcare team at these functional AB-HWCs.
Thirty-nine health calendar days are celebrated by each AB-HWCs . These centres also conduct various wellness related activities like Yoga, Zumba, Meditation etc. which not only enable improved physical health but also mental wellbeing of the community. It is envisaged that these centres will not only be the point of delivery for healthcare services, but at the same time enable the community to take health in their own hands.
Community Ownership: Jan Arogya Samiti (JAS), a facility based multi-stakeholder committee, led by the PRI member of the area, has been established at AB-HWCs to enhance ownership of the community on AB-HWCs. JAS is responsible for overall functioning of the AB-HWCs. The amount of untied funds to the SHC-HWC has also been increased from Rs.20,000 to Rs.50,000 per annum.
Infrastructure Augmentation - All HWCs are provided with sufficient space for outpatient care, for dispensing medicines, diagnostic services, adequate spaces for display of communication material of health messages, including audio visual aids and appropriate community spaces for wellness activities, including the practice of Yoga and physical exercises. Financing /provider payment reforms- All HWCs have system for population empanelment and record of services, team-based incentives are provided. This facilitates accountability to outputs/outcomes and provide individual centred care.
Government of India has launched Health and Wellness Ambassadors initiative under AB-HWCs to promote a healthy lifestyle among students. It includes school health activities; teachers are trained to serve as Health and Wellness Ambassadors and students as messengers. It is a joint initiative of the Ministry of Health and Family Welfare and the Department of School Education &Literacy, Ministry of Education enables creating healthy habits leading to early action and encouragement to adopt healthy behaviours at young age and prevent chronic diseases later in life.
IT enabled Reporting and Data management- An online portal- AB-HWC portal was developed in 2018 to capture progress on AB-HWC operationalization and is being used in all the States. An App version of the AB-HWC portal has also been developed to enable geo-tagging of the AB-HWCs and entering the daily service delivery parameters by the frontline healthcare workers. The AB-HWC team are equipped with IT equipment- Tablets at SHCs and Laptop/ Desktop at PHC/UPHC level to create electronic health record of the population covered by AB-HWCs.
The Minister of State (Health and Family Welfare), Dr. Bharti Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
Government has taken a numbers of steps for strengthening Routine Immunization and ensuring that amidst the COVID-19 pandemic crisis, immunization programme is properly implemented and access to immunization services remains uninterrupted, these include:
Development and dissemination of National guidelines to safely continue immunization services during the COVID-19 pandemic.
