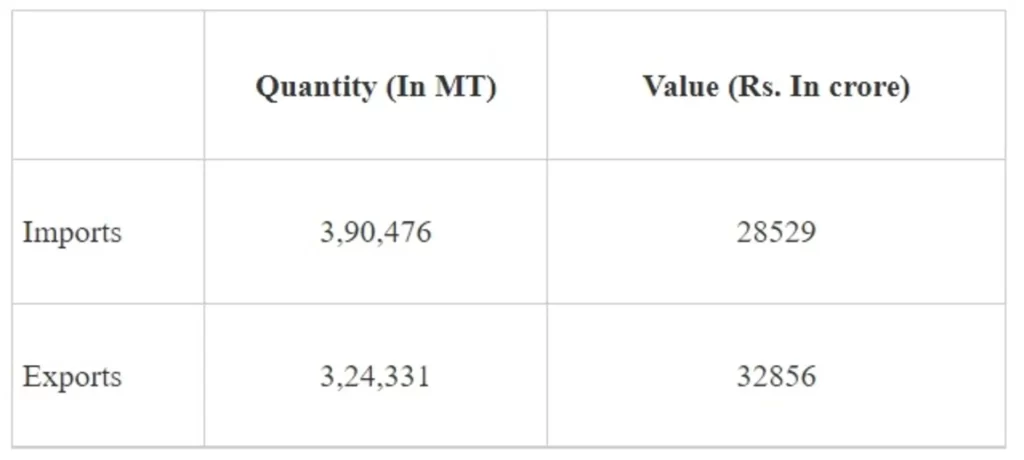
The quantity of API manufactured domestically is not readily available. However, the size of API and Intermediates market in India is estimated to be Rs. 96,000 crore in FY 2019-20. Further, as per data maintained by Directorate General of Commercial Intelligence and Statistics (DGCIS), the quantity and value of API imported and exported in FY 2020-21 is given below:
The Department of Pharmaceuticals is implementing the following three (03) schemes for promoting domestic manufacturing of APIs to ensure their sustainable domestic supply and make India AtmaNirbhar (self-reliant):
Production Linked Incentive (PLI) Scheme for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/ Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) In India: The scheme provides for financial incentives to manufacturers selected under the scheme for manufacturing of 41 Key Starting Materials (KSMs)/ Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs). The incentives will be provided on incremental sales to selected participants for a period of 6 years. The total financial outlay of the scheme is Rs. 6,940 crore and the tenure of the scheme is from FY 2020-2021 to 2029-30.
Scheme for Promotion of Bulk Drug Parks: The scheme provides for grant-in-aid to three (03) Bulk Drug Parks for creation of Common Infrastructure Facilities (CIF) with a maximum limit of Rs.1000 crore per park or 70% of the project cost of CIF, whichever is less. In case of North Eastern States and Hilly States (Himachal Pradesh, Uttarakhand, Union Territory of Jammu & Kashmir and Union Territory of Ladakh) financial assistance would be 90% of the project cost. The total financial outlay of the Scheme is Rs. 3000 crore and the tenure of the Scheme is from FY 2020-21 to 2024-25.
Production Linked Incentive Scheme for Pharmaceuticals: The scheme provides for financial incentives to manufacturers selected under the scheme for manufacturing of Key Starting Materials (KSMs)/ Drug Intermediates (DIs) and Active Pharmaceutical Ingredients (APIs) amongst other categories of formulations. The incentives will be provided on incremental sales to selected participants for a period of 6 years. The total financial outlay of the scheme is Rs. 15,000 crore and the tenure of the scheme is from FY 2020-2021 to 2028-29.
This information was given by the Minister of Health & Family Welfare and Chemicals & Fertilizers, Shri Mansukh Mandaviya in a written reply in Lok Sabha today.
As per Central Drugs Standard Control Organisation (CDSCO), isolated complaints regarding spurious drugs are received in CDSCO. As and when such complaints are received, based on merit, the matter is taken up by the CDSCO in coordination with State/UT Drugs Controller for action as per the provisions of the Drugs & Cosmetics Act, 1940 and the Drugs & Cosmetics Rules, 1945. The information received from Drugs Controllers of various States/UTs during last three years and current year in this context is as under:
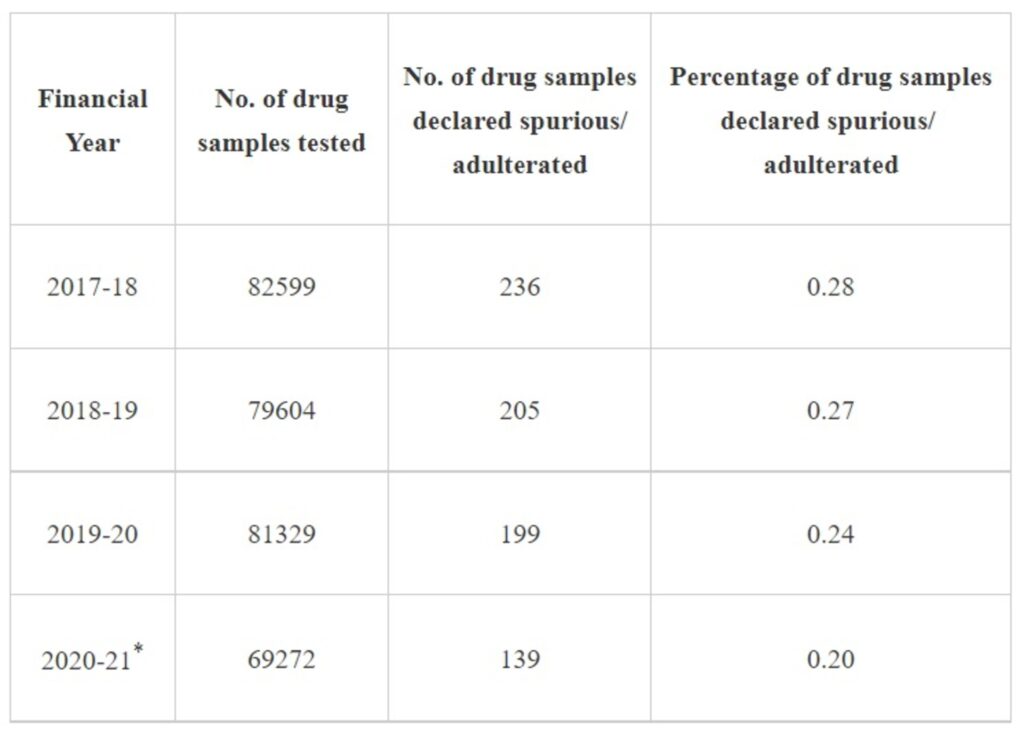
Corresponding information received from various Zonal/Sub-zonal offices of CDSCO is as under:
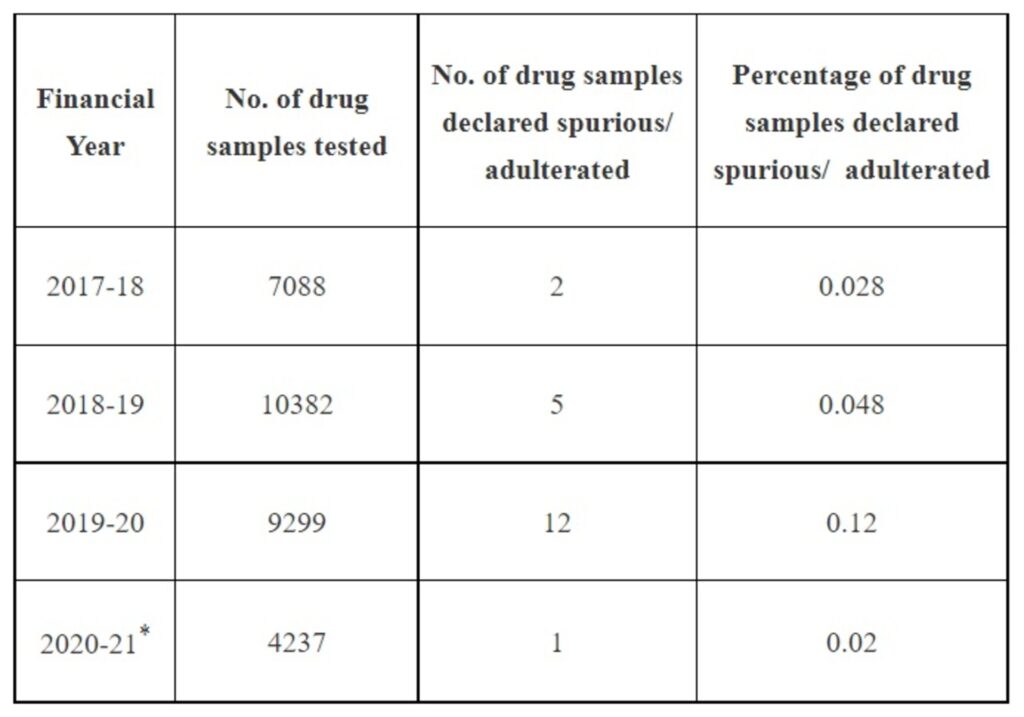
*The information of FY 2020-21 is upto 31st January, 2021 only.
