Next-generation sequencing (NGS) enables a better understanding of genetic mechanisms linked to specific diseases and is helping drive personalised healthcare
Preparing DNA for sequencing libraries is complex and prone to human error, which can interfere with accurate sequencing results
The new AVENIO Edge System is a fully-automated solution for the sample preparation workflow, from DNA sample input to the output of sequencing-ready libraries, and supports more comprehensive testing with consistent and high quality results for precision medicine for the future
Basel, 13 December 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the launch of the AVENIO Edge System, a core component of Roche’s strategy to advance sequencing technologies. Built on best-in-class foundational capabilities to deliver a fully-automated, integrated sequencing solution.
The AVENIO Edge System is a pre-analytical platform for sequencing library preparation, target enrichment and quantification steps that deliver integrated, end-to-end control with reliable, consistent high-quality results.
“Roche is committed to developing diagnostic solutions with the goal of providing the healthcare community with faster and more accurate medical information to predict risk and detect disease,” said Thomas Schinecker, CEO Roche Diagnostics. “We are pleased to offer next-generation sequencing laboratories and translational researchers the new automated AVENIO Edge System that aims to drastically reduce human error and help ensure fast, reliable and accurate results.”
Next-generation sequencing samples are precious. Every step of sample preparation has the potential to impact results. The AVENIO Edge System’s high reproducibility and low error rate can support the goal of reducing the number of rejected samples which otherwise might have to be re-collected.
The new AVENIO Edge System offers ready-to-use components in addition to customisable workflow parameters, making it a scalable, cost-efficient solution for sequencing laboratories seeking high performance and agility. With a setup to initiation of 20 minutes, this walkaway system demonstrated more than a 96 percent lower error opportunity and an 84 percent reduction in hands-on time.
The AVENIO Edge System will be available at select locations worldwide with timelines that vary country by country.
The AVENIO Edge System is Class I, In Vitro Diagnostics (IVD), 510(k) exempt, in the US. It is Class A in the EU in accordance with EU Regulation 2017/746 (IVDR) of the European Parliament and of the Council of 5 April 2017 on in vitro diagnostic medical devices. At launch, the AVENIO Edge Instrument can be used for Research Use Only (RUO) workflows. Consumables are General Laboratory Use (GLU). Compatible reagents and workflows are Research Use Only (RUO). Not for use in diagnostic procedures.
The AVENIO Edge System simplifies next-generation sequencing (NGS) and elevates automated sample preparation with integrated workflows, reagents, barcoded consumables and connectivity to enable reliable, high-quality results and the freedom to do more.
The AVENIO Edge System delivered high sequencing performance and demonstrated high uniformity, specificity and reproducibility in our in-house technical validation and early customer studies. At an early evaluation study site, the AVENIO Edge System replaced 384 manual steps while preparing 24 DNA libraries in one run versus manually preparing 24 DNA libraries.
The AVENIO Edge System is a fully automated liquid handling technology consisting of all-in-one hardware and traceable solutions that guides the operators through the process, provides real-time tracking of samples and delivery of the results to the laboratory information system (LIS). It is intended for routine laboratory tasks and designed to support multiple library prep, target enrichment and quantification workflow steps with customizable parameters. The AVENIO Edge System offers a wide set of modular, barcoded and ready-to-run reagents in addition to customizable workflow parameters, making it a scalable, cost-efficient solution for sequencing laboratories seeking high performance and agility.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
People with moderate or mild haemophilia A have significant unmet clinical needs, as this population may not use preventative treatments due to missed or delayed diagnoses of bleeding episodes and a lack of treatment guidelines1,
New data indicate that Hemlibra has a favourable safety profile in people with moderate or mild haemophilia A without factor VIII inhibitors, with no new safety signals identified
Hemlibra also achieved clinically meaningful bleed control, with 80.3% of participants experiencing no bleeding episodes that required treatment and 90.1% experiencing no joint bleeds that required treatment
A separate analysis of thrombosis-related events in people taking Hemlibra, including real-world data, further confirmed the safety profile of Hemlibra
Basel, 13 December 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced results from an interim analysis of the phase III HAVEN 6 study, which show Hemlibra® (emicizumab) demonstrated a favourable safety profile and effective bleed control in people with moderate or mild haemophilia A without factor VIII inhibitors.3 The data were presented at the 63rd American Society of Hematology (ASH) Annual Meeting and Exposition as an oral presentation on 12 December 2021.