Appropriate communication material has been developed and shared with the States and UTs for addressing vaccine hesitancy and strengthening routine immunization with due precautions, during COVID-19 pandemic.
Supply chain of vaccines and other logistics has been ensured during COVID-19 pandemic.
In addition, Intensified Mission Indradhanush (IMI-3.0) was also conducted in February 2021 & March 2021, to cover the unvaccinated and partially vaccinated children. These efforts have contributed in reduction of immunization gap coverage.
States/UTs have been asked to prepare specific plans to improve Full Immunization Coverage and to ensure designated days in a week only for Routine Immunization activity. The number of children fully immunized as reported by State/UTs in HMIS portal under the Universal Immunization Programme (UIP), State/UT-wise in 2018-19, 2019-20 and 2020-21 is as below.
Number of Children Fully Immunized, State/UT wise for 2018-19, 2019-20 & 2020-21
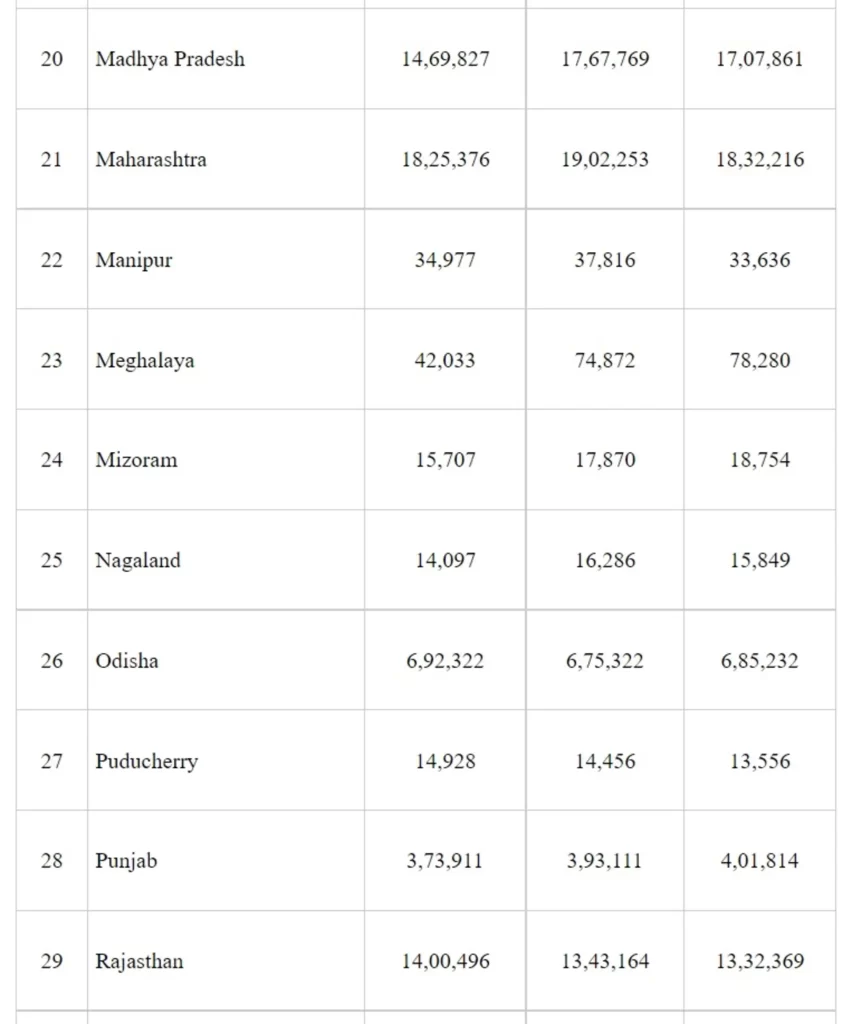
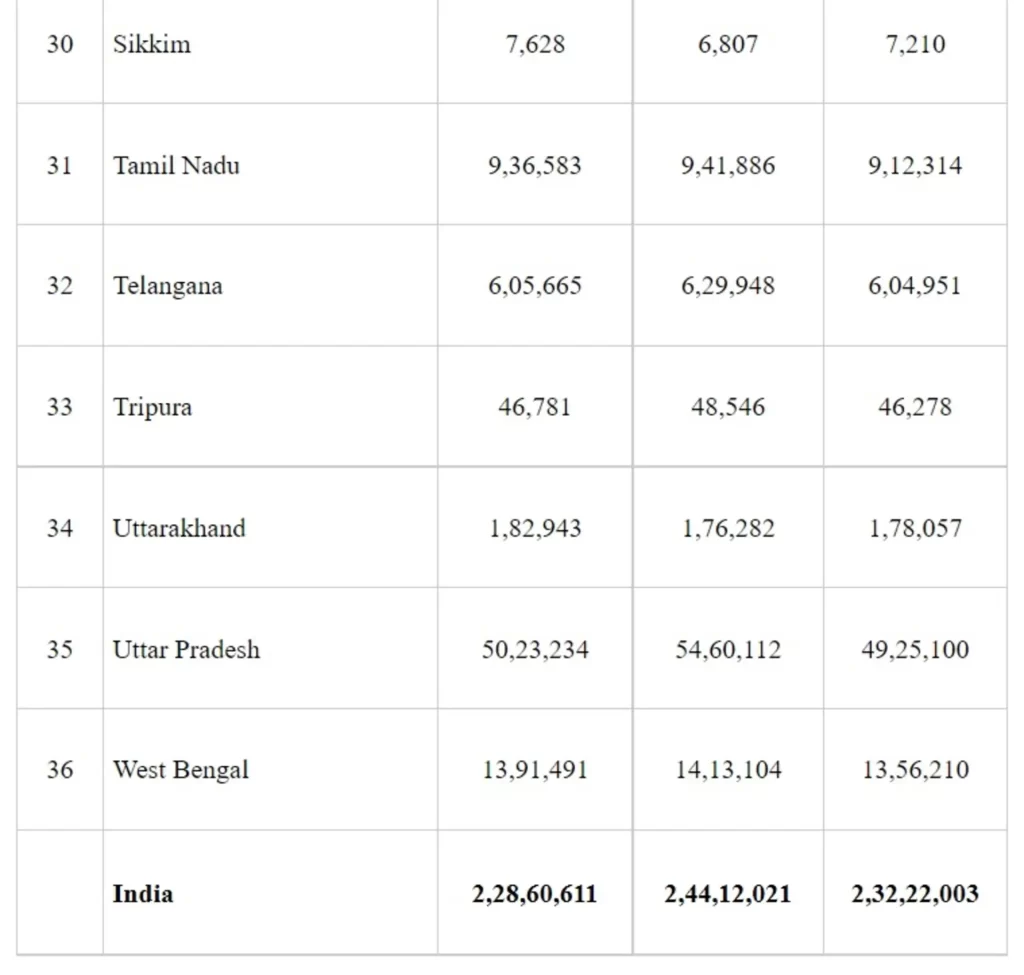
Data Source- Health Management Information System (HMIS) as on 21st July 2021
The Minister of State (Health and Family Welfare), Dr Bharti Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
National Pharmaceuticals Pricing Policy (NPPP), 2012 prescribes the guidelines for regulation of prices of drugs. The key principles of price regulation are (i) essentiality of drugs (ii) control of prices of formulations and (iii) MarketBased Pricing. Based on NPPP, 2012 and subsequent Drugs (Prices Control) Order, 2013 (DPCO, 2013), National Pharmaceutical Pricing Authority (NPPA) under the Department of Pharmaceuticals fixes the ceiling price of scheduled drugs specified in the first schedule of the DPCO, 2013 and monitors the prices of non-scheduled drugs.
NPPA has fixed the ceiling prices of scheduled drugs, including the essential medicines used for treatment of cancer, diabetes and HIV as well as heart and kidney diseases. Further, NPPA has put a cap on Trade Margin of 42 select non-scheduled anti-cancer medicines under ‘Trade Margin Rationalization (TMR)’ Approach resulting in reduction up to 90% of Maximum Retail Price (MRP) of526 brands of these medicines.
NPPA has also brought 106 non-scheduled anti-diabetic and cardiovascular drugs under price control by invoking extraordinary powers in public interest.The total annual savings on account of revision of ceiling prices of medicines under National List of Essential Medicines (NLEM), price control of anti-diabetic & cardiovascular, fixation of ceiling price of stents, knee implants and capping of TMR on anti-cancer are estimated to the tune of Rs. 12,500 crore. NPPA monitors the ceiling price of the scheduled formulations to ensure that the MRP of such formulations are within the range of ceiling price and monitors non-scheduled formulations to ensure that their MRP does not increase by more than 10% during the preceding twelve months. The details of retail/ceiling prices fixed/revised by NPPA are available on NPPA’s website www.nppaindia.nic.in.