Further, amidst reports received of fake & spurious Covid-19 management drugs, CDSCO has requested all States/UTs Drugs Controllers through several advisories to instruct their enforcement staff to keep strict vigil especially at sensitive places and to take stringent action against the offenders by conducting special drive of monitoring and investigation.
As per information available from various State Licensing Authorities, in cases of fake &spurious Covid management drugs, various enforcement actions like Drug seizure, Arrests of accused persons / registration of FIR etc. have been carried out by the States/UTs Drugs Controllers. CDSCO and Ministry of Health and Family Welfare have taken various regulatory measures to ensure the quality of medicines distributed in the country which are as under:
The Drugs and Cosmetics Act, 1940 was amended under the Drugs & Cosmetics (Amendment) Act 2008 to provide stringent penalties for manufacture of spurious and adulterated drugs. Certain offences have also been made cognizable and non-bailable.
The States / UTs were requested to set up special Courts for trial of offences under the Drugs and Cosmetics Act for speedy disposal. So far, 33 States have already set up designated special Courts.
Guidelines for taking action on samples of drugs declared spurious or not of standard quality in the light of enhanced penalties under the Drugs & Cosmetics (Amendment) Act, 2008 were forwarded to the State Drugs Controllers for uniform implementation.
The number of sanctioned posts in the Central Drugs Standard Control Organization (CDSCO) has been increased from 111 in 2008 to 492 (in Jan, 2021).
The testing capacities of Central Drugs Testing Laboratories under the CDSCO are being constantly strengthened to expedite testing of drug samples in the country.
On 3.4.2017, in order to ensure efficacy of drugs, the Drugs and Cosmetics Rules, 1945 have been amended providing that applicant shall submit the result of bioequivalence study along with the application for grant of manufacturing license of oral dosage form of drugs falling under the Category II and Category IV of the Biopharmaceutical Classification System.
On 27.10.2017, the Drugs and Cosmetics Rules, 1945 have been amended vide Gazette notification no. G.S.R. 1337 (E) making it mandatory that before the grant of manufacturing license, the manufacturing establishment is to be inspected jointly by the Drugs Inspectors of Central Government and State Government.
Joint inspection of the licensed manufacturing premises by the Drugs Inspectors of Central Government and State Government to verify the compliance with the conditions of license and the provisions of the Drugs & Cosmetics Act and Rules for not less than once in three years or as needed as per risk based approach is also provided for.
On 10.04.2018, the Drugs and Cosmetics Rules, 1945 have been amended vide Gazette notification no. G.S.R. 360 (E), making it mandatory for all drugs, that the applicants shall submit evidence of stability, safety of excipients etc. to the State Licensing Authority before grant of product manufacturing license by the Authority.
This information was given by the Minister of Health & Family Welfare and Chemicals & Fertilizers, Shri Mansukh Mandaviya in a written reply in Lok Sabha today.
Under Health Minister’s Discretionary Grant (HMDG) financial assistance up to a maximum of Rs. 1.25 lakhs are provided to the poor indigent patients to defray a part of the expenditure on hospitalization/treatment in Government Hospitals for life threatening diseases covered under Rashtriya Arogya Nidhi (RAN) in cases where free medical facilities are not available. Only those having annual family income up to Rs.1.25 lakhs and below are eligible for financial assistance under the scheme.
State wise list is at Annexure-I.
As on 30-07-2021 forty applications for grant of financial assistance under Health Minister’s Discretionary Grant (HMDG) are under process in the Ministry.

The Minister of State (The Ministry of Health and Family Welfare), Dr. Bharti Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
The Ministry of Health & Family Welfare administers a Centrally Sponsored Scheme (CSS) for “Establishment of new medical colleges attached with existing district/referral hospitals”. Establishment of 157 new Medical Colleges has been approved under the scheme in three phases. Out of these 157 medical colleges, 47 have become functional. Another 36 medical colleges have applied to the National Medical Commission (NMC) for permission. Under this scheme, the State Government is the implementing agency and the planning, execution and commissioning of the projects is done by the State Governments. Detailed status of these medical colleges along with funds released is at Annexure.
