Indian Council of Medical Research (ICMR) has informed that development of chronic diseases like diabetes, heart diseases & liver diseases are multi-factorial in nature and eating of junk food and/ or processed food is one of these factors. Various studies indicate that products with high content of fat, sugar or salt may increase the risk of overweight, obesity or certain Non-Communicable Diseases (NCDs).
Junk foods are high in calorie but low in nutritional value and lead to an excess metabolic weight leading to obesity. An obese individual is prone to life-threatening diseases which are not only limited to cholesterol or diabetes but also can cause stroke and other NCDs.
According to the study report "India: Health of the Nation's States"-The India State-Level Disease Burden Initiative 2017, the proportion of Chronic Diseases (NCDs) have increased in India from 30.5% in 1990 to 55.4 % in 2016. The overall metabolic risk factors / causes attributable to the rise in Chronic Diseases (NCDs) include unhealthy diet as per study.
Food Safety & Standards Authority of India (FSSAI) has informed that a nation-wide media campaign titled 'Aj Se Thoda Kam', to encourage consumers to make dietary modifications by gradually reducing the consumption of fat, sugar and salt has been launched. A series of short videos (with subtitles in 12 languages) have been created for the same. The campaign is supported with flyers, banners, audio clips and an ‘Eat Right India’ website with useful inputs on gradual reduction in consumption of fat, salt and sugar.
ICMR-NIN (National Institute of Nutrition) along with FSSAI has recommended guidelines for High Fat, Salt, Sugars (HFSS) Food Labels on all Ready to Eat Foods so that consumption of these foods is moderated.
Several awareness initiatives have been undertaken by the Government including use of print, electronic and social media. In addition, the national Programme for Prevention and Control of Cancer, Diabetes, Cardiovascular Diseases and Stroke (NPCDCS) supports awareness generation activities to be undertaken by the States.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the LokSabha here today.
The Government has undertaken several initiatives to promote transparency, foster ease of compliance in the export system and to facilitate exports, especially from MSMEs and small businesses with minimal capital. These include the following:
Directorate General of Foreign Trade (DGFT), under the Ministry of Commerce & Industry, has introduced multiple digital reforms to simplify and modernize foreign trade processes. The DGFT portal enables seamless online application and approval for various Foreign Trade Policy (FTP) benefits, including Advance Authorisation, Export Promotion Capital Goods (EPCG), and Status Holder certification. The portal is integrated with ICEGATE, GSTN, MCA, and PFMS for efficient data validation and processing.
Other key initiatives taken by DGFT include the self-certification mechanism for eBRCs, and the digitized e-Certificate of Origin (eCoO 2.0) system, enabling real-time authentication 2 and reducing manual burden. The revamped e-Certificate of Origin (eCoO) 2.0 system, operational for both Preferential and Non-Preferential CoOs, enables end-to-end digital issuance with QR code and Aadhaar-based e-signatures. It connects exporters with issuing agencies and chambers of commerce on a single online platform, thereby improving authentication and service delivery.
In order to assist Indian exporters, the Ministry has also launched the Trade Connect ePlatform (https://www.trade.gov.in/), as one-stop interface for addressing trade queries and enhancing access to international markets. Key features of the ePlatform include information about the Trade Agreements and provision of a Tariff Explorer to assess FTA benefits, Country and Product Guides for market access and compliance insights, Global e-Commerce Guide, and information on Trade Events Worldwide. Additionally, the platform hosts EXIM Paathshaala (learning modules), the “Ask an Expert” feature for resolving trade-related queries, the “Source from India” discovery tool for foreign buyers in order to connect to verified Indian exporters, and the unified Certificate of Origin issuance system for Indian exporters.
Further, in order to strengthen stakeholder interaction, the Ministry has operationalized the Jan-Sunwai facility, a digital grievance redressal platform that provides on-demand video conferencing between exporters and government officials. The system ensures real-time grievance handling, direct access to officers, and promotes transparency through audit trails.
Further, benefits of availing export promotion schemes under Chapters 4 and 5 of the Foreign Trade Policy (FTP) 2023 are available to all eligible exporters, including small businesses, third-party exporters, and merchant exporters. Therefore, in order to promote inclusive access and ease of compliance for such entities, the Government has introduced several supportive measures such as reduced application fees for MSMEs applying for benefits under export promotion schemes viz. Advance Authorisation and Export Promotion Capital Goods (EPCG).