While the treatment and management of severe haemophilia A are well-established, there is less information and treatment guidance on moderate and mild haemophilia A, which can lead to delayed or missed diagnosis of bleeding episodes.1 Considering this population may not use preventative treatments, they may experience worsened clinical burden, with less than 30% of people with moderate or mild haemophilia A living a bleed-free life.
“We are pleased to see that Hemlibra continues to show benefit in additional haemophilia A populations, regardless of severity,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “The clinical evidence for Hemlibra derives from one of the largest pivotal clinical trial programmes in haemophilia A, with and without factor VIII inhibitors. We remain committed to working together with the haemophilia community as we further explore the efficacy and safety of Hemlibra in broader populations.”
HAVEN 6 is a phase III study evaluating the safety, efficacy, pharmacokinetics and pharmacodynamics of Hemlibra in people with moderate or mild haemophilia A without factor VIII inhibitors. This interim analysis included data from 71 participants (69 men and two women); 20 of whom had mild haemophilia A without factor VIII inhibitors and 51 had moderate haemophilia A without inhibitors. Thirty-seven participants were on factor VIII prophylaxis at baseline.
This interim analysis was conducted after 50 participants with moderate haemophilia A completed at least 24 weeks in the study or withdrew. Data cut-off was on 16 April 2021. These data show Hemlibra demonstrated a favourable safety profile and effective bleed control in the HAVEN 6 study, with 80.3% of participants experiencing no bleeding episodes that required treatment and 90.1% experiencing no joint bleeds that required treatment.3 Annualised bleeding rates (ABR) remained low, consistent with previously reported observations from the HAVEN 1-4 studies.3,5,6,7,8 In addition, of the 50 participants aged 12 years or more who responded to the EmiPref questionnaire, 48 (96.0%) preferred Hemlibra to their previous treatment, one preferred their old treatment, and one expressed no preference.
The most common adverse events (AEs) occurring in 10% or more people in the HAVEN 6 study were headache (14.1%) and local injection site reactions (ISRs) (12.7%). Eleven people (15.5%) reported a Hemlibra-related AE, with ISRs being the most common (12.7%). There were no deaths, or cases of thrombotic microangiopathy (TMA) or serious thrombotic events (TEs) in the study as of the data cut-off, reinforcing Hemlibra’s favourable safety profile.
A separate analysis of TE and TMA events in people taking Hemlibra, including real-world data, will also be presented as a poster at ASH.4 These results showed that the evaluation of reported events without concomitant activated prothrombin complex concentrate (aPCC) remains similar to previous analyses as exposure increases, and the benefit/risk profile of Hemlibra remains unchanged. These data further confirm the favourable safety profile of Hemlibra, consistent with results from previous HAVEN and STASEY studies.
Hemlibra is approved to treat people with haemophilia A with factor VIII inhibitors in more than 100 countries worldwide and for people without factor VIII inhibitors in more than 90 countries worldwide, including the US, EU and Japan. Hemlibra has been studied in one of the largest clinical trial programmes in people with haemophilia A with and without factor VIII inhibitors, including eight phase III studies.
Hemlibra is a bispecific factor IXa- and factor X-directed antibody. It is designed to bring together factor IXa and factor X, proteins involved in the natural coagulation cascade, and restore the blood clotting process for people with haemophilia A. Hemlibra is a prophylactic (preventative) treatment that can be administered by an injection of a ready-to-use solution under the skin (subcutaneously) once-weekly, every two weeks, or every four weeks (after an initial once-weekly dose for the first four weeks). Hemlibra was created by Chugai Pharmaceutical Co., Ltd. and is being co-developed globally by Chugai, Roche and Genentech. It is marketed in the United States by Genentech as Hemlibra (emicizumab-kxwh), with kxwh as the suffix designated in accordance with Nonproprietary Naming of Biological Products Guidance for Industry issued by the U.S. Food and Drug Administration.