Annexure
Details of medical colleges approved under Phase-I of Centrally Sponsored Scheme for ‘Establishment of new Medical Colleges attached with existing District/Referral Hospitals’
Rs. in crore
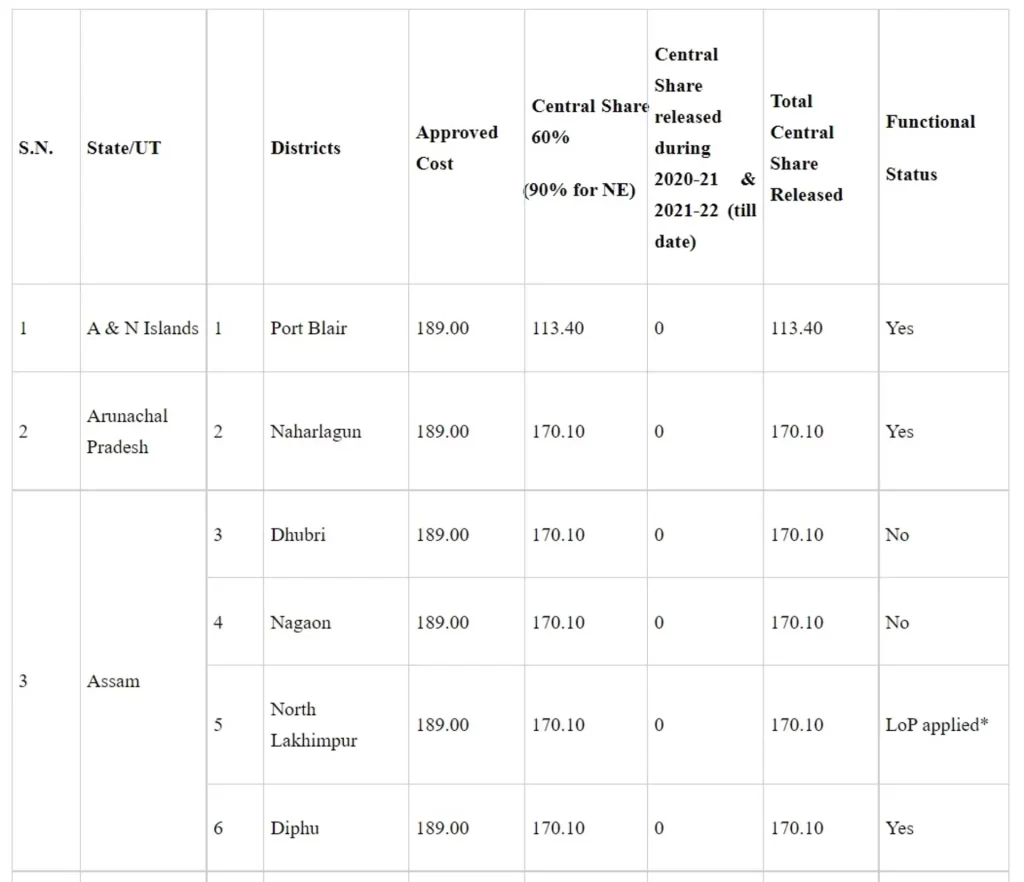
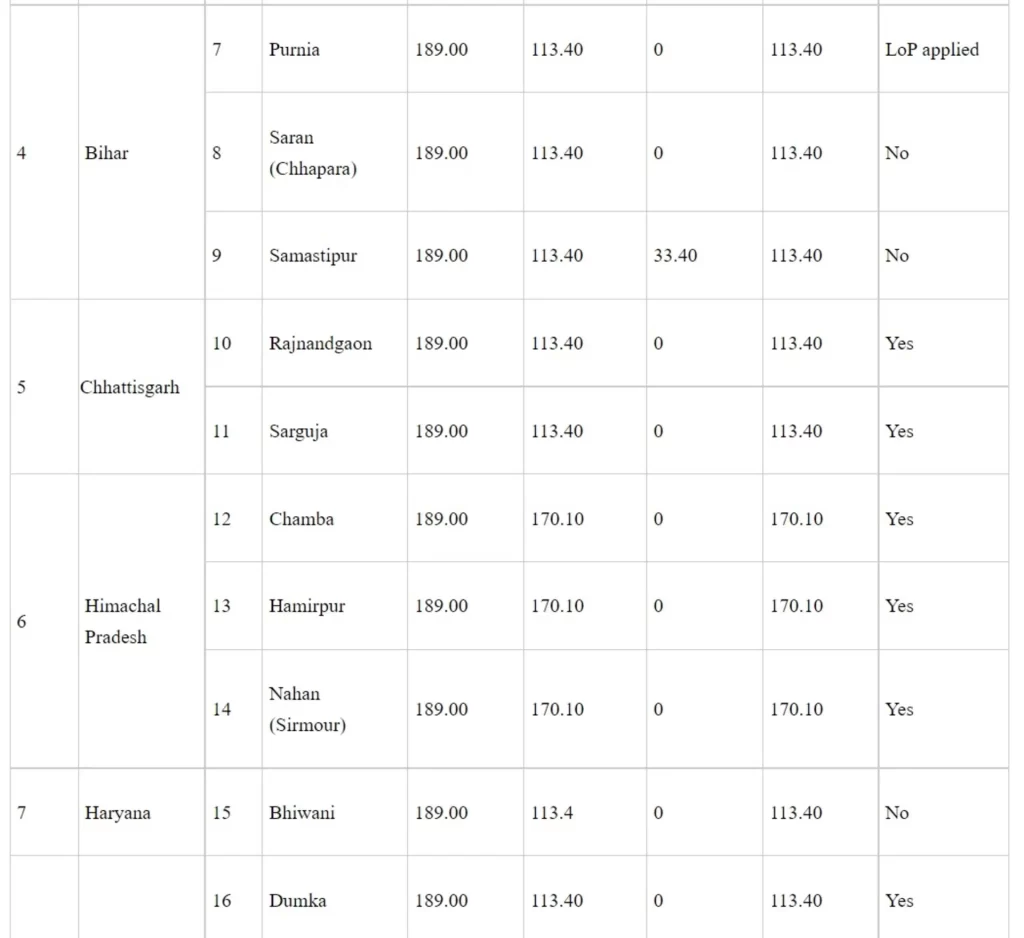
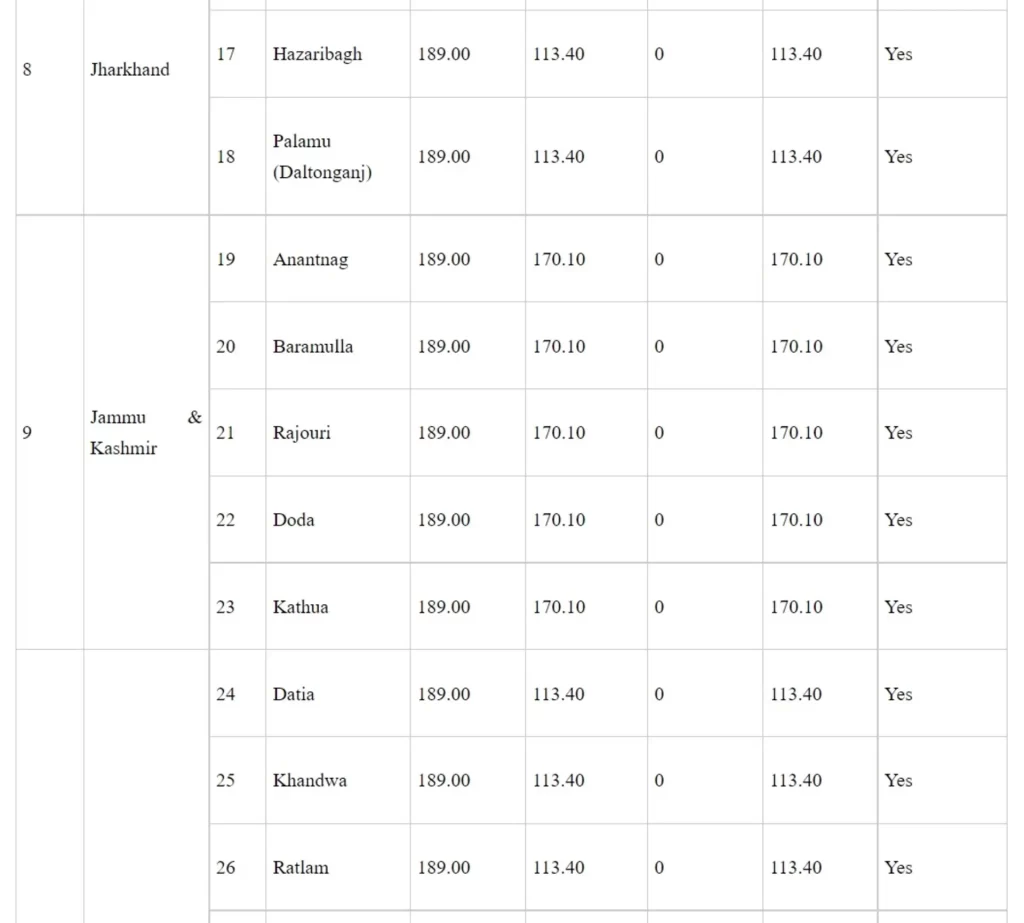
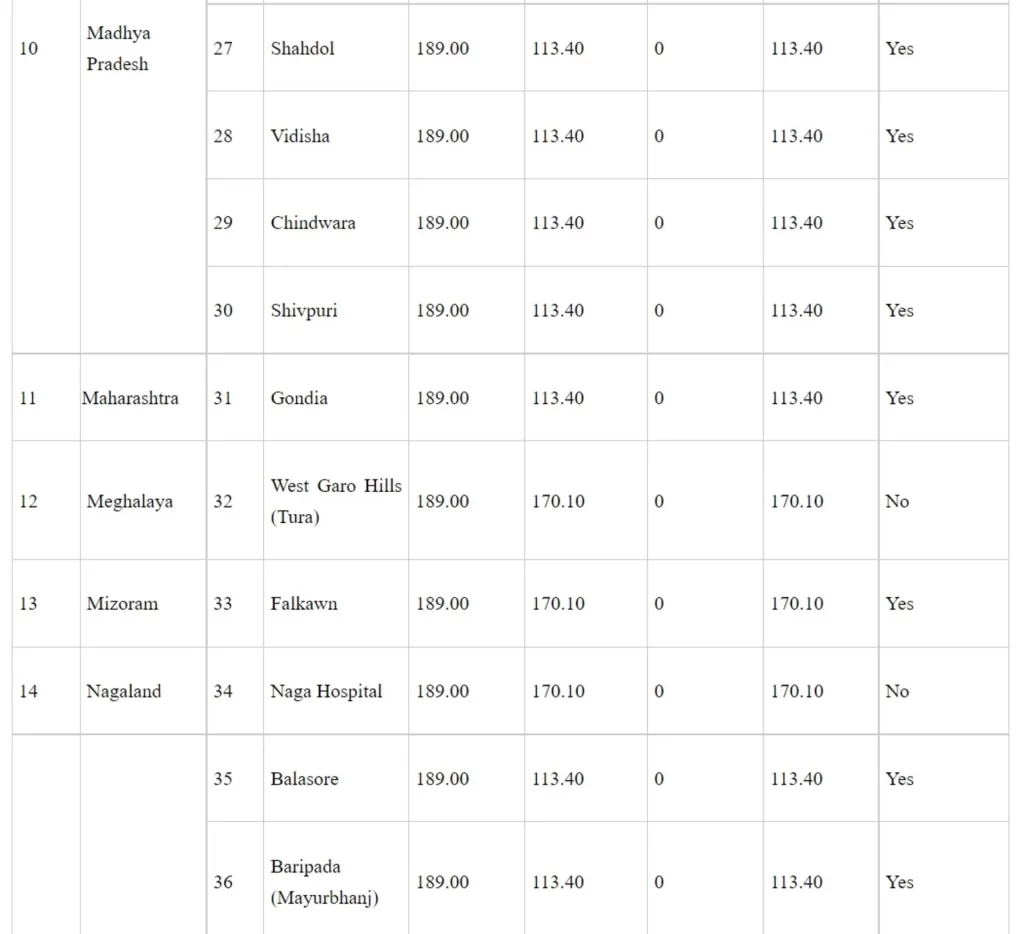
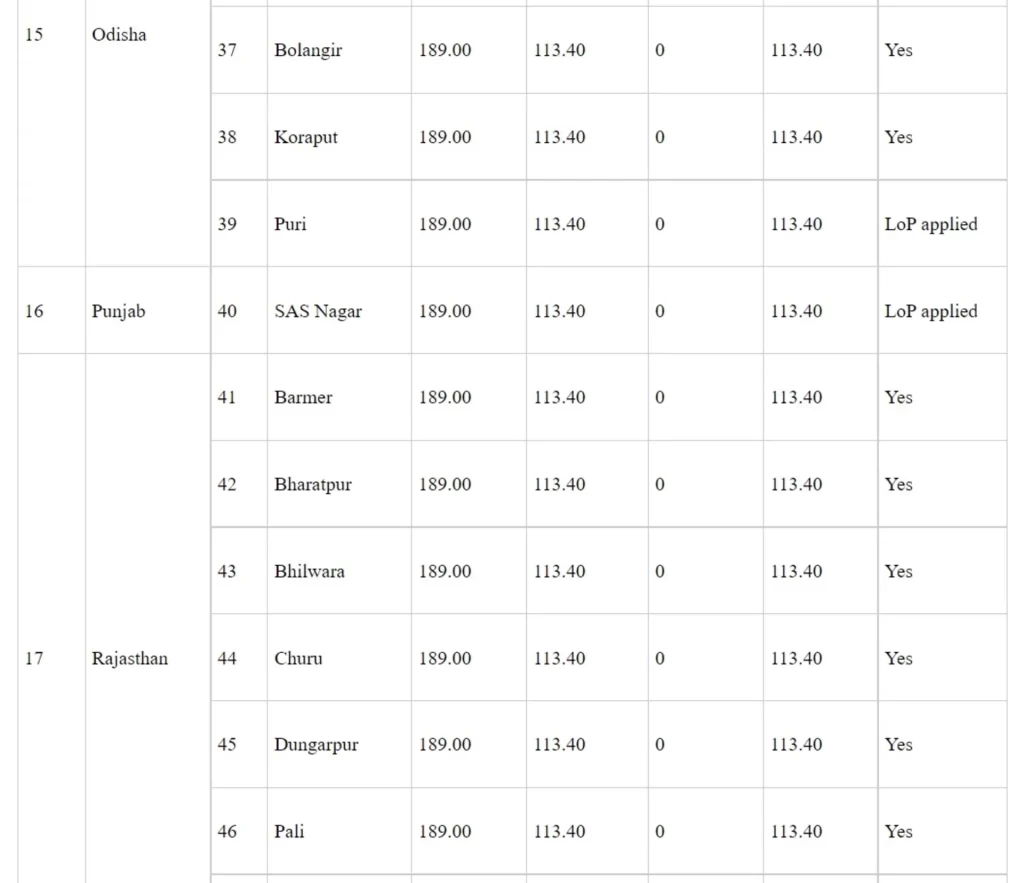
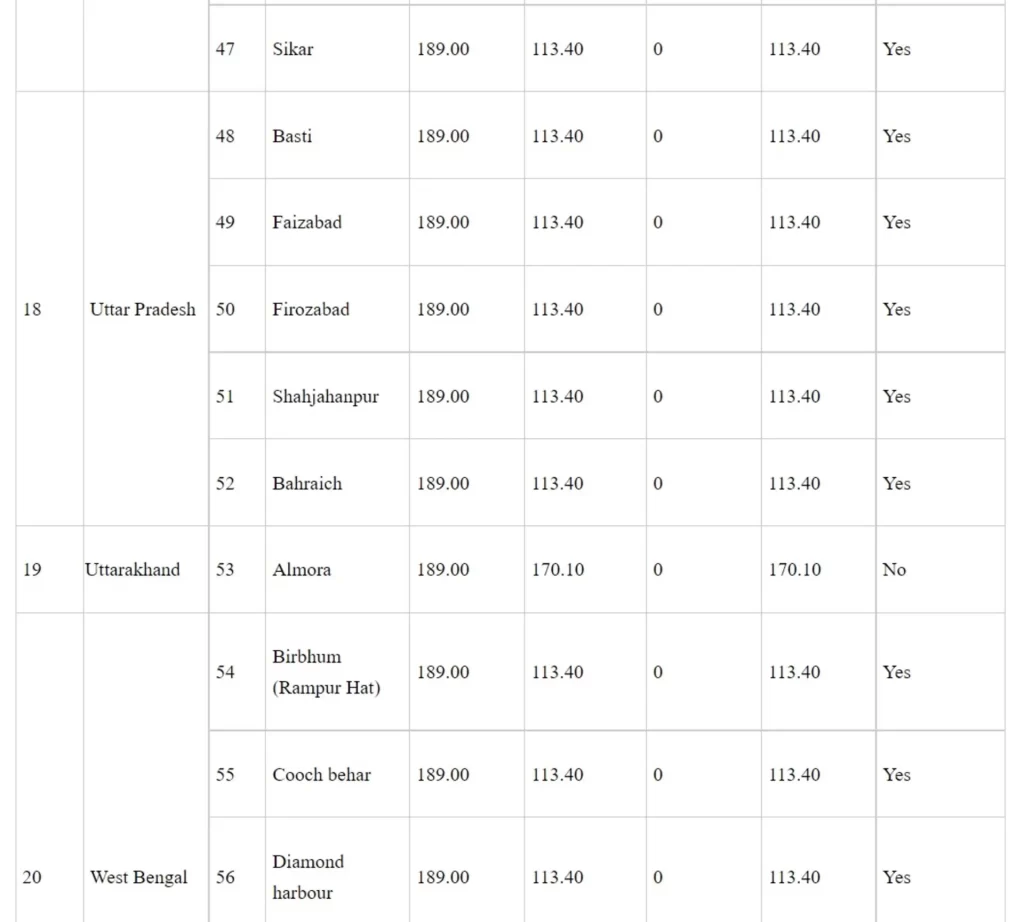
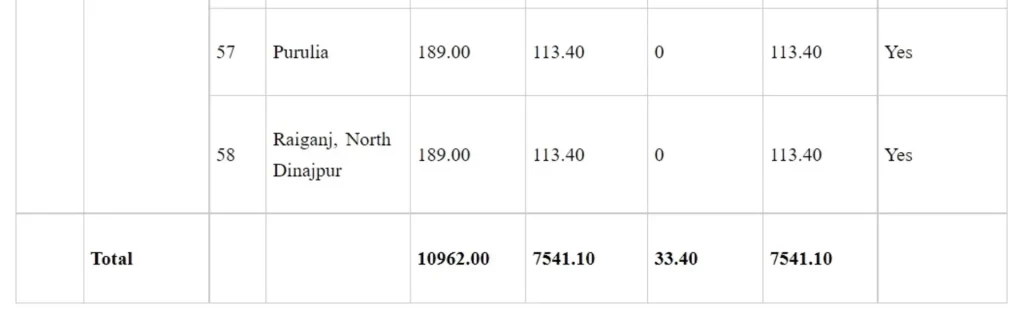
*LoP applied: These medical colleges have applied to the National Medical Commission for Letter of Permission (LoP)
The Minister of State (The Ministry of Health and Family Welfare), Dr. Bharti Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
Union Ministry of Health & Family Welfare has rushed a multidisciplinary team to Maharahstra to monitor the Zika virus situation and support the State government in management of Zika cases. A case of Zika has been reported in Pune district recently.
The three member Central team consists of a public health expert from the office of Regional Director, Pune; a gynecologist from Lady Hardinge Medical College, New Delhi; and an entomologist from National Institute of Malaria Research (NIMR), ICMR, New Delhi.