The Niryat Bandhu Scheme, a flagship outreach initiative by DGFT, is designed to support and empower new and small exporters by providing them with expert guidance, mentorship, and handholding support throughout their export journey. This scheme aims to facilitate their entry into the international trade and enhance India's overall export competitiveness.
These initiatives have enhanced accessibility for small exporters while reducing procedural burdens and manual intervention.
The Government has taken measures to improve ease of compliance in the export system for all types of exporters, including exporters in agro-focused regions such as Erode. Developing District as Export Hub Initiative under Chapter 3 of FTP 2023 identifies the products/services (including GI products, agricultural clusters and toy clusters) with export potential in all the districts of the country. Institutional mechanism in the form of State Export Promotion Committees (SEPC) at State/UT level and District Export Promotion Committees (DEPCs) at the district level has been created in all districts of the country to provide support for export promotion and address the bottlenecks for export growth in the districts.
This information was given by the Minister of State for Ministry of Commerce & Industry, Shri Jitin Prasada, in a written reply in the Lok Sabha today.
Dr. Harsh Vardhan, Union Minister of Health and Family Welfare reviewed the preparedness for prevention and control of Vector Borne Diseases (VBD)with the UT of Delhi today in the presence of Shri Ashwini Kumar Choubey, Minister of State for Health and Family Welfare.Lt. Governor Shri Anil Baijaland Health Minister (Delhi) Shri Satyendar Jain were also present along with Mayors of three Municipal Corporations- ShriRaja Iqbal Singh (North Delhi Municipal Corporation), Shri Mukesh Suryan (South Delhi Municipal Corporation),Shri Shyam Sunder Aggarwal (East Delhi Municipal Corporation)and Shri Naresh Kumar, Chairperson, New Delhi Municipal Council.

The main focus of the meeting was geared towards the elimination of Malaria from Delhi by the year 2022. Dr. Harsh Vardhan emphasized on the need to make Malaria a notifiable disease so that each and every case of Malaria could be tracked and treated and the transmission foci could be identified. This issue was taken up with the UT govt. last year also, he stated.
Reminding everyone that Delhi is a Category 1 State for Malaria elimination, Dr. Harsh Vardhan said, “Delhi had originally planned to eliminate Malaria in 2020. The deadline has been now shifted to 2022.”
On the critical step of making Malaria notifiable in Delhi, he explained, “Capturing actual data from the Hospitals and taking preventive actions in the areas of infection is critical to achieve elimination. So is the regular reporting from the private sector essential for taking timely preventive measures and for accuracy of data pertaining to incidence of malaria. Further,as we move towards elimination, 10% of all fever cases need to be screened for malaria so that no cases of malaria can escape detection.” To encourage the administrators present, the Union Minister said, “India’s feat of achieving a reduction of 83.34% in Malaria morbidity and 92% in Malaria mortality between the year 2000 and 2019 has been acknowledged by WHO.”
The main component of VBD control programmebeing elimination and reduction in the risk of transmission and outbreak containment by stringent control of vector organisms, Dr. Harsh Vardhan said, “No effective drug or vaccine against Dengue & Chikungunya is available yet, so prevention is mainly focused on controlling of vector mosquito. Vector control like application of larvicides in breeding habitats can be carried out simultaneously with COVID19 activities. Precautions should be taken if fogging is undertaken, as COVID19 patients in Hospitals and quarantined at home or other places may experience breathing problem.”
He was apprised by the UT officers that the introduction of larvae eating fishes like Gambusia and Guppy have showed promising results. He advised the municipal authorities present to inspect buildings that are under construction to ensure that there is no breeding of vectors in these sites.
A planned preparation against vector borne diseases was elaborated upon:
· Adequate quantity of test kits for diagnosis of Dengue, chikungunya and malaria cases are to be made available in all hospitals.
· All the Hospitals are to prepare for ensuing transmission season with a contingency plan for Dengue. Regular testing of samples needs to be ensured for early warning signal.
· Blood banks need to be alerted to be ready with adequate blood components.
· Sensitization of the clinicians and laboratories on VBDs is also to be undertaken.
· Logistics for vector control including equipment and insecticides are to be ensured at all levels.
Dr. Harsh Vardhan stressed on community empowerment for their pro-active and effective contribution in vector control activitiesso that timely and appropriate action can be taken. The Union Minister highlighted that prevention and control of VBDs necessitates an effective inter-sectoral approach, requiring close collaboration and partnerships between the health and non-health sectors (both government and private), non-governmental organizations (NGOs) and local communities. He invited partners organisations to work in a coordinated way to prevent Dengue, chikungunya and malaria amidst COVID19, in order to prevent mortality and morbidity.
