All beneficiaries who have received vaccines in India are registered on CoWIN portal. CoWIN portal is single source of truth for vaccination status.
As on 16th July 2021, a total of 3.48 lakh doses (0.09% of the total doses administered) have been administered to persons without Identity Cards. State/UT-wise details are at Annexure.
Registration and vaccination of people without access to digital technology can be done through:
Walk-in registration of either single individuals or groups of individuals at COVID-19 Vaccination Centre (CVC).
Registration at Common Service Centres
Registration of up to 4 persons using a single mobile number to facilitate registration of people without mobile phones.
Detailed SOP has been issued for vaccination of people without prescribed photo ID documents through key facilitators at identified Government CVCs.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
“Public Health and Hospitals” being a State subject, the primary responsibility of strengthening public healthcare system including provision of essential medicines lies with the respective State Governments. However, under National Health Mission (NHM), technical and financial support is provided to the States/UTs to strengthen the public healthcare system including support for ensuring sufficient supply of life saving medicines and other medical equipment at public healthcare facilities towards the objective of providing accessible, affordable and quality healthcare to all those who access public health facilities.
To ensure availability of essential drugs and reduce the Out-of-Pocket Expenditure (OOPE) of the patients visiting the public health facilities, Government has rolled out the Free Drugs Service Initiative (FDSI) under National Health Mission (NHM).
Under this, financial support is provided to States / UTs for provision of free essential medicines in public health facilities based on the requirements posted by them in their Programme Implementation Plans (PIPs) within their overall resource envelope.
Support under the scheme is available for Procurement of drugs and strengthening /setting up robust systems of procurement, Quality Assurance, Supply chain management and warehousing, Prescription audit, grievance redressal, dissemination of Standard Treatment Guidelines and Establishment of IT enabled platform DVDMS (Drugs & Vaccine Distribution Management System) for monitoring the real status of procurement and availability of essential medicines.
Ministry has recommended facility wise Essential Medicines List (EML) to be made available at the public healthcare facilities:
Sub Health Centre Health & wellness centre (SHC-HWCs)– 105
Primary Health Centre Health & wellness centre (PHC-HWCs)- 172
Community Health Centres (CHCs)- 455
District Hospital (DHs)- 544
However, States have the flexibility to add more medicines and Essential Drugs List (EDL) of States vary from State to State. Quality of drugs procured, under Free Drugs Service Initiative, is ensured through the operational guidelines of the initiative that
All drugs must be sourced from Good Manufacturing Practices (GMP) compliant manufacturers through robust procurement mechanism.
Post supply testing of every batch before distributing to the health facilities.
To address the healthcare challenges, particularly in rural areas, the National Rural Health Mission (NRHM) was launched in 2005 to supplement the efforts of the State/UT governments to provide accessible, affordable and quality healthcare to all those who access public health facilities. Currently, NRHM is a sub-mission of National Health Mission (NHM).
This support under NHM includes provision of a host of free services related to maternal health, child health, adolescent health, family planning, universal immunisation programme, and for major diseases such as Tuberculosis, HIV/ AIDS, vector borne diseases like Malaria, Dengue and Kala Azar, Leprosy etc.
Other major initiatives include Janani Shishu Suraksha Karyakram (JSSK) (under which free drugs, free diagnostics, free blood and diet, free transport from home to institution, between facilities in case of a referral and drop back home is provided), Rashtriya Bal Swasthya Karyakram (RBSK) (which provides newborn and child health screening and early interventions services free of cost for birth defects, diseases, deficiencies and developmental delays to improve the quality of survival), implementation of Free Drugs and Free Diagnostics Service Initiatives, PM National Dialysis Programme and implementation of National Quality Assurance Framework.
Mobile Medical Units (MMUs) & Telemedicine are also being implemented with NHM support to improve healthcare access particularly in rural areas.
As part of Ayushman Bharat, the Government is supporting the States for strengthening Sub Centres and Primary Health Centres as Health and Wellness Centres (AB-HWCs) for provision of comprehensive primary health care that includes preventive healthcare and health promotion at the community level with continuum of care approach.
Further, Ayushman Bharat, Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) provides health coverage up to Rs. 5.00 lakh per family per year to around 10.74 crore poor and vulnerable families as per Socio Economic Caste Census (SECC). In so far as implementation of PMJAY is concerned, public hospitals are reimbursed for the healthcare services at par with the private hospitals under the scheme.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
Life insurance benefits are being provided under Pradhan Mantri Garib Kalyan Package (PMGKP): Insurance Scheme for Health Workers Fighting COVID-19. The scheme provides a comprehensive personal accident cover of Rs. 50 lakhs to healthcare providers, including community health workers and private health workers drafted by the Government for COVID19 related responsibilities, who may have to be in direct contact and care of COVID-19 patients and who may be at risk of being impacted by it. The benefits under the said scheme have been extended for a further period of 180 days (w.e.f. 24.04.2021).
As on 15th July 2021, a total no. of 921 doctors and healthcare workers have been paid insurance claim 50 lakh each under the said scheme.
Further, realizing the criticality of human resources at the time of pandemic, Ministry of Health & Family Welfare has taken a number of steps to improve safety and wellbeing of medical officers and staff.
Few of the actions taken in this regard include:
In the context of COVID-19, the Epidemic Diseases (Amendment) Ordinance, 2020 was promulgated on 22nd April 2020. Further this ordinance was brought before the Parliament & was passed and notified on 29th September 2020. The amendment provides for safety and security of Health Care Service Personnel (HSPs) from acts of violence.