The team shall work closely with the State Health Department, take a stock of the situation on ground and assess whether the Union Health Ministry’s Action Plan for Zika Management is being implemented, and recommend necessary public health interventions for management of Zika in the state.
To address the healthcare challenges, particularly in rural areas, the National Rural Health Mission (NRHM) was launched in 2005 to supplement the efforts of the State/UT Governments to provide accessible, affordable and quality healthcare to all those who access public health facilities including tribal, backward, slum areas. Currently, NRHM is a sub-mission of National Health Mission (NHM).
Under the National Health Mission (NHM), financial and technical support is provided to States/UTs to strengthen their health care systems including for setting-up/upgrading public health facilities and augmenting health human resource on contractual basis for provision of equitable, affordable healthcare to all its citizens including the people of tribal, backward and slum areas based on requirements posed by the States in their Programme Implementation Plans (PIPs).
Various interventions that are implemented for better healthcare in tribal areas and beneficiaries are as follows;
The population norms for setting up Health Facilities in tribal areas are relaxed. Against the population norms of 5,000, 30,000, and 1,20,000 for setting up of Sub Centre, PHC and CHC respectively, in tribal and desert areas its 3,000, 20,000 and 80,000. Relaxed population norms for Mobile Medical Units (MMUs) & Ambulances SC/ST households are covered under Ayushman Bharat, Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) for health coverage up to Rs 5 Lakh per family per year as per Socio Economic Caste Census (SECC).
Further, all tribal majority districts whose composite health index is below the State average have been identified as High Priority Districts (HPDs) and these districts receive more resources per capita under the NHM as compared to the rest of the districts in the State. These districts receive higher per capita funding, have enhanced monitoring and focussed supportive supervision and are encouraged to adopt innovative approaches to address their peculiar health challenges.
Due to above interventions under NHM, there has been 73% increase in Health Facilities available in Tribal areas as compared to 10% increase in all India.