Shri Ashwini Kumar Choubey said, “Since the elimination of breeding spots for vector constitutes the pillar of Public Health response to VBDs, greater focus should be on community engagement through multi-media IEC campaigns.” He added that making Delhi Malaria free would put another feather on the cap of the country.
Shri Anil Baijal, Lt. Governor assured that the process of making Malaria a notifiable disease is already underway.
Shri Anil Baijalalso informed about the important efforts being put by the administration to stop VBDs: “We can’t afford to have an outbreak of Malaria, Dengue or Chikungunya during COVID times. The three Municipal Corporations have been onboarded who are to spearhead the action on controlling the vector in their localities. Since a major success was achieved in the past through educating children who conveyed vector control information to their parents, the Education Department has been instructed to include this IEC topic in their online education.”

Shri Rajesh Bhushan, Union Health Secretary, Ms. Rekha Shukla, Jt. Secretary (Health), Dr. Sunil Kumar, DGHS, Dr. Sujeet Singh,Director, NCDC, Dr Neeraj Dhingra, Director, NVBDCPand other senior officials of the Ministry were present at the event along with the Director/Medical Superintendents of 4 Central Government Hospitals –Dr. Randeep Guleria, Director, AIIMS, Dr. Rana A. K. Singh, MS, RML Hospital, Dr. S. V. Arya, MS, Safdarjung Hospital and Dr. Aparna Agarwal, Director, LHMC.
Sh. Vikram Dev Dutt, Principal Secretary (H&FW), NCT of Delhi along with the Commissioners and Medical Health Officers of North, South and East Municipal Corporations,The District Magistrates, Deputy Superintendents of Police of all of Delhi’s districts and Heads/Medical Superintendents of Delhi Hospitals dedicated to VBDs attended the event.
Under the visionary guidance of the Prime Minister Shri Narendra Modi, Ministry of Health and Family Welfare has taken a historic and a landmark decision for providing 27% reservation for OBCs and 10% reservation for Economically Weaker Section (EWS) in the All India Quota (AIQ) Scheme for undergraduate and postgraduate medical / dental courses (MBBS / MD / MS / Diploma / BDS / MDS) from the current academic year 2021-22 onwards.
Hon'ble Prime Minister in a meeting held on 26th July (Monday),2021 had directed the concerned Union Ministries to facilitate an effective solution to this long pending issue.
This decision would benefit every year nearly 1500 OBC students in MBBS and 2500 OBC students in postgraduation and also around 550 EWS students in MBBS and around 1000 EWS students in postgraduation.
The All India Quota (AIQ) Scheme was introduced in 1986 under the directions of the Hon’ble Supreme Court to provide for domicile-free merit based opportunities to students from any State to aspire to study in a good medical college located in another State. All India Quota consists of 15% of total available UG seats and 50% of total available PG seats in government medical colleges. Initially, there was no reservation in AIQ Scheme up to 2007. In 2007, the Hon’ble Supreme Court introduced reservation of 15% for SCs and 7.5% for STs in the AIQ Scheme. When the Central Educational Institutions (Reservation in Admission) Act became effective in 2007 providing for uniform 27% reservation to OBCs, the same was implemented in all the Central Educational Institutions viz. Safdarjung Hospital, Lady Harding Medical College, Aligarh Muslim University and Banaras Hindu University etc. However, this was not extended to the AIQ seats of State medical and dental colleges.
The present Government is committed to providingdue reservation both to the backward category as well as the EWScategory. The Union Government has now taken a historic decision to provide for 27% reservation for OBCs and 10% reservation for EWS in the AIQ Scheme. The OBC students from across the country shall now be able to take benefit of this reservation in AIQ Scheme to compete for seats in any State. Being a Central Scheme, the Central List of OBCs shall be used for this reservation. Around 1500 OBC students in MBBS and 2500 in postgraduation will be benefitted through this reservation.
In order to provide benefit to students belonging to EWS category in admission to higher educational Institutions, a Constitutional amendment was made in 2019 which enabled the provision of 10% reservation for EWS category. Accordingly, seats in medical / dental colleges were increased over two years in 2019-20 and 2020-21 to accommodate this additional 10% EWS reservation so that the total number of seats available for unreserved category do not reduce. In the AIQ seats, however, this benefit had not been extended so far.