The details of the formulations for which price have been fixed by NPPA in the last 3 years are as follows:
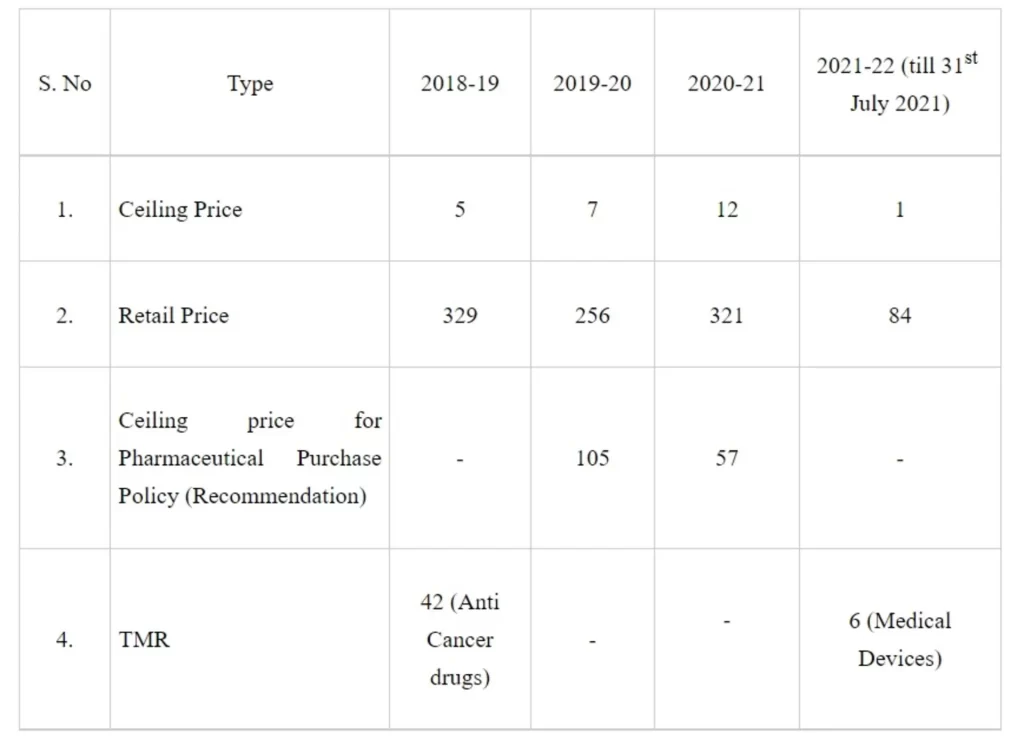
Under the Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP), till 04.08.2021, about 8,004 Pradhan Mantri Bhartiya Janaushadhi Kendras (PMBJKs) are open across the country to provide generic medicines at cheaper rates to the citizens. Till 04.08.2021, about 236 PMBJKs are open in the State of Chhattisgarh. State/UT wise list of PMBJKs is as:
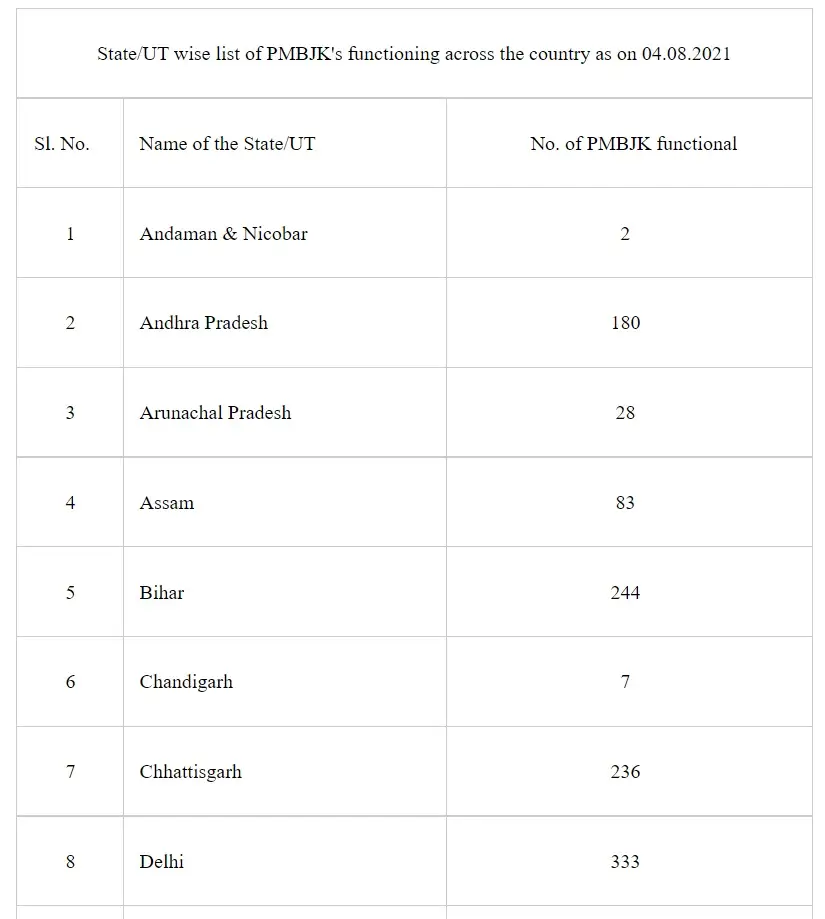
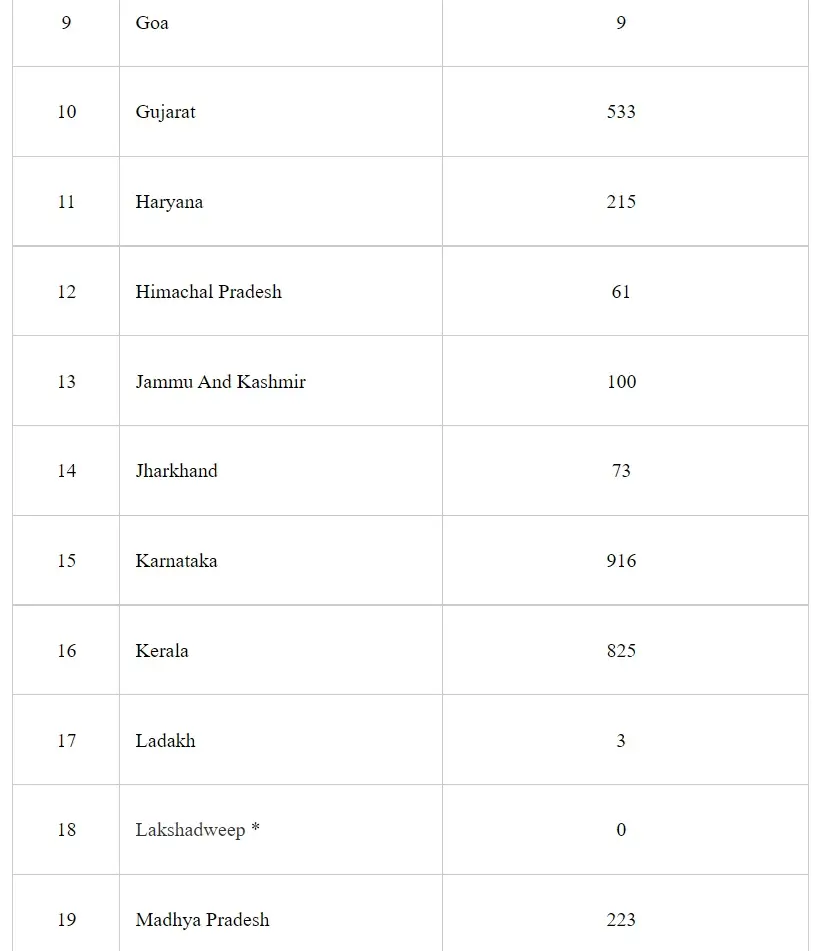
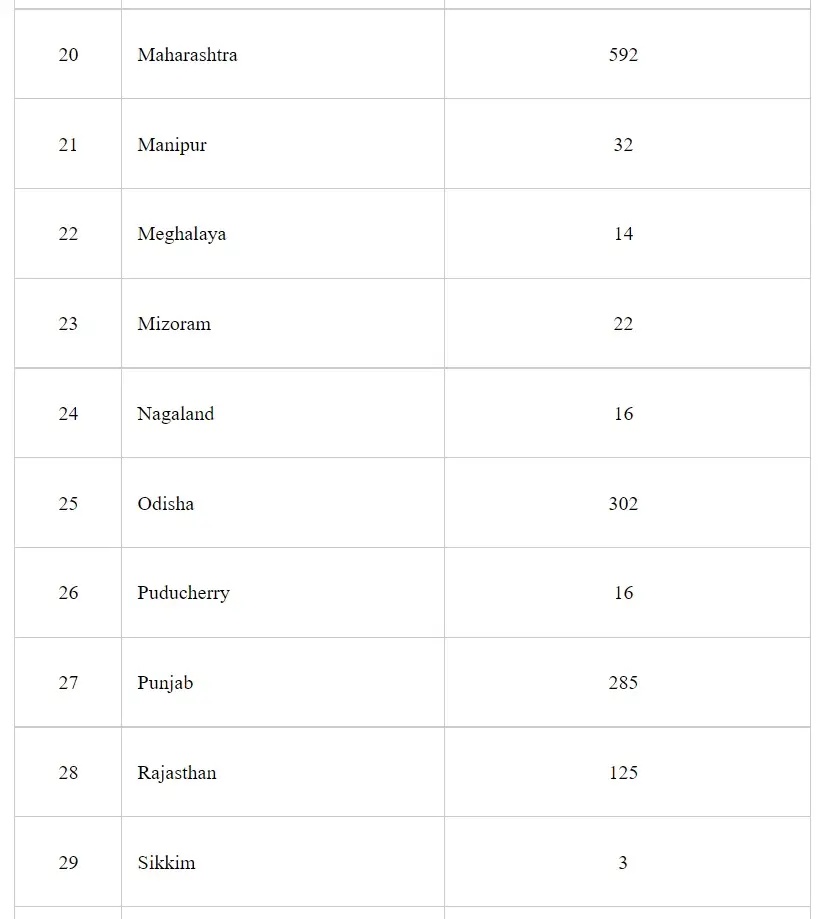
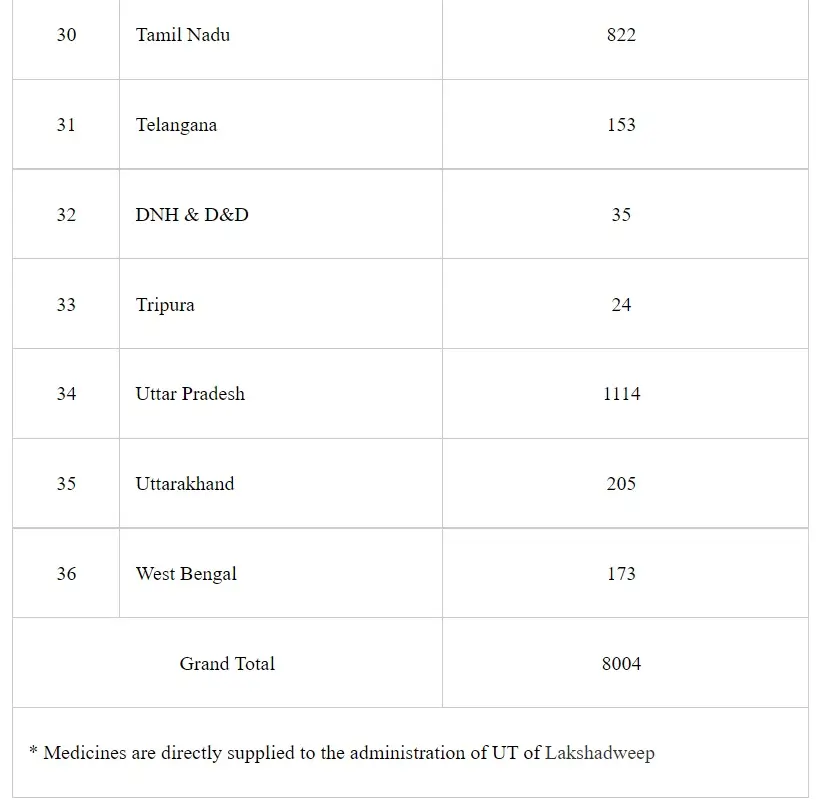
This information was given by the Minister of Health & Family Welfare and Chemicals & Fertilizers, Shri Mansukh Mandaviya in a written reply in Lok Sabha today.