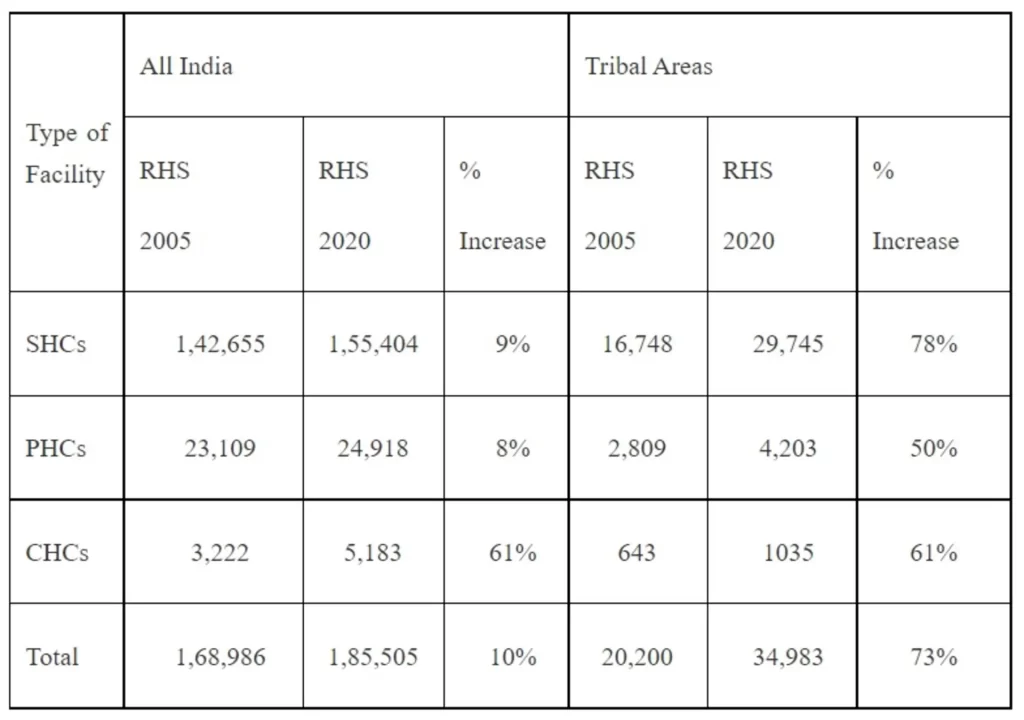
NHM support is also provided for provision of a host of free services in public healthcare facilities in tribal, backward and slum areas, related to maternal health, child health, adolescent health, family planning, universal immunisation programme, and for major diseases such as Tuberculosis, vector borne diseases like Malaria, Dengue and Kala Azar, Leprosy etc.
As part of Ayushman Bharat, the Government is supporting the States for transformation of Sub Health Centres and Primary Health Centres into 1.5 lakh Health and Wellness Centres across the country by December, 2022 for provision of Comprehensive Primary Health Care (CPHC) that includes preventive healthcare and health promotion at the community level with continuum of care approach. Under this programme, CPHC services of an expanded range of servicesthat are universal and free to users, with a focus on wellness, are provided, closer to the community. Further Ayushman Bharat, Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) provides health coverage up to Rs 5 Lakh per family per year to around 10.74 crore poor and vulnerable families as per Socio Economic Caste Census (SECC).
Financial support is also provided to States for providing hard area allowance, performance-based incentives, providing accommodation and transport facilities in rural and remote areas, sponsoring training programmes, etc to engaged human resources to address the issue of shortage of doctors and specialists in the public health facilities.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the LokSabha here today.
In a significant development, over 2.27 lakh pregnant women have received first dose of COVID-19 vaccine under the ongoing National COVID Vaccination drive. This can be seen as a result of regular counseling of the pregnant women by health workers and medical officers about risks of COVD-19 infection to pregnant women and benefits of COVID-19 vaccination. This sustained campaign has empowered pregnant women to make an informed choice on taking the COVID vaccination.
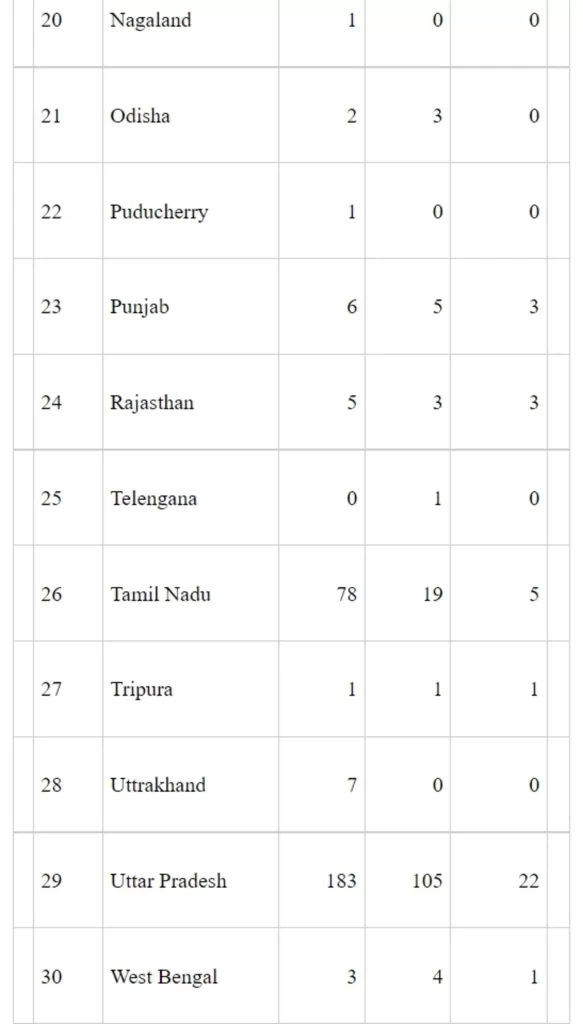
State of Tamil Nadu is leading the way by vaccinating over 78,838 pregnant women, followed by Andhra Pradesh with 34,228, Odisha with 29,821, Madhya Pradesh with 21,842, Kerala with 18,423 and Karnataka with 16,673 pregnant women vaccinated.
As part of the campaign to allay fears, apprehensions, misinformed rumours and some social taboos and issues, a guidance note to assist the States/UTs in operationalizing the COVID vaccination programme for pregnant women was shared by the Union Health Ministry on 2nd July 2021. This was followed up by training of the programme managers, service providers and frontline health workers to equip them to counsel the pregnant women and their family during antenatal care about importance of COVID-19 vaccination during pregnancy. The States/UTs further sensitized their vaccination teams in the Government and private COVID Vaccination Centres (CVCs) regarding COVID vaccination for pregnant women.
Several initiatives have been taken by States to encourage pregnant women to be motivated for the vaccine, such as special vaccination sessions for the pregnant women. Counselling the eligible beneficiaries by the frontline workers and counsellors at the antenatal clinics, mobilization by ASHAs for vaccination and innovative practices like issue of certificate by Deputy Commissioner to the pregnant and lactating women who were first to get themselves vaccinated have resulted in building confidence and acceptance in the community.

Studies have shown that COVID-19 infection during pregnancy may result in rapid deterioration of health of pregnant women, escalating the increased risk of severe disease and it might affect the fetus too. Pregnant women with COVID-19 infection are at an increased risk for preterm birth and other adverse pregnancy outcomes including higher chances of neonatal morbidity. Additionally, the experts have also highlighted pre-existing co-morbidities, advanced maternal age, and high Body Mass Index as factors for severe COVID-19 in pregnancy.
The National Technical Advisory Group on Immunization (NTAGI) had recommended vaccination for pregnant women leading to inclusion of pregnant women in India’s COVID Vaccination Drive. The National Expert Group on Vaccine Administration for COVID-19 (NEGVAC) had unanimously recommended it earlier. Additionally, a national level Consultation on COVID-19 vaccination for pregnant women was convened by Union Ministry of Health and Family Welfare to build consensus on COVID Vaccination of pregnant women. Professional bodies like FOGSI, representatives of State Governments, CSOs, NGOs, Development Partner agencies, technical experts etc., participated in this consultation. The recommendation of NTAGI to vaccinate pregnant women was unanimously welcomed. The combined effort of the Union Government, States / UTs and other stakeholders is helping in building confidence in the pregnant women and their families with increased uptake of COVID-19 vaccination thus, helping in protecting two lives from the risk of COVID-19 infection.