Therefore, alongwith the 27% reservation for OBCs, 10% reservation for EWS is also being extended in AIQ seats for all the undergraduate / postgraduate medical/dental courses from the current academic year 2021-22. This will benefit every year around more than 550 EWS students for MBBS and around 1000 EWS students for PG medical courses.
The above decision is the reflection of the Government’s commitment to provide due reservation for backward and EWS category students.
This decision is also in sync with the significant reforms carried out in the field of medical education since 2014. During the last six years, MBBS Seats in the country have increased by 56% from 54,348 seats in 2014 to 84,649 seats in 2020 and the number of PG seats have increased by 80% from 30,191seats in 2014 to 54,275 seats in 2020.During the same period, 179 new medical colleges have been established and now the country has 558 (Govt: 289, Pvt: 269) medical colleges.
The International Society for Pharmaceutical Engineering (ISPE) is a not-for-profit association serving its Members through leading scientific, technical, and regulatory advancement across the entire pharmaceutical lifecycle. The 18,000 Members of ISPE are building solutions in the development and manufacture of safe, effective pharmaceutical and biologic medicines, and medical delivery devices in more than 90 countries around the world. Founded in 1980, ISPE has its worldwide headquarters and training center in North Bethesda, Maryland, USA, and its operations center in Tampa, Florida, USA. Visit ISPE.org for more information.
The governmentlaunched the National Policy for Rare Diseases (NPRD), 2021, in March 2021 for the treatment of rare disease patients. The salient features of NPRD, 2021, are as follows:
The rare diseases have been identified and categorized into 3 groups, namely Group 1, Group 2, and Group 3.
Group 1: Disorders amenable to one-time curative treatment.
Group 2: Diseases requiring long-term/lifelong treatment having relatively lower cost of treatment and benefit have been documented in literature, and annual or more frequent surveillance is required.
Group 3: Diseases for which definitive treatment is available but challenges are to make optimal patient selection for benefit, very high cost, and lifelong therapy.
Provision for financial support of up to Rs. 50 lakhs to the patients suffering from any category of the rare diseases and for treatment in any of the Centers of Excellence (CoE) mentioned in NPRD-2021, outside the Umbrella Scheme of Rashtriya Arogaya Nidhi.
In order to receive financial assistance for treatment of a rare disease, the patient of the nearby area may approach the nearest Center of Excellence to get him assessed and avail the benefits.
Eight (08) Centers of Excellence (CoEs) have been identified for diagnosis, prevention, and treatment of rare diseases.
Five Nidan Kendras have been set up for genetic testing and counseling services.
The NPRD, 2021, has provisions for promotion of research and development for diagnosis and treatment of rare diseases; promotion of local development and manufacture of drugs; and creation of a conducive environment for indigenous manufacturing of drugs for rare diseases at affordable prices.
The Department of Pharmaceuticals has initiated the implementation of the Production Linked Incentive Scheme for Pharmaceuticals. The scheme provides for financial incentives to manufacturers selected under the scheme for domestic manufacturing of various product categories, which also include orphan drugs.
The Department of Revenue, Ministry of Finance, vide their Notification No. 46/2021-Customs dated 30.09.2021, gives a full waiver of Basic Customs Duty (BCD) and Integrated Goods and Services Tax (IGST) to drugs imported (personal use only) for the treatment of Spinal Muscular Atrophy (SMA), a rare disease, thereby making the medicines for SMA more affordable.
In addition, the Department of Revenue, Ministry of Finance, vide their Notification No. 02/2022-Customs dated 01.02.2022, has given exemption from Basic Customs Duty to drugs or medicines that are used in the treatment of rare diseases when imported by Centers of Excellence (CoEs) as specified in NPRD, 2021, or any person or institution on the recommendation of any Center of Excellence listed in NPRD, 2021, certifying that the person (by name) for whom the drugs or medicines are imported is suffering from a rare disease (to be specified by name) and requires the drugs or medicines for the treatment of said rare disease.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar, stated this in a written reply in the Lok Sabha today.
Minister of State for Chemicals and Fertilizers Shri Mansukh Mandaviya along with Minister of State for Home Affairs Shri G. Kishan Reddy today visited Bharat Biotech's vaccine manufacturing and Bio Safety Level-3 facility to review the production of Vaccines in Hyderabad. Secretary, Department of Pharmaceuticals, Ms S. Aparna was also present.