In a landmark initiative to ensure safety from COVID-19, Ministry of Health and Family Welfare has taken a decision to allow foreign nationals residing in India to get registered on CoWin portal to take COVID-19 vaccine. They can use their passport as identity document for the purpose of registration on CoWIN portal. Once they are registered on this portal, they will get a slot for vaccination.
A significant number of foreign nationals are living in India, especially in large metropolitan areas. In these areas, the potential of spread of Covid-19 is high due to higher population density. To counter any possibility of such occurrence, it is important to vaccinate all eligible persons.
This initiative will ensure the safety of foreign nationals residing in India. It will also bring down the possibilities of further transfer of infection from unvaccinated persons residing in India. It will also ensure overall safety from further transmission of COVID-19 virus.
The National Covid-19 Vaccination Programme is being implemented across all states and Union Territories since 16th January, 2021. The vaccination programme in its current phase covers all citizens aged 18 years and above. As on 9th August, 2021, India has administered more than 51 crore vaccine doses across the country.
The National Telemedicine Service of the Ministry of Health & Family Welfare has completed 90 lakh teleconsultations in the country. The last 10 lakh consultations came up in record 17 days.
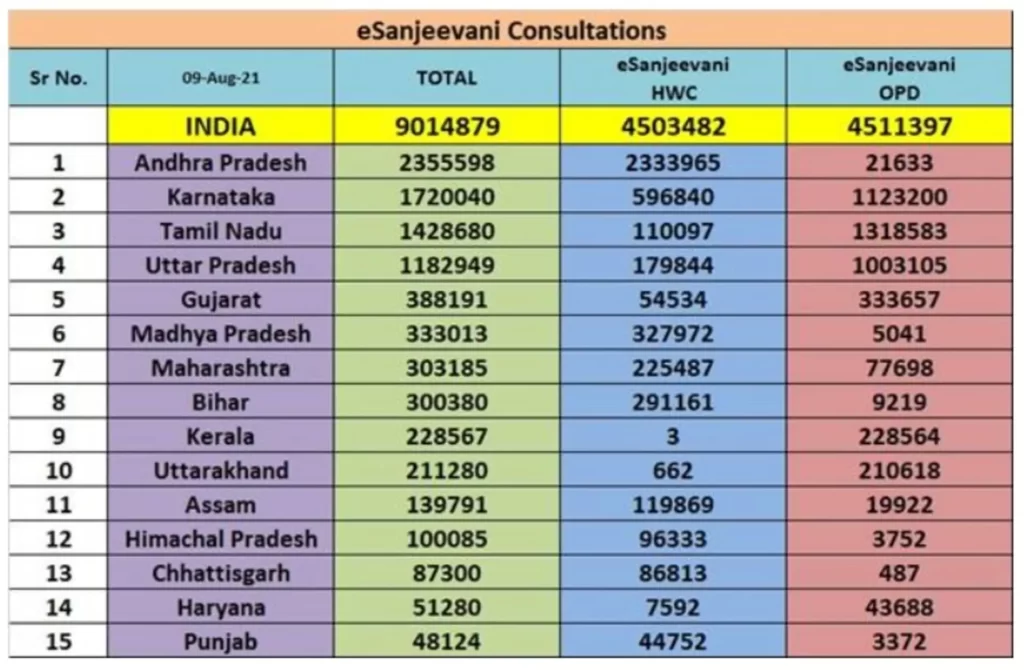
Government of India’s eSanjeevani initiative has gained popularity amongst doctors and patients across country. New users are continuously adopting telemedicine as eSanjeevani has proved to be a feasible, easy to use, practical means of seeking health services remotely. This is evident from the fact that in past one month, eSanjeevani network’s utilisation has gone up by a factor of around 1.5 times.