Haemophilia A is an inherited, serious disorder in which a person’s blood does not clot properly, leading to uncontrolled and often spontaneous bleeding. Haemophilia A affects around 900,000 people worldwide, approximately 35-39% of whom have a severe form of the disorder. People with haemophilia A either lack or do not have enough of a clotting protein called factor VIII. In a healthy person, when a bleed occurs, factor VIII brings together the clotting factors IXa and X, which is a critical step in the formation of a blood clot to help stop bleeding. Depending on the severity of their disorder, people with haemophilia A can bleed frequently, especially into their joints or muscles. These bleeds can present a significant health concern as they often cause pain and can lead to chronic swelling, deformity, reduced mobility and long-term joint damage. A serious complication of treatment is the development of inhibitors to factor VIII replacement therapies. Inhibitors are antibodies developed by the body’s immune system that bind to and block the efficacy of replacement factor VIII, making it difficult, if not impossible, to obtain a level of factor VIII sufficient to control bleeding.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for over 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematological diseases. Our approved medicines include MabThera®/Rituxan® (rituximab), Gazyva®/Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie, and Hemlibra® (emicizumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies, glofitamab and mosunetuzumab, targeting both CD20 and CD3, and cevostamab, targeting both FcRH5 and CD3; Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1; and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Data showcase the potential of glofitamab and Lunsumio to address diverse patient needs.
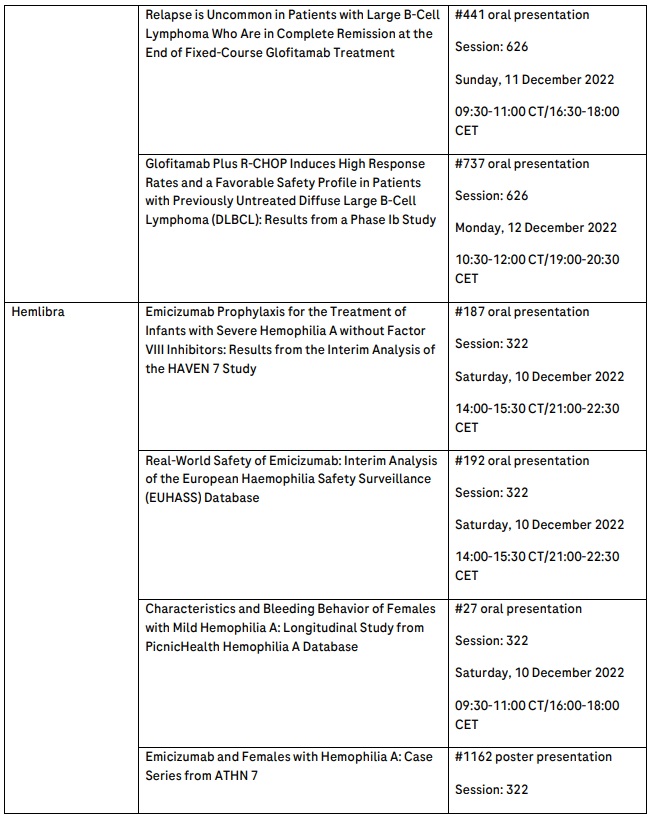
Data presented at ASH 2022 and simultaneously published in the New England Journal of Medicine showed that glofitamab, given as a fixed course, induced early and durable responses in people with heavily pre-treated large B-cell lymphoma.
27-month follow-up data showed Lunsumio continued to induce high and durable responses in people with relapsed or refractory follicular lymphoma, with 60% experiencing a complete response.
Basel, 12 December 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that updated clinical data for its CD20xCD3 T-cell engaging bispecific antibodies, including five oral presentations, were presented at the 64th American Society of Hematology (ASH) Annual Meeting & Exposition, 10-13 December 2022. Updated results for investigational bispecific glofitamab in people with relapsed or refractory (R/R) large B-cell lymphoma (LBCL) suggest glofitamab has the potential to be the first, off-the-shelf CD20xCD3 T-cell engaging bispecific antibody that can be given for a fixed period of time to people with heavily pre-treated aggressive lymphoma.1 These data will be presented at the meeting, and simultaneously published online in the New England Journal of Medicine (NEJM).2 Additionally, updated data for Lunsumio® (mosunetuzumab) continued to demonstrate clinically meaningful outcomes in people with heavily pre-treated follicular lymphoma (FL).3 Lunsumio is a fixed-duration treatment that can be administered in the outpatient setting, which could allow people the possibility of experiencing a lasting remission with a treatment-free period.
“We pioneered the development of T-cell engaging bispecific antibodies for lymphoma with the aim of expanding treatment options for people with difficult-to-treat blood cancers,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “New glofitamab and Lunsumio data continue to demonstrate durable and impressive patient responses, including complete remissions, when given for a fixed period of time. We believe these medicines could potentially transform treatment and offer new hope for people with lymphomas.”