Speaking on the occasion Shri Mandaviya said that the Government under the leadership of Prime Minister Shri Narendra Modi is committed to support all our vaccine developers and manufactures for ensuring vaccine for all.
The Ministers also discussed ramping up the production of Vaccines with the Manufacturers.
Later in the day, Ministers visited Biological_E Ltd, the Vaccine developer of 'Corbevax'-One of the indigenous COVID-19 Vaccines of India.

They also had a meeting with Dr Reddy’s team regarding the status of single shot COVID-19 vaccine 'Sputnik Light' that will be imported from Russia. The team apprised the Ministers on the domestic manufacturing of Sputnik vaccine.
44.19 Cr. Vaccine Doses administered so far under Nationwide Vaccination Drive
3,06,21,469 total recoveries across the country so far
Recovery Rate currently at 97.39%
42,363 patients recovered during last 24 hours
India reports less than 30,000 daily cases after 132 days; 29,689 new cases in last 24 hours
India's Active caseload less than 4,00,000 after 124 days; currently at 3,98,100
Active cases constitute 1.27% of total cases
Weekly Positivity Rate remains below 5%, currently at 2.33%
Daily positivity rate at 1.73%, remains below 5%
Testing capacity substantially ramped up – 45.91 cr tests total conducted
An amount of Rs. 1113.21 crore and Rs. 8147.28 crore was released to States/UTs as Grant-in-aid under National Health Mission (NHM) for management and control of COVID-19 during financial year 2019-20 and under the “India COVID-19 Health System Preparedness and Emergency Response Package” during financial year 2020-21, respectively.
All States/UTs were requested to ensure that the incentive of Rs. 2000/- to ASHAs for the routine and recurring activities is continued to be paid in full during COVID-19 Pandemic. Further, States/UTs were also asked to continue to pay other task based incentives to ASHAs such as Janani Suraksha Yojana (JSY), Home Based Newborn Care (HBNC), etc., as per National/State norms during the course of this pandemic period as per the activities undertaken by them.
In view of the significant contribution towards the COVID-19 pandemic related work by ASHAs, States were advised to pay an additional incentive of Rs.1000/- per month for those ASHAs engaged in COVID-19 related work using the resources of “COVID-19 Health System Preparedness and Emergency Response Package” from January, 2020.
Under the Pradhan MantriGaribKalyan Package, Insurance Scheme has been introduced for all health workers, including ASHAs. This insurance scheme provides an insurance cover of Rs. 50.00 Lakhs in-case of loss of life on account of COVID-19 related duty.
As per reports received from States/UTs, a total no. of 8,78,071ASHAs, ASHA Facilitators and other community volunteers have received supplementary COVID-19 payment till March, 2021. No such delay has been reported by the States/UTs.
43 claims have been disbursed to ASHAs under Pradhan MantriGaribKalyan (PMGK) Package till June, 2021. No such delay in disbursements has been reported by the States/UTs.
ASHAs do not fall under the Code on Social Security and the Code of Wages, 2020. Theyare envisaged to be community health volunteer and are entitled to task/activity based incentives. ASHAs benefits include the following:
Pradhan Mantri Jeevan JyotiBeemaYojana (PMJJBY) with a benefit Rs. 2.00 Lakh in case of death of the insured (annual premium of Rs. 330 contributed by GOI).
Pradhan Mantri Suraksha BeemaYojana (PMSBY) with a benefit of Rs. 2.00 lakh for accidental death or permanent disability; Rs. 1.00 lakh for partial disability (annual premium of Rs. 12 contributed by GOI).
Pradhan MantriShram Yogi MaanDhan (PM-SYM) with pension benefit of Rs. 3000 pm after age of 60 years (50% contribution of premium by GOI and 50% by beneficiaries).
The Union Minister of State for Health and Family Welfare, DrBharati Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
The National Pharmaceutical Pricing Authority (NPPA) under the Department of Pharmaceuticals, as an on-going process, with the help of Price Monitoring & Resource Units (PMRUs) and State Drug Controllers (SDCs) undertake activities related to market-based data collection, monitoring of the notified prices of medicines and detection of violation of the provisions of Drug (Price Control) Order, 2013 (DPCO, 2013).