The Government of India has undertaken stem cell research through various Departments/ Institutions.
Department of Health Research-Indian Council of Medical Research (DHR-ICMR) is supporting biomedical research and stem cell is one of the priority areas promoted through centres for advance research/excellence, task force activities in identified thematic areas, viz. Basic stem cell biology, Cancer stem cell, Translational research and Generation of induced pluripotent stem cells (iPSC), open ended investigator initiated ad-hoc projects and fellowships for past twenty years. Many laboratories have been strengthened through above programmes.
Under Stem Cells & Regenerative Medicine, the Department of Biotechnology (DBT) has implemented projects on embryonic, adult and induced pluripotent stem cells (iPSC). Projects have been supported in areas of basic biology of stem cells; early and late translational research; developing gene editing technology for potential therapeutic applications; creation of animal models for various human diseases and training programmes on various components of stem cells and regenerative medicine. Studies have also been implemented to develop improved methods and techniques for isolating and culturing stem cells from different origin and also to generate induced pluripotent stem cells from various somatic/adult tissues.
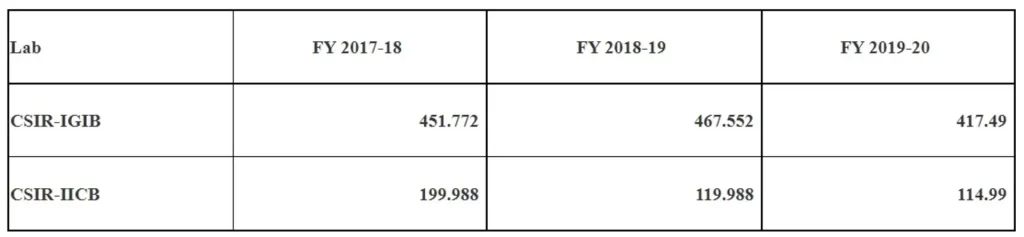
Council of Scientific and Industrial Research (CSIR), through its constituent laboratories namely CSIR-Institute of Genomics and Integrative Biology (CSIR-IGIB), New Delhi and CSIR-Indian Institute of Chemical Biology (CSIR-IICB), Kolkata has undertaken a Sickle Cell Anemia Mission during FY 2017-2020.
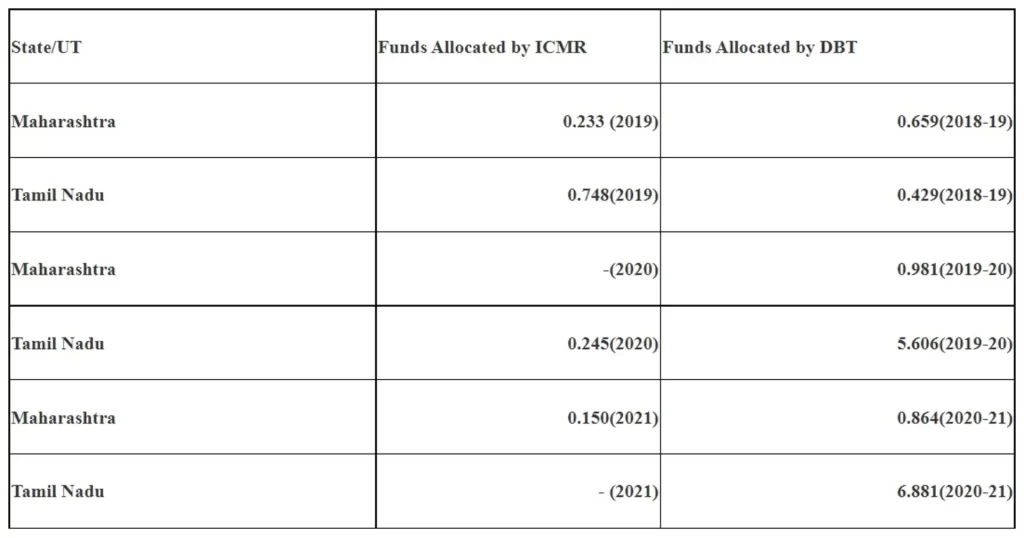
Financial assistance for stem cell research activities is being provided by the Government. Details of funds released to the States of Maharashtra and Tamil Nadu during last three years are as follows:-
Further, budget released by CSIR during the FY 2017-18 to 2019-20 under Sickle Cell Anemia Mission is as under:
Government has supported the ethical and scientific conduct of stem cell research through guidelines for stem cell research. These guidelines are available since 2007 and were revised in 2013 and again in 2017 based on the advancement in the field and released as National Guidelines for Stem Cell Research (NGSCR). This document provides guidance to all stakeholders including scientists, clinicians and industry and can be accessed online.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the LokSabha here today.
As per information from National Medical Commission (NMC), 90 medical colleges have been established during the last three years. State/UT-wise details are as below:
1 Arunachal Pradesh
TomoRiba Institute of Health & Medical Sciences, Narahlagun
2 Mizoram
Mizoram Institute of Medical Sciences Education & Research, Falkawn, Mizoram
3 Himachal Pradesh
Dr. Radhakrishnan Government Medical College, Hamirpur, Himachal Pradesh
4 Madhya Pradesh
Govt. Datia Medical College, Datia
5 Maharashtra
Government Medical College, Jalgaon, Maharashtra
6 Odisha
Govt. Medical College & Hospital. Balasore, Odisha
7 Odisha
Govt. Medical College & Hospital, Balangir, Odisha
8 Rajasthan
Govt. Medical College, Bhilwara
9 Rajasthan
Govt. Medical College, Bharatpur, Rajasthan
10 Rajasthan
Govt. Medical College, Churu, Rajasthan
11 Rajasthan
Govt. Medical College, Dungarpur, Rajasthan
12 Rajasthan
Govt. Medical College, Pali, Rajasthan
13 Telangana
Govt. Medical College, Siddipet, Telangana
14 Madhya Pradesh
Govt. Medical College, Ratlam
15 Madhya Pradesh
Govt. Medical College, Vidisha
16 Madhya Pradesh
Govt. Medical College, Khandwa
17 Maharashtra
AIIMS, Nagpur
18 Andhra Pradesh
AIIMS, Mangalgiri
19 Uttar Pradesh
Mulayam Singh Yadav Medical College & Hospital, Meerut, U.P.
20 West Bengal
Jagannath Gupta Institute of Medical Sciences & Hospital, Buita, Budge Budge , Kolkata, West Bengal
21 Telangana
Ayaan Institute of Medical Sciences, Teaching Hospital & Research Centre, Kanaka Mamidi, Moinabad Mandal, R.R. District, Hyderabad, Telangana
22 Gujarat
Zydus Medical College, Dahod, Gujarat
23 Gujarat
Banas Medical College & Research Institute, Palanpur, Gujarat
24 Dadar & Nagar Haveli
Shri VinobhaBhave Institute of Medical Sciences & Research, Dadra AND Nagar Haveli
25 Jammu & Kashmir
Govt. Medical College, Anantnag, Jammu & Kashmir
26 Jammu & Kashmir
Govt. Medical College & Hospital, Kathua, Jammu (J & K)
27 Jammu & Kashmir
Govt. Medical College , Baramulla, Kashmir
28 Jammu & Kashmir
Govt. Medical College , Rajouri , Kashmir
29 Karnataka
Bowring & Lady Curzon Medical College & Research Institute, Shivajinagar, Bangalore
30 Madhya Pradesh
Govt. Medical College, Chhindwara , M.P.