eSanjeevani platform has been consistently getting diffused in the ecosystem and currently it is serving 70,000 patients on daily basis. eSanjeevani – the National Telemedicine Service is serving patients through two variants namely - doctor to doctor (eSanjeevaniAB-HWC) and patient to doctor (eSanjeevani OPD). eSanjeevani AB-HWC was launched by Ministry of Health and Family Welfare in November 2019 under Govt. of India’s Ayushman Bharat Scheme in ‘Hub & Spoke’ model and by December 2022 it would be implemented at 1,55,000 Health & Wellness Centres across India. eSanjeevaniAB-HWC is functional at over 25,000 Health and Wellness Centres as spokes and these spokes are being served by medical officers, specialists and super-specialists seated in over 2000 hubs.
The second variant of this mammoth initiative i.e. eSanjeevani OPD was rolled out on 13th of April 2020 during the first lockdown when the OPDs across the country were shutdown. eSanjeevaniOPD is enabling delivery of health services to patients in the confines of their homes. At present eSanjeevani OPD is hosting over 430 online OPDs, around 400 of these are speciality and super-speciality online OPDs. Over 60,000 doctors and health-workers across the country have been trained on both the variants of eSanjeevani.
Considering the potential that telemedicine has especially in view of the ongoing pandemic, States and Union Territories are consistently working towards strengthening the IT infrastructure within the health care delivery set ups. These efforts are aimed at stepping up the practice of telemedicine at all levels, they are utilising eSanjeevaniAB-HWC platform to ensure access of specialised health services to the populace living in rural and isolated areas. The States and UTs are also stepping up this digital health modality by opening newer speciality and super-speciality onlineOPDs on eSanjeevaniOPD. Centre for Development of Advanced Computing in Mohali is providing end-to-end services in maintaining the National Telemedicine Service and in addressing the needs of the users in States and Union Territories.
The top ten States which have registered highest consultations through eSanjeevani and eSanjeevani OPD platforms are Andhra Pradesh (2355598), Karnataka (1720040), Tamil Nadu (1428680), Uttar Pradesh (1182949), Gujarat (388191), Madhya Pradesh (333013), Maharashtra (303185), Bihar (300380), Kerala (228567), Uttarakhand (211280).
Vice President of India and Chairman of Rajya Sabha Shri Venkaiah Naidu along with Speaker of Lok Sabha Shri Om Birla chaired an event today to sensitise Parliamentarians on the country’s efforts to eliminate TB. Shri Mansukh Mandaviya, Union Minister for Health and Family Welfare and Dr. Bharati Pravin Pawar, Union Minister of State for Health and Family Welfare apprised the audience on the situation of Tuberculosis in the country and explained the need to rapidly eliminate the disease by 2025, as has been envisioned by Prime Minister Shri Narendra Modi in 2018.

Thanking the parliamentarians for being present at this sensitization exercise, the Vice President stated that “coordination at the central, state, district and local levels would help in making this a people’s movement and would expedite our efforts in eliminating TB by 2025.” Significant progress has been achieved since independence on this front and several others due to increasing penetration of healthcare systems across the country. For e.g. 63 million lives have been saved since 2000 through TB diagnosis and treatments, he added. He urged the members to set a goal in their constituencies for proper planning and execution of govt programs and help in eliminating TB. A pledge was taken by Parliamentarians for a TB Mukt Bharat.

Lok Sabha Speaker Shri Om Birla, calling for a collective action against TB, emphasized the crucial role played by the Parliament in disseminating the message in a country full of diversity. He said that it is responsibility of the people's representatives to ensure that their constituencies have constant identification and monitoring of the TB patients, look after the needs of patients during and post treatment. For smooth governance and health administration, parliamentarians would be provided with all necessary data related to TB and any assistance needed from the government, he added.

In his address, Shri Mansukh Mandaviya noted, “Prime Minister Shri Narendra Modi ji played a huge role in moulding thinking of the country towards making health a ‘comprehensive’ subject by linking it with development.” Giving examples of programmes targeting malnutrition and those aimed at promoting cleanliness run by line ministries, he said that all these programmes ultimately improve the social determinants of health thereby improving the living standards of India’s population. He exemplified the model in the country’s efforts to eliminate TB which requires nutritional support and wide social awareness. He highlighted the increased budgetary provisions that aim to provide holistic health to India’s citizens.
The Union Health Minister underscored that a staggering 65% of TB cases are in the age group 15-45 which is the most economically productive population segment. This coupled with the fact that 58% of TB cases are in rural areas translates to entire families being excluded from upward mobility due to Tuberculosis. He thus asked all the MPS to proactively engage in sensitizing citizens about Tuberculosis and its treatment in the events they organise and attend.
Dr. Bharati Pawar spoke on the difficulties in the journey towards TB elimination with the onset of COVID-19: “Globally, 2020 witnessed a sweeping COVID-19 pandemic devastate lives, economies, health systems and health programmes across the world with record-breaking speed. In just a few months, the pandemic has reversed years of progress made in the fight against Tuberculosis. The onset of the pandemic in March 2020 triggered lockdowns, restrictions in movement, repurposing of available health system resources, infrastructure, diagnostics, treatment centers and manpower to fight Covid-19, disrupted ongoing TB elimination efforts and services all over the country.” She informed her colleagues in Parliament on the steps being taken by the Ministry to rapidly make up for this interruption in the elimination of TB. She also suggested the effective sharing of collective responsibility with States in tackling the social determinants of TB for better and rapid health outcomes.