With the launch of COVID-19 vaccination initiative on 16th January 2021, Healthcare workers were identified as one of the first priority group. State Governments/ UT administrations have been repeatedly urged to ensure that the medical professionals engaged in Covid related work are suitably vaccinated.
Ministry of Health and Family Welfare provided guidelines on Infection Prevention and Control practices to the State Governments to minimize the risk of infection to healthcare workers.
Training on Infection Prevention and Control was also made available for all category of healthcare workers on the iGoT platform.
Guidelines on rational use of PPEs for hospital and community settings (including front line workers) were issued. These guidelines followed a risk-based approach and recommended type of PPE that needs to be used in various risk settings.
The healthcare workers were provided with hydroxychloroquine for prophylaxis and prevention of infection. An Advisory to that affect was also issued by MoHFW on 23rd March 2020.
For high-risk exposures, healthcare workers are being provided with a quarantine period initially for one week and thereafter taking the profile of the health worker a decision to be taken for further period of one week. An advisory to this effect for managing health care workers working in COVID and Non-COVID areas of the hospital was issued by Ministry of Health and Family Welfare on 18thJune, 2020.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
As per Central Drugs Standard Control Organisation (CDSCO), CDSCO has granted manufacturing permission to private manufacturers for following COVID-19 Vaccines for restricted use in emergency situation: -
ChAdOx1 nCoV- 19 Corona Virus Vaccine (Recombinant) manufactured by M/s Serum Institute of India Pvt., Ltd., Pune on 03.01.2021
Whole Virion Inactivated Corona Virus Vaccine manufactured by M/s Bharat Biotech International Limited, Hyderabad on 03.01.2021.
Gam-COVID-Vac Combined vector vaccine (SPUTNIK-V) manufactured by M/s Ra (biologicals) Panacea Biotec Ltd., New Delhi on 02.07.2021.
Department of Biotechnology has informed that to support vaccine manufacturing in India, the Government of India has launched ‘Mission COVID Suraksha- the Indian COVID- 19 Vaccine Development Mission’, being implemented by Biotechnology Industry Research Assistance Council (BIRAC), a Public Sector Undertaking (PSU) of Department of Biotechnology (DBT). Under the Mission, clinical trial lot manufacturing of promising vaccine candidates including DNA vaccine candidate (ZydusCadila); mRNA vaccine candidate (Gennova Biopharmaceuticals); intranasal vaccine candidate (Bharat Biotech) are being supported.
Further, as part of efforts for augmentation of Covaxin production, capacity enhancement of Bharat Biotech is being supported under Mission COVID Suraksha. Also, technology transfer of Covaxin production from Bharat Biotech to Gujarat COVID Vaccine Consortium (GCVC), comprising of Hester Biosciences and OmniBRx Biotechnologies Pvt Ltd, is being facilitated by the Department of Biotechnology.
The information was given by the Union Minister of Chemicals and Fertilizers, Shri Mansukh Mandaviya in a written reply in the Lok Sabha today.
“Public Health and Hospitals” being a State subject, the primary responsibility of strengthening public healthcare system, including for provision of quality healthcare and advanced treatment and diagnostic facilities, lies with the respective State Governments. However, under National Health Mission (NHM), technical and financial support is provided to the States/UTs to strengthen the public healthcare delivery at public healthcare facilities.
To address the healthcare challenges, particularly in rural areas, the National Rural Health Mission (NRHM) was launched in 2005 to supplement the efforts of the State/UT governments to provide accessible, affordable and quality healthcare to all those who access public health facilities. Currently, NRHM is a sub-mission of National Health Mission (NHM). NUHM was also launched in 2013 as a sub-mission of NHM.
NHM support is provided to States/ UTs for setting up of new facilities as per norms and upgradation of existing facilities for bridging the infrastructure gaps based on the requirement posed by them.
NHM support is also provided for provision of a range of free services related to maternal health, child health, adolescent health, family planning, universal immunisation programme, and for major diseases such as Tuberculosis, vector borne diseases like Malaria, Dengue and Kala Azar, Leprosy etc.
Other major initiatives supported under NHM include Janani Shishu Suraksha Karyakram (JSSK) (under which free drugs, free diagnostics, free blood and diet, free transport from home to institution, between facilities in case of a referral and drop back home is provided), Rashtriya Bal Swasthya Karyakram (RBSK) (which provides newborn and child health screening and early interventions services free of cost for birth defects, diseases, deficiencies and developmental delays to improve the quality of survival), implementation of Free Drugs and Free Diagnostics Service Initiatives, PM National Dialysis Programme and implementation of National Quality Assurance Framework in all public health facilities including in rural areas.
Mobile Medical Units (MMUs) & Tele-consultation services are also being implemented to improve access to healthcare particularly in rural areas.
As part of Ayushman Bharat, the States are supported for establishment of 1.5 lakh Health and Wellness Centres across the country by December, 2022 for provision of comprehensive primary care that includes preventive healthcare and health promotion at the community level with continuum of care approach. Further Ayushman Bharat, Pradhan Mantri Jan Arogya Yojana (AB-PMJAY) provides health coverage up to Rs 5 Lakh per family per year to around 1074 crore poor and vulnerable families as per Socio Economic Caste Census (SECC).
In the 15 years of implementation, the NHM has enabled achievement of the Millennium Development Goals (MDGs) for health. It has also led to significant improvements in maternal, new-born, and child health indicators, particularly for maternal mortality ratio, infant and under five mortality rates, wherein the rates of decline in India are much higher than the global averages and these declines have accelerated during the period of implementation of NHM. The major achievements under NHM are given at Annexure.