31 Madhya Pradesh
Govt. Medical College, shahdol, M.P.
32 Madhya Pradesh
Govt. Medical College, Shivpuri, Madhya Pradesh.
33 Maharashtra
Government Medical College, Baramati, Maharashtra
34 Rajasthan
Govt. Medical College, Barmer, Rajasthan
35 Tamil Nadu
Govt. Medical College, Sanapiratti Village KarurDistt., Tamil Nadu
36 Telangana
Govt. Medical College, Nalgonda, Telangana
37 Telangana
Govt. Medical College, Suryapet, Telangana
38 Uttar Pradesh
Govt. Allopathic Medical College, Faizabad, U.P.
39 Uttar Pradesh
Govt. Medical College, Shahjahanpur, U.P.
40 Uttar Pradesh
Government Institute of Medical Sciences, Greater Noida, U.P.
41 Uttar Pradesh
Govt. Medical College, Badaun, Uttar Pradesh
42 Uttar Pradesh
Govt. Medical College, Firozabad
43 Uttar Pradesh
Rajkiya Allopathic Medical College, Bahraich, KDC College Road, Bahraich, Uttar Pradesh
44 West Bengal
Diamond Harbour Govt. Medical College & Hospital, West Bengal
45 West Bengal
Raiganj Govt. Medical College & Hospital, Raiganj, West Bengal
46 West Bengal
Cooch Behar Govt. Medical College & Hospital, Cooch Behar, West Bengal
47 West Bengal
Rampurhat Govt. Medical College & Hospital, Rampurhat, West Bengal
48 Uttar Pradesh
Rajkiya Allopathic Medical College, Ramnagar, Basti, U.P.
49 Jharkhand
Palamu Medical College, Medininagar, Palamu
50 Jharkhand
Hazaribagh Medical College, Hazaribah
51 Jharkhand
Dumka Medical College, Dumka
52 West Bengal
AIIMS, Kalyani
53 Uttar Pradesh
AIIMS, Gorakhpur
54 Uttar Pradesh
AIIMS, Raibareli
55 Telangana
AIIMS, Bibinagar
56 Punjab
AIIMS, Bhatinda
57 Jharkhand
AIIMS, Deoghar
58 Bihar
Lord Buddha Medical College& Hospital, Saharsa
59 Bihar
Madhubani Medical College, Madhubani , Bihar
60 Gujarat
Shantabaa Hospital and Medical College, civil Hospital, Campus, Amreli, Gujarat.
61 Gujarat
Dr. N.D. Desai Faculty of Medical Science and Research, Dharmsinh Desai University Campus, Nadiad, Gujarat
62 Gujarat
NootanSarvaVidhyalayaKelavani Mandal, Visnagar
63 Haryana
Al Falah School of Medical Science & Research Centre, Village, Dhauj, Tikri , KhedaFatehpurTaga Road, Faridabad, Haryana
64 Karnataka
Sri Siddhartha Institute of Medical Sciences & Research Centre, T-Bengur, NH4, Nelamangala Taluk, Bangalore Rural District-562123.
65 Tamil Nadu
Kovai Medical Center & Hospital Ltd., Coimbatore
66 Telangana
Dr. PatnamMahender Reddy Institute of Medical Sciences, Chevella Village, Ranga Reddy Distt. Telangana
67 Telangana
Surabhi Institute of Medical Sciences, Siddipet, Distt. Medak, Telangana
68 Telangana
Mamata Academy of Medical Sciences, Bachupally, Medchal-Malajgiri, Districts, Hyderabad
69 West Bengal
Shri Ramkrishna Institute of Medical Sciences and Sanaka Hospitals, Malandighi, Durgapur , West Bengal
70 Assam
Assam Hills Medical College & Research Institute , Diphu, Assam
71 Bihar
JannayakKarpoori Thakur Medical College & Hospital, Madhepura, Bihar
72 J & K
Govt. Medical College, Doda, Kashmir
73 Maharashtra
Govt. Medical College, Nandurbar, Maharashtra
74 Rajasthan
Shri Kalyan Govt. Medical College, Sikar , Rajasthan
75 West Bengal
Purulia Govt. Medical College & Hospital, Purulia, West Bengal
76 Bihar
Netaji Subhas Medical College & Hospital, Amhara, Bihta, Patna
77 Gujarat
Dr. Kiran C. Patel Medical College & Research Institute, Bharuch
78 Jharkhand
Manipal Tata Medical College, Baridih, Jamshedpur, Jharkhand
79 Karnataka
Dr. Chandramma Dayananda Sagar Institute of Medical Education & Research, Harohalli, Karnataka
80 Madhya Pradesh
Mahaveer Institute of Medical Sciences & Research, Badwai, Bhopal
81 Maharashtra
SindhudurgShikshanPrasarak Mandal Medical College & Lifetime Hospital, Maharashtra
82 Maharashtra
DattaMeghe Medical College, Wanadongri, Hingna, Nagpur
83 Maharashtra
Symbiosis Medical College for Woman, Pune, Maharashtra
84 Punjab
GianSagar Medial College & Hospital, Ramnagar, Rajpura, Distt. Patiala.
85 Tamil Nadu
Indira Medical College & Hospitals, Thiruvallur Taluk & District, Tamil Nadu
86 Tamil Nadu
Panimalar Medical College Hospital & Research Institute Varadharajapuram, Poonamallee, Chennai, Tamil Nadu
87 Tamil Nadu
Bhaarath Medical College & Hospital, Agaram Village, Selaiyur, Chennai, Tamil Nadu
88 Telangana
TRR Institute of Medical Sciences, Patancheru, Medak Dist., Telangana
89 Uttar Pradesh
United Institute of Medical Sciences, District Allahabad, Uttar Pradesh
90 Uttar Pradesh
Noida International Institute of Medical Sciences, GautamBhudh Nagar, Uttar Pradesh
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Lok Sabha here today.
The Government of India has issued Telemedicine Practice guidelines on 25th March, 2020 which provide a robust framework for practice of telemedicine.
(https://www.mohfw.gov.in/pdf/Telemedicine.pdf).
These guidelines comprehensively prescribe norms and protocols covering all aspects of telemedicine practice like physician-patient relationship; issues of liability and negligence; management and treatment; informed consent; continuity of care; medical records; privacy and security of the patient records, exchange of information etc.
The guidelines also provide detailed information on technology platforms & tools to be utilized for effective health care delivery. Further, Ministry of Health and Family Welfare (MoHFW) has also developed a telemedicine application known as National Telemedicine Service, eSanjeevani to initiate OPD services free of cost to all citizens. This application works on a hub and spoke model to provide telemedicine services for citizen to doctor and doctor to doctor consultation. This application has also been integrated with 3.74 lakh Common Service Centers (CSCs) thereby facilitating access to equitable health care in the remotest areas of the country.
To ensure that only valid health professionals are able to provide services under eSanjeevani, the telemedicine practitioners are first vetted by the State Nodal Officer for eSanjeevani before being onboarded.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the LokSabha here today.