Union Health Secretary Shri Rajesh Bhushan, Shri Utpal Kumar Singh, Secretary General, Lok Sabha and Ms. Arti Ahuja, Additional Secretary (Health) were also present.
Government of India has provided the required technical support and has also supported the states through logistic and financial support to further strengthen the existing health infrastructure to tackle COVID-19 pandemic.
Some of the ongoing initiatives to further strengthen healthcare infrastructure include:
With the intent to reduce the risk of cross infection to non-COVID patients as well as to maintain continuity of non-COVID essential health services in the country, a three-tier arrangement of dedicated COVID-19 health facilities [(i) COVID Care Center (CCC); (ii) Dedicated COVID Health Centre (DCHC) and (iii) Dedicated COVID Hospital (DCH)] has been implemented in the country.
Government of India, to supplement the hospital facilities has roped in tertiary care hospitals under ESIC, Defence, Railways, paramilitary forces, Steel Ministry etc. Further, many large temporary treatment facilities were established by DRDO to manage surge in COVID-19 cases in the country. The isolation bed capacity and ICU bed capacity which was only 10,180 and 2,168 before the first lockdown (as on 23rd March 2020) in being enhanced continuously and is currently at 18,03,266 isolation beds and 1,24,598 ICU beds (as on 3rd August 2021).
The daily liquid medical oxygen (LMO) supply, which was about 1292 MTs per day in February 2021 increased to 8593 MTs in April 2021. On 28th May 2021, a total of 10,250 MTs of LMO was allocated to the states. This was done by enhancement of LMO production in steel plants as well as in other LMO plants. Restrictions were imposed on industrial use of oxygen. A dynamic and transparent framework for allocation of medical oxygen in consultation with States/UTs and all the stakeholders such as relevant Ministries, manufacturers/suppliers of liquid oxygen etc. was prepared.
Online digital solutions viz. Oxygen Demand Aggregation system (ODAS) and Oxygen Digital Tracking System (ODTS) have been developed to ascertain the demand for medical oxygen from all medical facilities and to track their transportation. To avoid wastage of medical oxygen, guidelines on rational use of oxygen were issued on 25th September 2020, and further revised and disseminated to States on 25th April 2021. 1,02,400 oxygen cylinders were procured in April and May of 2020 and distributed to States. Further orders for additional 1,27,000 cylinders have been placed on 21.04.2021 (54,000 jumbo cylinders (D type) and 73,000 regular cylinders (B type). Deliveries of the same have started and 73,352 (56,108 B-type and 14,244 D-type) cylinders have been delivered as on 3rd August 2021.
To generate oxygen at the health facility level, PSA plants are being established in each district hospitals, especially in far flung areas enabling the hospitals to become self-sufficient in generation of oxygen for their needs and thereby, reduce the burden on the medical oxygen supply grid across the country.
Further, to fast-track the availability of Medical Oxygen in rural and peri-urban areas, more than 39,000 oxygen concentrators have been allocated to various States.
A COVID Drugs Management Cell (CDMC) has been set up in the Department of Pharmaceuticals (DoP) to oversee the management of smooth supply of drugs used in COVID-19 management.
A Drugs Coordination Committee (DCC) has been constituted as an institutional mechanism under Department of Pharmaceuticals for efficient decision making on all the issues with respect to COVID-19 related drugs including availability through inter-departmental consultations.
Remdesivir is a patented drug, manufactured in India under voluntary licenses granted by Gilead Life Sciences USA (the patent holder) to 7 Indian pharmaceutical companies. Manufacturing capacity was augmented from 38 lakh vials per month in March 2021 to nearly 122 lakh vials per month in June 2021. In addition, 40 additional manufacturing sites were approved by the CDSCO, thus increasing the manufacturing sites from 22 (in March 2021) to 62 (June 2021).
All States/UT and State Drugs Controllers have been requested to verify stock of drugs and check other malpractices and take effective steps to curb hoarding and black marketing of some drugs like Remdesivir.
Department of Pharmaceuticals and the Drug Controller General of India (DCGI) have actively coordinated with the industry to enhance availability of Amphotericin B through identification of manufacturers, alternate drugs and expeditious approvals of new manufacturing facilities.
Besides, the existing five manufacturers, DCGI had issued permissions to manufacturing / marketing of Amphotericin B Liposomal Injection to six additional firms. The guiding principle to avert/ minimize the risk of future resurgence of COVID-19 cases in the country remains the five-fold strategy of test-track-treat-vaccinate and COVID appropriate behavior.
Ministry of Health & Family Welfare continues to provide technical guidance for managing various aspects of COVID-19. So far more than 150 guidelines/advisories/SoPs/plans have been provided to States/UTs. Taking note of ingress of COVID-19 pandemic in peri-urban and rural areas, Ministry of Health & Family Welfare on 16th May 2021 issued an SOP on COVID-19 Containment & Management in Peri-urban, Rural & Tribal areas.
Further COVID-19 treatment protocols and advisories both for adults as well as pediatric age groups were issued and widely disseminated to promote rational use of drugs and oxygen. During the F.Y. 2019-20. funds to the tune of Rs.1113.21 crore was released to the States/UTs under NHM towards management and containment of COVID-19 pandemic.
In September 2020, the Union Government further allowed use of SDRF by the States for oxygen generation and storage plants in hospitals; strengthening ambulance services for transport of patients; and setting up containment zones, COVID-19 care centres. States were allowed to spend maximum 35% of annual allocation of funds under SDRF for the financial year 2019-20. The ceiling was further enhanced to 50% during the financial years 2020-21 and 2021-22 for containment measures of COVID-19.