Improvements in Core Health Outcomes:
Reduction in OOPE: The average medical expenditure per hospitalization and childbirth also declined in this period, from Rs. 5636 to Rs. 4290 in rural areas, and Rs. 7670 to Rs. 4837 in urban areas for hospitalization and from Rs. 1587 to Rs. 1324 in rural areas and Rs. 2117 to Rs. 1919 in urban areas for institutional delivery, indicating the impact of NHM in reduction of Out of Pocket Expenditures for health care.
Maternal Mortality Rate (MMR) has declined from 556/lakh live births in 1990 to 113/lakh live births in 2016-2017. Rate of decline of MMR in India at 77% is much higher than global average decline of 44% over the same period.
Infant Mortality Rate (IMR), declined from 80 in 1990 to 32 in the year 20181.
Under 5 Mortality Rate (U5MR), declined from 52 in 2012 to 36 in 2018,
Total Fertility Rate (TFR) declined from 2.3 in 2013 to 2.2 in the year 20181.
The incidence of Tuberculosis per 1,00,000 population reduced from 234 in 2012 to 193 in 2019. The mortality due to TB per 1,00,000 population also reduced from 42 in 2012 to 33 in 2019.
The number of districts that achieved the target of leprosy elimination increased from 543 districts in 2011-2012 to 554 by March 2017 and to 571 districts by March 2018. The prevalence rate of Leprosy reduced to less than 1/10000 population in 610 Districts in 2020.
For Malaria, Annual Parasitic Index (API) was sustained at levels of less than 1 and declined from 0.89 in 2014 to 0.32 in 2018, and 0.25, in 2019. Malaria cases and deaths declined from 21.27% and 20% respectively in 2019 from 2018.
By the end of December 2019, 94% Kala-azar endemic blocks have achieved the elimination target of <1 KA case per 10,000 population at block level.
The National target for sustaining Dengue related case fatality rate to less than 1% was met. Dengue related case fatality rate in 2014 was 0.3% and reduced to 0.1% in 2019.
The prevalence of blindness (visual acuity <3/60) has reduced to 0.36% in 2019) from 0.68% in 2010.
There is a 17.3% relative reduction in prevalence of tobacco use from 34.6% to 28.6% from 2009-10 to 2016-17.
Increased Access to Health Services:
As a result of the health systems strengthening efforts of NHM, the proportion of those seeking care from public health facilities, increased from 28.3% to 32.5% in rural areas and 21.2% to 26.2% in urban areas between 2014 to 2017.; and utilization of public health facilities for institutional deliveries increased from 56% to 69.2% in rural areas and from 42% to 48.3% in urban areas.
Growth in Public Health Facilities:
NHM adopts a health system approach and targets to build a network for public health facilities with Health & Wellness Centres at the grassroot level and District Hospitals, with robust referral linkage, to offer Comprehensive primary and secondary care services to citizens. The growth in the number of facilities at various levels since 2025 is given in Table 1 -
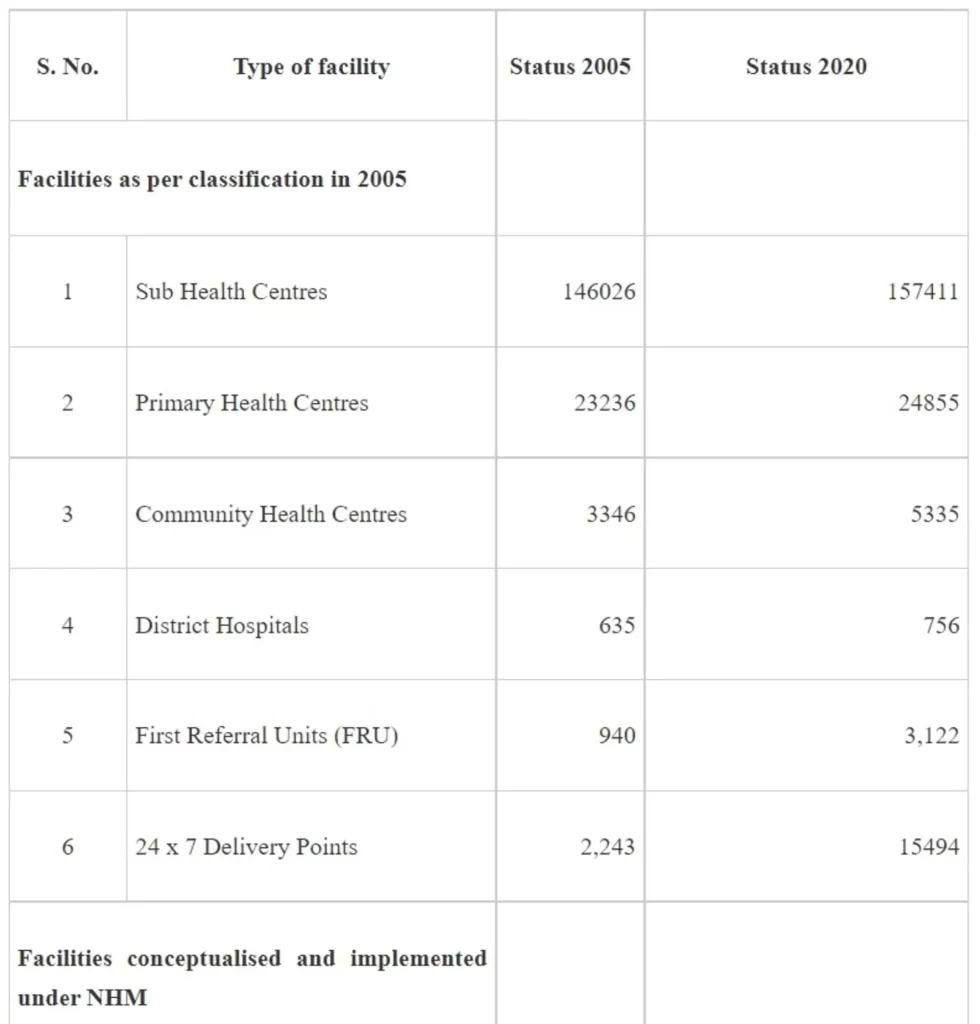
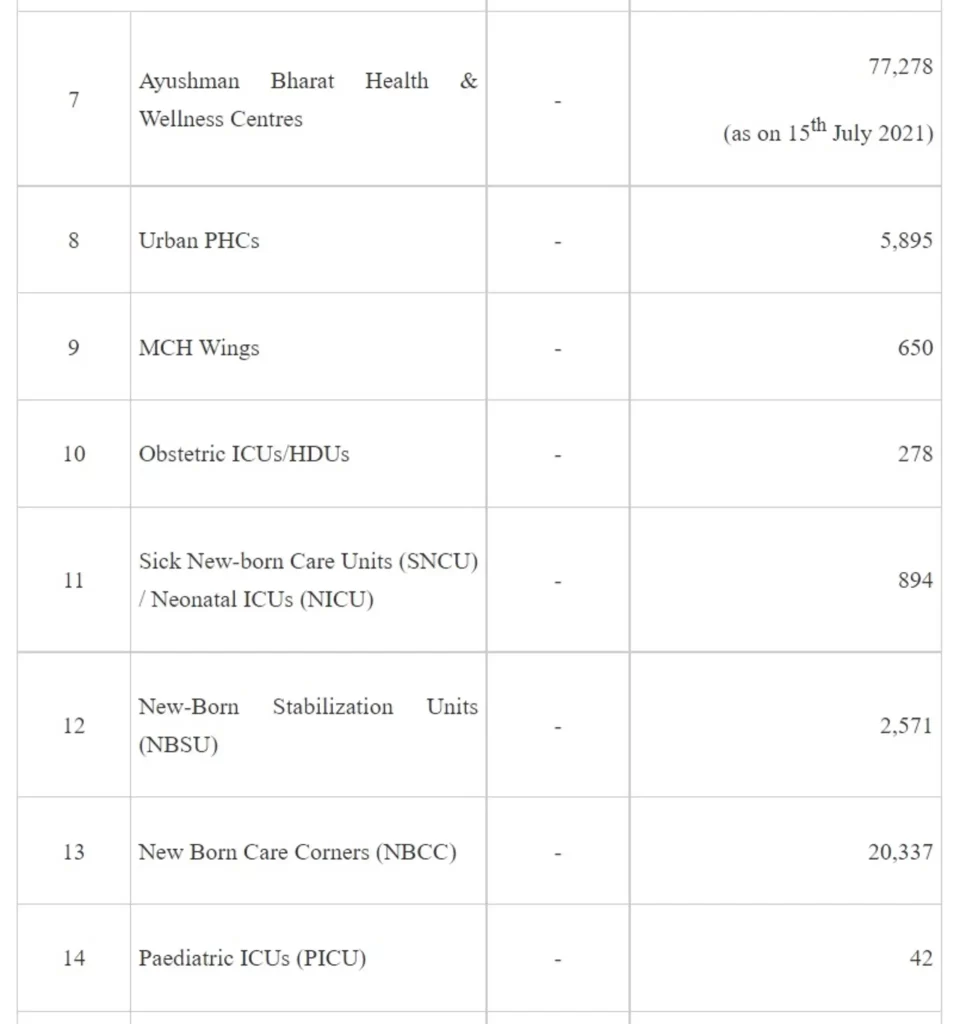
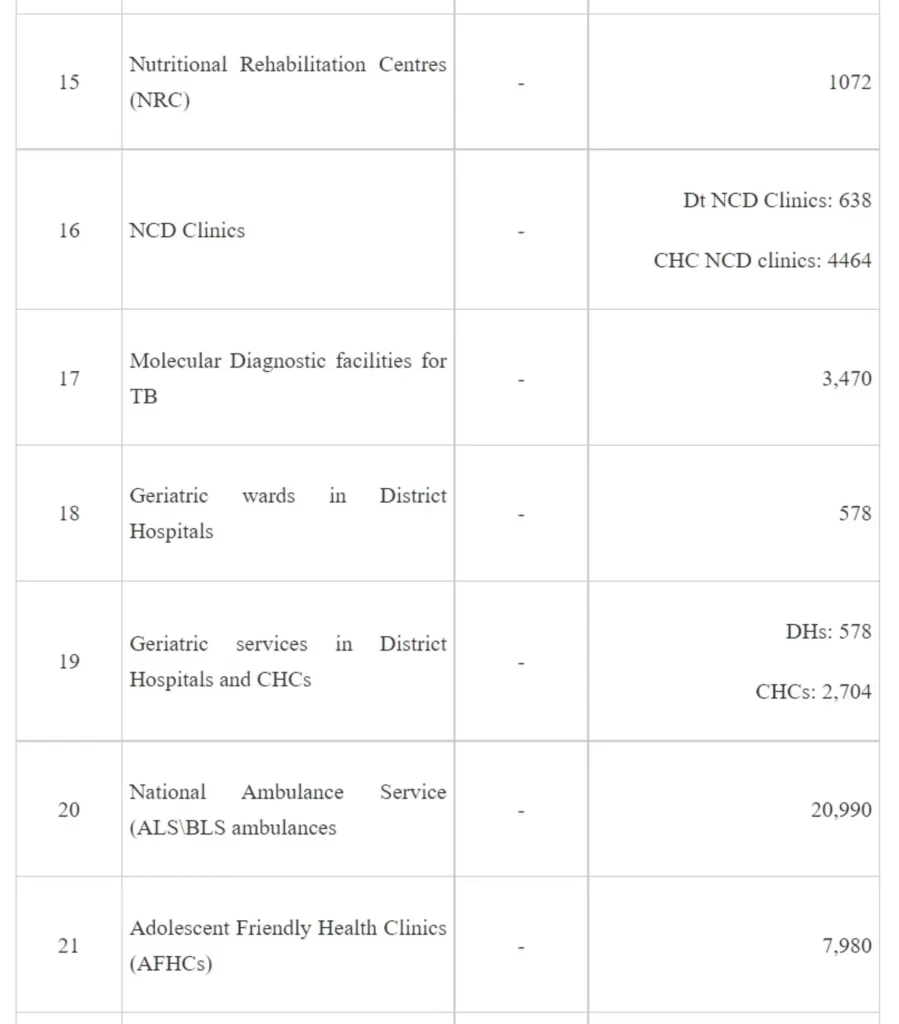
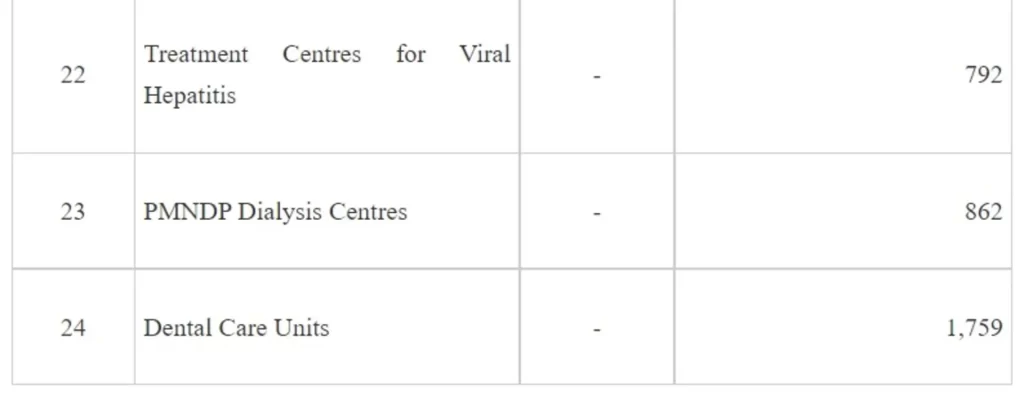
It may be seen from the above that implementation of NHM has not only contributed to increase in the institutional capacities for service delivery but also has led to development of capacities for targeted interventions of the various National Programmes under the NHM.
3. Equitable development: There was also a sustained focus on the health of tribal populations, those in Left Wing Extremism areas, and the urban poor. A more recent effort at ensuring equity in access and use, is the Aspirational district initiative, in which 115 districts across 28 states, with weak social and human development indicators have been identified for allocation of additional resources and capacity enhancement to catch up with more progressive districts.
4. National Ambulance Services: At the time of launch of NRHM (2005), ambulance networks were non-existent. So far, 20,990 Emergency Response Service Vehicles are operational under NRHM. Besides 5,499 patient transport vehicles are also deployed, particularly for providing “free pickup and drop back” facilities to pregnant women and sick infants.