Updated data from the pivotal phase II NP30179 study in people with R/R LBCL showed glofitamab given as a fixed course induced early and durable responses that were maintained beyond the end of treatment. Most patients who had achieved a complete response (CR; a disappearance of all signs of cancer) at the end of treatment experienced durable responses, with a median CR follow-up from end of treatment of 11.5 months (95% confidence interval [CI]: 10.5-16.4). Twelve months after the end of treatment with glofitamab, 61% of patients (n=37/61) maintained a CR, 92.6% remained progression-free and only one patient (n=1/44) experienced disease progression.
Simultaneously, an earlier data cut from the phase II NP30179 study in R/R diffuse large B-cell lymphoma (DLBCL) was published online in NEJM.2
Data from this pivotal phase II study have been submitted for review to the European Medicines Agency, and submissions to additional health authorities worldwide, including the U.S. Food and Drug Administration (FDA), are ongoing.
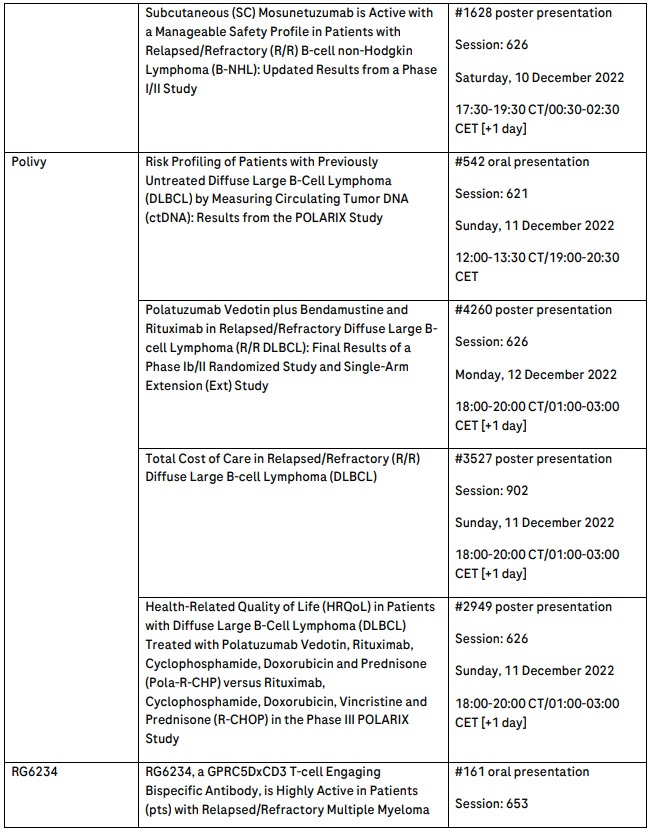
An updated analysis from the pivotal phase II GO29781 study of Lunsumio in people with R/R FL who had received two or more prior therapies showed 60.0% (n=54/90; 95% CI: 49.1–70.2) achieved a CR and 77.8% (95% CI: 67.8–85.9) achieved an objective response (a CR or a partial response, a decrease in the amount of cancer in their body) at a median follow-up of 28.3 months. After 24 months of achieving a CR, 62.7% of patients remained in remission (95% CI: 37.7–87.7). Overall, 48.3% of patients remained progression-free (95% CI: 36.2-60.3). The median duration of response, median duration of CR, and median progression-free survival were not reached. Safety was consistent with the previous analysis of study data, with no new cytokine release syndrome (CRS) events or Grade 3 or higher adverse events (AEs) reported. CRS events were experienced by 44% of patients, and were predominately low grade and during cycle one.3
The European Commission granted conditional marketing authorisation for Lunsumio for the treatment of people with R/R FL who have received at least two prior systemic therapies in June 2022, making it the first and only fixed-duration bispecific antibody to be approved in Europe for lymphoma. Lunsumio is under Priority Review with the FDA, with a decision expected by 29 December 2022.
Roche continues to evaluate Lunsumio and glofitamab as part of its commitment to providing off-the-shelf therapies for people with lymphomas that can meet their diverse needs, including fixed-duration treatment options. Additional data presented at ASH 2022 include the following:
A subcutaneous (SC) formulation of Lunsumio (administered as an injection given under the skin) demonstrated comparable efficacy with the intravenous formulation and a manageable safety profile in people with R/R non-Hodgkin lymphoma (NHL). The most common AEs were injection site reactions (60.9%; n=53/87) and CRS events (27.6%; n=24/87), which were all Grade 1 or 2. These findings suggest that a SC formulation of Lunsumio may offer patients a treatment option that could reduce their time spent in treatment centres.