During the FY 2020-21, funds to the tune of Rs.8257.88 crore has been released to the States/UTs towards the India COVID-19 Emergency Response and Health System Preparedness Package. In addition, ‘India COVID-19 Emergency Response & Health System Preparedness Package: Phase-II’ has also been approved by the Cabinet with Rs 23,123 crores (with Rs. 15,000 Cr as Central Component & Rs 8,123 Cr as State component) and is to be implemented from 1st July 2021 to 31st March 2022. So far Rs. 1827.78 crore has been released to States/UTs in 2021-22 under ECRP Phase-II in FY 2021-22.
It includes support to State/UT level for ramping up Health Infrastructure including those in rural, tribal and peri-urban areas closer to the community, providing support for procurement of drugs and diagnostics to enhance service delivery at district and sub district levels for management of COVID-19 cases (including pediatric care) and for maintaining a buffer of drugs, support for IT Interventions such as implementation of Hospital Management Information System and expanding access to tele-consultations in all districts, and support for capacity building and training for all aspects of management of COVID-19.
Further, under the National COVID Vaccination Program, Government of India is procuring vaccines and providing them free of cost to States and UTs. As on 3rd August 2021, a total of about 50.21 crore doses have been supplied to States/UTs from all sources i.e., Government of India’s Covid vaccine supply free of cost to all States/UTs, State/UTs and Private Hospitals procured Covid vaccine.
The COVID-19 response strategy since inception is supported by expert groups comprise of epidemiologists, scientists, microbiologists, clinicians etc. drawn from eminent institutions including World Health Organization. Further, the research community and pharmaceutical industry including vaccine manufacturers have supported COVID-19 response in terms of research and development of vaccines, as well as their production.
Union Ministry of Health through Department of Health Research has also been commissioning studies to find answers to pressing research questions related to COVID-19. These projects have been awarded and funds provided to ICMR and non-ICMR scientists in various important areas.
In addition, recently Indian Council of Medical Research had put out a call for proposals, inviting concept proposals in various areas related to COVID-19. These include clinical research, epidemiology, operational & socio-behavioral research and diagnostics & biomarkers. Selected proposals with public health translational value will be funded after scientific review by ICMR.
The Minister of State (The Ministry of Health and Family Welfare), Dr. Bharti Pravin Pawar stated this in a written reply in the Lok Sabha here today.
The manufacturing cost of medicine/vaccine is dependent of factors such as development cost, Intellectual Property Rights (IPR) cost, technology, vaccine platform & scale of production in different companies.
Department of Biotechnology has informed that under “Mission COVID Suraksha- the Indian COVID-19 Vaccine Development Mission”, being implemented by Biotechnology Industry Research Assistance Council (BIRAC), a Public Sector Undertaking (PSU) of DBT, facility augmentation of Bharat Biotech and 3 Public Sector Enterprises (PSEs) including Haffkine Biopharmaceutical Corporation Ltd, Mumbai; Indian Immunologicals Limited (IIL); Hyderabad; Bharat Immunologicals Biologicals Corporation Limited (BIBCOL), Bulandshahr; is being supported, for augmented production of Covaxin. Further, Technology transfer of Covaxin production to Gujarat COVID Vaccine Consortium (GCVC), comprising Hester Biosciences, OmniBRx Biotechnologies Pvt Ltd and Gujarat Biotechnology Research Centre(GBRC), Department of Science and Technology, Govt. of Gujarat; is being facilitated by the Department of Biotechnology, with a view to enhance the production of Covaxin in the coming months.
Ministry of Health and Family Welfare has provided 100% advance to domestic vaccine manufacturers in respect of procurement order placed with them. These funds can be used by such manufacturers for their capacity augmentation. Government has also provided financial assistance to one of the vaccine manufacturer i.e. M/s Biological E for ‘At-risk manufacturing’ of COVID-19 vaccine.
Further, the Central Drug Standards and Control Organisation (CDSCO) under the Ministry of Health and Family Welfare has put in place a system for fast track processing of application for clinical trial & approval for COVID-19 Vaccines.
Ministry of Health and Family Welfare, Government of India has ensured improved accessibility of safe COVID-19 vaccination services for eligible beneficiaries irrespective of their socio-economic status. This has been done through increase in vaccination centres, citizen friendly upgradation of Co-WIN establishment of 24x7 national call center helpline to address queries from general public on COVID-19 and the vaccination process.
Government of India is also providing 15 days advance visibility of vaccine availability to States/UTs with an advice to prepare and publicize in advance district-wise and COVID-19 Vaccination Centers wise (CVCs) plan for accelerating the coverage of COVID-19 vaccination and for convenience of citizens. Further, a communication strategy is in place which is being implemented across all States/UTs with a focus to sustain vaccine confidence and address vaccine hesitancy. Accessibility to vaccination is being facilitated by involving Private CVCs, workplace CVCs & ‘Near to Home’ CVCs. Government of India regularly reviews the progress of National COVID-19 Vaccination Programme with all States/UTs to expedite its progress.
Government of India has made all arrangements to secure COVID-19 vaccines for the eligible beneficiaries as per production and the availability of COVID-19 vaccines in the country. It is expected that adequate quantity of COVID vaccine will be available between January 2021 to December 2021 to vaccinate eligible beneficiaries aged 18 years and above.
This information was given by the Minister of Health & Family Welfare and Chemicals & Fertilizers, Shri Mansukh Mandaviya in a written reply in Rajya Sabha today.