5. Human Resource Augmentation: NHM supports states for engaging of service delivery HR such as doctors, nurses and health workers and also implements the world’s largest community health volunteer programme through the ASHAs. More than 10 lakhs ASHAs and ASHA facilitators are engaged under NHM. NHM has also supported states to acquire staff with skills in public health, finance, planning, management and M&E to plan and implement interventions, freeing up clinical staff to deliver health services. Differential remuneration and incentives linked to remote/underserved areas of posting, resulted in increased availability and retention of HR in difficult and remote areas. Human Resource Information System (HRIS) is a key administrative HR reform implemented under NHM for regular and contractual staff, providing comprehensive real time information on the HR posted in facilities and in managerial positions, facilitating salary payments and transfer postings. NHM invests in capacity building and mentoring for all cadres of personnel, with a focus on multi-skilling and task sharing to address HR shortages.
6. Health Sector Reforms: The National Health Mission (NHM) enabled the design and implementation of reforms specifically related to Governance (Decentralized Management Units, and creation of flexibility in administrative and fund management decisions, creation of Rogi Kalyan Samitis, establishment of community collectives for accountability, such as the Village Health, Sanitation and Nutrition Committees and Mahila Arogya Samitis), Procurement (creating the institutional mechanisms needed for effective procurement modalities such as the Medical services Corporations in several states, improve supply chain and logistics management, and expand procurement capacity), and Technology (to facilitate reporting, follow up of mothers and children in need of services, IEC and for tracking vaccine use).
7. Addressing high Out-of-Pocket Expenditure (OOPE): Recognising the need for reducing the current high levels OOPE, and that, almost 70% of the OOPE is on account of drugs and diagnostics, the Free Drugs and Free Diagnostics Services Initiatives have been implemented under the NHM. The Essential Drugs Lists (EDL) and the Essential Diagnostics Lists have been notified and are periodically updated to include more essential drugs based on new initiatives undertaken. These interventions coupled with strengthening of health facilities’ capacities to deliver an expanded range of services, closer to communities, teleconsultation services are leading to significant reduction in OOPE.