Updated results from the phase I/II G050554 study of Lunsumio monotherapy in elderly/unfit patients with previously untreated DLBCL and additional analyses from the phase I/II G040516 study of Lunsumio in combination with Polivy® (polatuzumab vedotin) in heavily pre-treated people with DLBCL continued to show promising efficacy and manageable safety, highlighting the potential of Lunsumio in these patient populations.
Results from the phase I/II NP30179 study evaluating glofitamab as a monotherapy following pre-treatment with Gazyva®/Gazyvaro® (obinutuzumab) in patients with heavily pre-treated R/R mantle cell lymphoma continued to show early, high and durable response rates in this difficult-to-treat disease. After a median follow-up of eight months, the overall response rate (ORR) was 83.8%, with the majority of patients showing durable complete responses at the data cut off (74.1%; n=20/27). The most common AE was CRS (75.5%; n=28/37), with the majority low grade.
Data from the safety and expansion cohorts of the phase Ib NP40126 study evaluating glofitamab in combination with MabThera®/Rituxan® (rituximab) plus cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP) in patients with previously untreated DLBCL showed, after a median follow-up of 8.5 months, a best ORR of 92.7% (n=51/55) and a complete metabolic response rate of 72.7% (n=40/55). In the safety cohort, CRS events were all low grade (Grade 1 or 2 [10.7%; n=6/56]), and serious AEs were reported in 18 patients (32.1%).
Both Lunsumio and glofitamab are being investigated as SC formulations and in phase III studies that will expand the understanding of their impact in earlier lines of treatment, with the aim of continuing to address the diverse needs and preferences of people with blood cancers. This includes the confirmatory phase III CELESTIMO study investigating Lunsumio plus lenalidomide as a chemotherapy-free option for patients with R/R FL; the phase III SUNMO study investigating Lunsumio plus Polivy versus MabThera/Rituxan in combination with gemcitabine plus oxaliplatin (R-GemOx) in patients with R/R aggressive B-cell NHL who are ineligible for autologous stem cell transplant (ASCT); and the phase III STARGLO study evaluating glofitamab in combination with gemcitabine and oxaliplatin (GemOx) versus MabThera/Rituxan in combination with GemOx in patients with R/R DLBCL who are ineligible for ASCT.
Glofitamab is an investigational CD20xCD3 T-cell-engaging bispecific antibody designed to target CD3 on the surface of T-cells and CD20 on the surface of B-cells. Glofitamab was designed with a novel 2:1 structural format. This T-cell-engaging bispecific antibody is engineered to have one region that binds to CD3, a protein on T-cells, a type of immune cell, and two regions that bind to CD20, a protein on B-cells, which can be healthy or malignant. This dual-targeting brings the T-cell in close proximity to the B-cell, activating the release of cancer cell-killing proteins from the T-cell. A robust clinical development program for glofitamab is ongoing, investigating the molecule as a monotherapy and in combination with other medicines for the treatment of people with B-cell non-Hodgkin’s lymphomas, including diffuse large B-cell lymphoma and other blood cancers.
Lunsumio is a CD20xCD3 T-cell engaging bispecific antibody designed to target CD20 on the surface of B-cells and CD3 on the surface of T-cells. This dual-targeting activates and redirects a patient’s existing T-cells to engage and eliminate target B-cells by releasing cytotoxic proteins into the B-cells. A robust clinical development programme for Lunsumio is ongoing, investigating the molecule as a monotherapy and in combination with other medicines, for the treatment of people with B-cell non-Hodgkin lymphomas, including follicular lymphoma and diffuse large B-cell lymphoma, and other blood cancers.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera®/Rituxan® (rituximab), Gazyva®/Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie, Hemlibra® (emicizumab) and Lunsumio® (mosunetuzumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies glofitamab, targeting both CD20 and CD3, cevostamab, targeting both FcRH5 and CD3, Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1, and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The phase III COMMODORE 3 study of crovalimab met primary endpoints of transfusion avoidance and haemolysis control in people with paroxysmal nocturnal hemoglobinuria (PNH).