NPPA fixes the ceiling prices of scheduled formulations as per provisions of DPCO, 2013 and monitors the prices of non-scheduled drugs so as to ensure that their prices do not increase beyond 10 percent in a year. NPPA has fixed ceiling prices of 355 medicines and 882 formulations for medicines under National List of Essential Medicines, 2015 (Schedule-I of DPCO, 2013). Most of the drugs that are part of COVID management protocol have ceiling prices, viz., Paracetamol, Dexamethasone, Methyl Prednisolone, IVIGs, Enoxaparin, Budesonide, Heparin and Amphotericin etc. In the case of few non-scheduled medicines like Remdesivir, which are part of COVID protocol and are non-scheduled, on Government intervention, MRPs of various brands of Remdesivir have been reduced voluntarily by the major manufacturers/marketers of the Remdesivir Injection (lyophilized). Inter-brand MRPs that varied up to Rs 5400/ per vial were brought down to less than Rs 3500/-.
Furthermore, to facilitate availability and ensure affordability, NPPA has capped the trade margin for Oxygen Concentrators at 70% on Price to Distributor (PTD) level vide notification dated 3rd June, 2021 and on Pulse Oximeter, Glucometer, BP Monitor, Nebulizer and Digital Thermometer vide notification dated 13th July, 2021.
In addition, Retail prices for 1,640 formulations have been fixed under DPCO, 2013 till date and in recent years exercising extraordinary powers under DPCO, 2013 in public interest, NPPA has fixed the prices of following drugs and medical devices:
(i) Ceiling price of Stents in February, 2017
(ii) Ceiling price of Knee Implants in August, 2017
(iii) Capped trade margin on selected 42 anti-cancer drugs in February, 2019.
The information was given by the Union Minister of Chemicals and Fertilizers, Shri Mansukh Mandaviya in a written reply in the LokSabha today.
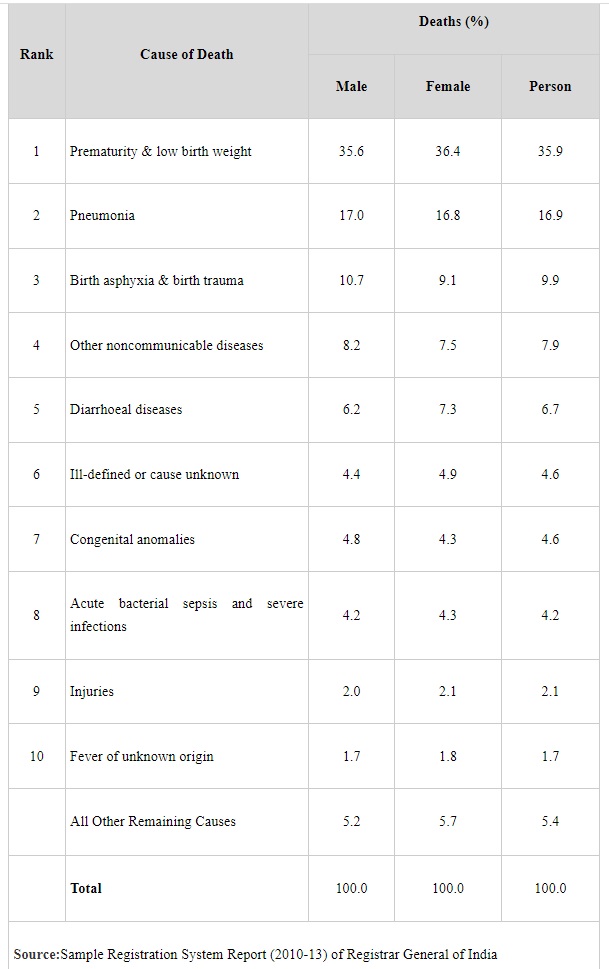
As per Sample Registration System Report (2010-13) of Registrar General of India, Pneumonia contributes 16.9% of infant deaths and it is the 2nd highest cause of infant mortality.
The list of major causes of infant mortality in India as per the above report is placed at annexure.
In order to accelerate actions for reducing deaths due to childhood Pneumonia, an initiative named “Social Awareness and Actions to Neutralize Pneumonia Successfully (SAANS)” has been launched in 2019. The SAANS initiative encompasses three pronged strategies:
Guidelines on treatment and management of childhood Pneumonia including introduction of use of Amoxicillin by ANM. Capacity building of service providers for identification and standardized management of Pneumonia. Communication campaign to ensure greater awareness on childhood Pneumonia among families and parents.
The Union Minister of State for Health and Family Welfare, Dr Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