NHM supported health system reforms have resulted in development of resilient health systems, albeit at different stages of maturity that enable successful scaling up of existing interventions and the addition of newer reforms given the rapid transitions from rural to urban, gradual demographic transition to aging populations, increasing burden of chronic diseases, and the emergence of newer infectious diseases.
Of note is that the NHM reforms were oriented towards addressing inequity, across and within states. A study done in 2020 by the National Institute of Public Finance and Policy (NIPFP) on health spending in states under NHM, suggests that NHM contributed to reduction of inequality in health spending across states and added funds to the lower tiers of the health pyramid.
The Union Minister of State for Health and Family Welfare, Dr. Bharati Pravin Pawar stated this in a written reply in the Rajya Sabha here today.
Union Health Ministry reviewed preparations under the recently approved, Rs. 23,123 Cr. “India COVID-19 Emergency Response and Health Systems Preparedness Package: Phase II” with Health Secretaries and senior officials of all States/UTs, through a video conference (VC) today.
Union Cabinet chaired by Hon’ble Prime Minister approved the new scheme "India COVID-19 Emergency Response and Health Systems Preparedness Project: Phase-II" amounting to Rs. 23,123 Crore for FY 2021-22 on 8th July 2021. This will be implemented from 01 July 2021 to 31 March 2022.
Phase-II of the Emergency COVID19 Response Package (ECRP) has Central Sector (CS) and Centrally Sponsored Schemes (CSS) components.
This scheme aims to accelerate health system preparedness for immediate responsiveness for early prevention, detection and management with sharp focus on health infrastructure development including Paediatric care and with measurable outcomes. This will help in strengthening decentralized public health actions and public health facilities in rural, peri-urban and tribal areas.
During the review meeting, States/UTs were guided on policies and guidelines from Union Health Ministry on different aspects of COVID management which would help the states/UTs in strengthening their healthcare infrastructure to streamline COVID-19 response. States/UTs were requested to send their expenditure proposals at the earliest to enable expeditious approval and sanctioning from the Union Government.
Focus on the following was highlighted during the meeting:
Need for ramping up test, track, treat & isolate strategy
Requirement for scaling up testing capacity, additional beds including for Paediatric care and makeshift hospitals in sub-district levels
Ensuring availability of critical drugs, testing kits and PPEs
Enhancing Oxygen availability and strengthen home and village/ community isolation centres/ COVID care centres
Sustaining and enhancing skilled medical and para-medical HR in line with the new guidelines issued by the Union Health Ministry in consultation with National Medical Commission (NMC) and Indian Nursing Council (INC)
The following objectives of ECRP: Phase-II were reiterated at the review meeting:
Support the States to establish dedicated Paediatric Care Unit in all 736 districts of the country for responding to the needs of Paediatric COVID-19 management
Support the States to establish Paediatric Centre of Excellence in each State/UT (either in State Medical Colleges or State Hospitals or Central Hospitals such as AIIMS, Institute of National Importance, etc) for providing Tele-ICU services, mentoring and technical hand-holding to these District Paediatric units.
Increase the availability of ICU beds including 20% Paediatric ICU beds as per evolving needs
Support to States to have 1050 Liquid Medical Oxygen (LMO) Storage Tanks along with Medical Gas Pipeline System (MGPS) (at least one per District) to augment the availability of medical Oxygen in public healthcare system.
Strengthen Tele-consultation Platform to provide up to 5 lakhs Tele-consultation services per day, duly augmenting the availability of Spokes and Hubs
Support the States in implementation of Hospital Management Information System (HMIS) in all the District Hospitals, duly strengthening the national architecture.
Increase access to ambulance services to eliminate delays in transport and referral of COVID-19 patients Enhance testing capacity and corresponding supportive diagnostics at the public healthcare facilities, closer to the public
Support the States to utilize the UG and PG Interns, Final Year MBBS, BSc and GNM Nursing students for effective management
States/UTs were advised to conduct a quick gap analysis for various infrastructure components under ECRP-II including their IT infrastructure readiness. The hub and spoke model for Tele-consultation services may to be improved including for the services at the COVID Care Centres, through District level Hubs. The Final year MBBS students, UG Interns and PG residents may be utilized under the supervision of the faculty as per the National Medical Commission Guidelines for providing services of mild COVID management through Tele-consultation and similarly, final Nursing graduates (BSc and GNM) may be utilized for full-time COVID nursing duties at government facilities under the supervision of Senior Faculty as per the Indian Nursing Council Guidelines and support of remuneration / incentives to these Human Resources for Health under the ECRP-II may be leveraged by the States effectively in the times of need. All facilities in the District (COVID/Non-COVID), Health and Wellness Centers (HWCs), eSanjeevaniOPD, Common Service Centers (CSCs) and other healthcare facilities like Sub-Centres (SCs), Primary Health Centres (PHCs), Community Health Centres (CHCs) etc., are required to be connected with District hospitals as spoke.
It was also pointed out that procurement of drugs for effective COVID management is an essential component of ECRP-II; guidelines on procurement and buffer stocks have been already shared with the states which may be adopted for this purpose. Guidance can be tweaked to local needs and states must come up with their own assessments based on stocks and costs involved.
Ms. Vandana Gurnani, AS&MD (NHM) and Shri Vikas Sheel, Addl. Secy with senior officers from the Union Health Ministry were present during the meeting.
Union Health Ministry reviewed preparations under the recently approved, Rs. 23,123 Cr. “India COVID-19 Emergency Response and Health Systems Preparedness Package: Phase II” with Health Secretaries and senior officials of all States/UTs, through a video conference (VC) today.
Union Cabinet chaired by Hon’ble Prime Minister approved the new scheme "India COVID-19 Emergency Response and Health Systems Preparedness Project: Phase-II" amounting to Rs. 23,123 Crore for FY 2021-22 on 8th July 2021. This will be implemented from 01 July 2021 to 31 March 2022.
Phase-II of the Emergency COVID19 Response Package (ECRP) has Central Sector (CS) and Centrally Sponsored Schemes (CSS) components.
This scheme aims to accelerate health system preparedness for immediate responsiveness for early prevention, detection and management with sharp focus on health infrastructure development including Paediatric care and with measurable outcomes. This will help in strengthening decentralized public health actions and public health facilities in rural, peri-urban and tribal areas.
During the review meeting, States/UTs were guided on policies and guidelines from Union Health Ministry on different aspects of COVID management which would help the states/UTs in strengthening their healthcare infrastructure to streamline COVID-19 response. States/UTs were requested to send their expenditure proposals at the earliest to enable expeditious approval and sanctioning from the Union Government.
Focus on the following was highlighted during the meeting:
Need for ramping up test, track, treat & isolate strategy
Requirement for scaling up testing capacity, additional beds including for Paediatric care and makeshift hospitals in sub-district levels
Ensuring availability of critical drugs, testing kits and PPEs
Enhancing Oxygen availability and strengthen home and village/ community isolation centres/ COVID care centres
Sustaining and enhancing skilled medical and para-medical HR in line with the new guidelines issued by the Union Health Ministry in consultation with National Medical Commission (NMC) and Indian Nursing Council (INC)
The following objectives of ECRP: Phase-II were reiterated at the review meeting:
Support the States to establish dedicated Paediatric Care Unit in all 736 districts of the country for responding to the needs of Paediatric COVID-19 management
Support the States to establish Paediatric Centre of Excellence in each State/UT (either in State Medical Colleges or State Hospitals or Central Hospitals such as AIIMS, Institute of National Importance, etc) for providing Tele-ICU services, mentoring and technical hand-holding to these District Paediatric units.
Increase the availability of ICU beds including 20% Paediatric ICU beds as per evolving needs
Support to States to have 1050 Liquid Medical Oxygen (LMO) Storage Tanks along with Medical Gas Pipeline System (MGPS) (at least one per District) to augment the availability of medical Oxygen in public healthcare system.
Strengthen Tele-consultation Platform to provide up to 5 lakhs Tele-consultation services per day, duly augmenting the availability of Spokes and Hubs
Support the States in implementation of Hospital Management Information System (HMIS) in all the District Hospitals, duly strengthening the national architecture.
Increase access to ambulance services to eliminate delays in transport and referral of COVID-19 patients Enhance testing capacity and corresponding supportive diagnostics at the public healthcare facilities, closer to the public
Support the States to utilize the UG and PG Interns, Final Year MBBS, BSc and GNM Nursing students for effective management
States/UTs were advised to conduct a quick gap analysis for various infrastructure components under ECRP-II including their IT infrastructure readiness. The hub and spoke model for Tele-consultation services may to be improved including for the services at the COVID Care Centres, through District level Hubs. The Final year MBBS students, UG Interns and PG residents may be utilized under the supervision of the faculty as per the National Medical Commission Guidelines for providing services of mild COVID management through Tele-consultation and similarly, final Nursing graduates (BSc and GNM) may be utilized for full-time COVID nursing duties at government facilities under the supervision of Senior Faculty as per the Indian Nursing Council Guidelines and support of remuneration / incentives to these Human Resources for Health under the ECRP-II may be leveraged by the States effectively in the times of need. All facilities in the District (COVID/Non-COVID), Health and Wellness Centers (HWCs), eSanjeevaniOPD, Common Service Centers (CSCs) and other healthcare facilities like Sub-Centres (SCs), Primary Health Centres (PHCs), Community Health Centres (CHCs) etc., are required to be connected with District hospitals as spoke.
It was also pointed out that procurement of drugs for effective COVID management is an essential component of ECRP-II; guidelines on procurement and buffer stocks have been already shared with the states which may be adopted for this purpose. Guidance can be tweaked to local needs and states must come up with their own assessments based on stocks and costs involved.
Ms. Vandana Gurnani, AS&MD (NHM) and Shri Vikas Sheel, Addl. Secy with senior officers from the Union Health Ministry were present during the meeting.
The Union Government is committed to accelerating the pace and expanding the scope of COVID-19 vaccination throughout the country. The new phase of universalization of COVID-19 vaccination commenced from 21st June 2021. The vaccination drive has been ramped up through availability of more vaccines, advance visibility of vaccine availability to States and UTs for enabling better planning by them, and streamlining the vaccine supply chain.
As part of the nationwide vaccination drive, Government of India has been supporting the States and UTs by providing them COVID Vaccines free of cost. In the new phase of the universalization of the COVID19 vaccination drive, the Union Government will procure and supply (free of cost) 75% of the vaccines being produced by the vaccine manufacturers in the country to States and UTs.
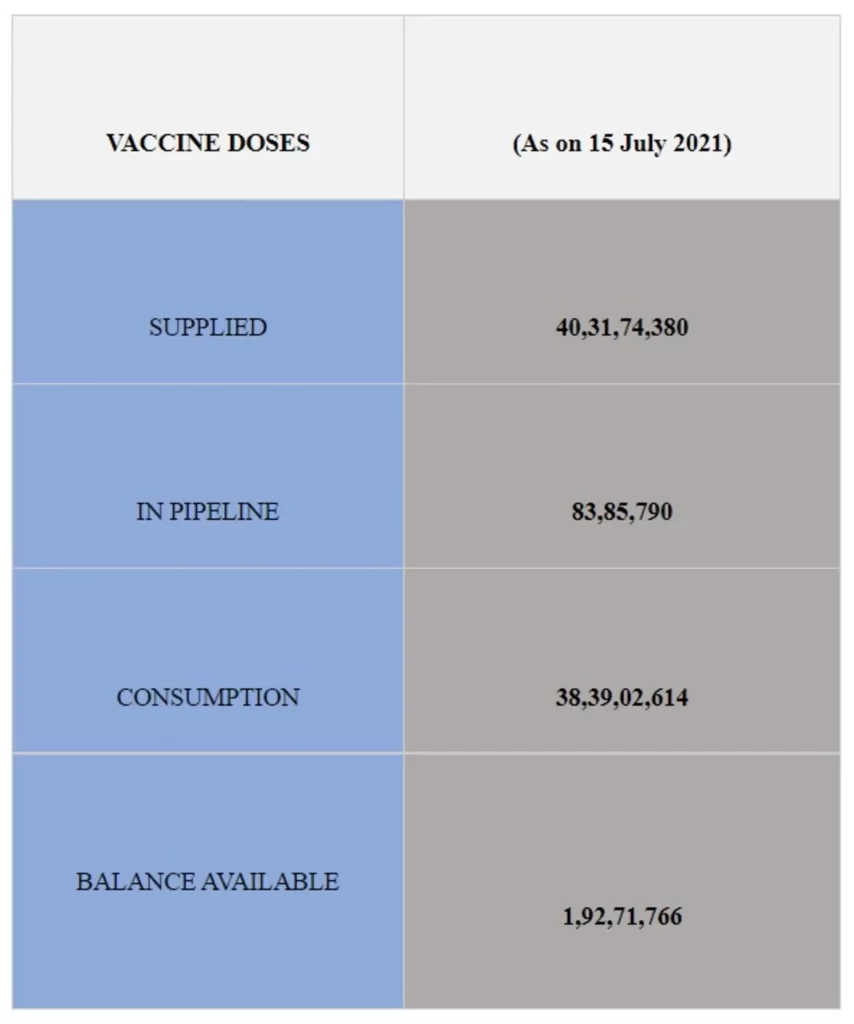
More than 40.31 crore (40,31,74,380) vaccine doses have been provided to States/UTs so far, through all sources and a further 83,85,790 doses are in the pipeline.
Of this, the total consumption including wastages is 38,39,02,614 doses (as per data available at 8 AM today).
More than 1.92 Cr (1,92,71,766) balance and unutilized COVID Vaccine doses are still available with the States/UTs and private hospitals to be administered.
Keeping in view the evolving COVID pandemic, and the continuing demand of the medical devices, the Government has decided to regulate their prices for affordable healthcare and COVID management.By invokingextraordinary powers under Para 19 of the DPCO, 2013 in larger public interest National Pharmaceutical Pricing Agency (NPPA) has vide its notification dated 13.07.2021, capped the Trade Margin up to 70% on Price to Distributor (PTD) level on (i) Pulse Oximeter, (ii) Blood Pressure Monitoring Machine, (iii) Nebulizer, (iv) Digital Thermometer, and (v) Glucometer. Earlier, in February 2019 NPPA hadpreviouslycapped the Trade Margin on Anti-Cancer Drugs and on 3rd June 2021 for Oxygen Concentrators. Based on the notified Trade Margin, NPPA has instructed the manufacturers / importers to report revised MRP within seven days. Revised MRPs will be informed in public domain thereafter by NPPA. The revised prices will come into effect from 20th July 2021.
Every retailer, dealer, hospital and institution shall display price lists of these medical devicesas furnished by the manufacturer, on a conspicuous part of the business premises in a manner so as to be easily accessible to any person wishing to consult the same. The manufacturers / importers not complying with the revised MRPafter Trade Margin capping, shall be liable to deposit the overcharged amount along with interest @15% andpenalty up to 100% under the provisions of the Drugs (Prices Control) Order, 2013 read with Essential Commodities Act, 1955. State Drug Controllers (SDCs) shall monitor the compliance of the order to ensure that no manufacturer, distributer, retailer shall sellthese medical devices to any consumer at a price exceeding the revised MRP, to prevent instances of black-marketing.
The Order shall be applicable up to 31st January 2022, subject to review.
The Union Cabinet chaired by the Prime Minister Shri Narendra Modi has approved the Memorandum of Understanding (MoU) between the Ministry of Health & Family Welfare of the Government of the Republic of India and the Ministry of Health of the Kingdom of Denmark on Cooperation in the field of Health and Medicine.
Benefits:
The bilateral Memorandum of Understanding will encourage cooperation between the Ministry of Health and Family Welfare of the Republic of India and the Ministry of Health of the Kingdom of Denmark through joint initiatives and technology development in the health sector. It will strengthen bilateral ties between India and Denmark.
The bilateral Memorandum of Understanding shall encourage cooperation between the Ministry of Health and Family Welfare of the Republic of India and the Ministry of Health of the Kingdom of Denmark through joint initiatives and development of research in the health sector. This will facilitate in improving the public health status of the people of both the countries.