COMMODORE 3 is the first China-specific study in PNH. Current treatment options are extremely limited in China, resulting in significant levels of disease-related morbidity and mortality for people living with PNH.
Based on these data crovalimab has received Breakthrough Therapy Designation and is under Priority Review for approval in China.
Basel, 11 December 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced positive new data from the phase III COMMODORE 3 study in China, demonstrating that crovalimab, a novel anti-C5 recycling monoclonal antibody, is efficacious and well-tolerated in people with paroxysmal nocturnal haemoglobinuria (PNH). The study met its co-primary efficacy endpoints of transfusion avoidance (TA) and haemolysis control, demonstrating that participants with PNH, who have not been treated previously with complement inhibitors and who received subcutaneous crovalimab injections every four weeks, achieved disease control.1 The data were presented at the American Society of Hematology (ASH) congress, taking place from 10-13 December 2022.
PNH is an ultra-rare and life-threatening blood condition, where red blood cells are targeted and destroyed by the complement system — part of the innate immune system — causing symptoms such as anaemia, fatigue, blood clots and kidney disease. 3 C5 inhibitors — treatments that block part of the complement system — have been shown to be effective in treating the condition. Crovalimab has been engineered to be recycled into circulation, enabling sustained complement inhibition, and potentially reducing the treatment burden associated with currently available treatments. 4, 5, 6, 7 It is being investigated in a comprehensive clinical development programme, including five ongoing global phase III studies in PNH and other complement mediated diseases. 8, 9, 10, 11, 12
“We are pleased with the strength of these first phase III data for crovalimab, which we hope will address the urgent need for efficacious and well-tolerated treatment options for this life-threatening condition in China.” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “As crovalimab has been developed to be taken subcutaneously and infrequently, with the option to self-administer, it has the potential to become an important treatment option for people everywhere living with paroxysmal nocturnal haemoglobinuria.”
The COMMODORE 3 study included data from 51 participants with PNH, who received crovalimab subcutaneously every four weeks during the primary study period. Results showed that the co-primary efficacy endpoints of haemolysis control and TA, indicators of disease control, were met. The mean proportion of participants with haemolysis control from week five through to week 25 was 78.7% (95% CI: 67.8%, 86.6%).1 The difference between the proportion of participants with TA within 24 weeks prior to screening (0.0%) and the proportion of participants with TA from baseline through to week 25 (51.0%) was statistically significant (p<0.0001).1 TA is defined as people who become transfusion-free and do not require transfusion per protocol-specified guidelines. Blood transfusion requirements are important clinical measures of haemolysis caused by complement dysregulation in PNH.
In addition, the proportion of participants with breakthrough haemolysis (used to measure a loss of disease control) from baseline through week 25 was 3.9% (95% CI: 0.7%, 14.6%), and the proportion of participants who achieved haemoglobin stabilisation was 51% (95% CI: 36.8%, 65.1%).1 A rapid and clinically meaningful improvement in fatigue status within two weeks after treatment with crovalimab was also reported and sustained over time, as measured by the FACIT-Fatigue scale.1 The overall safety data were consistent with the known safety profile of C5 inhibitors and underlying disease, showing that crovalimab was well-tolerated with no new safety signals identified.
Data from the COMMODORE 3 study have been submitted via China’s Centre for Drug Evaluation Breakthrough Therapy Designation pathway. This submission has been accepted under Priority Review for approval consideration in PNH by China’s National Medical Products Administration. As the availability of C5 inhibitors is extremely limited in China, there remains a high clinical need for people living with PNH there.
Data from the global crovalimab COMMODORE 1 and COMMODORE 2 PNH studies are expected.
The COMMODORE 3 study is a phase III, single-arm study in China evaluating the efficacy, safety, pharmacokinetics and pharmacodynamics of crovalimab in people who have not previously been treated with complement inhibitors.8 The study included 51 participants with paroxysmal nocturnal hemoglobinuria (PNH), who received crovalimab subcutaneously every four weeks for 24 weeks. Participants enrolled in the study received a loading series of crovalimab including an intravenous dose on day one, followed by weekly crovalimab subcutaneous (SC) doses for four weeks on week one day two, then on weeks two, three, and four. Maintenance SC dosing began at week five and continued every four weeks thereafter for a total of 24 weeks of study treatment. After 24 weeks of crovalimab treatment, participants who derived benefit from the drug can continue to receive crovalimab.
Crovalimab is an investigational, novel anti-C5 recycling monoclonal antibody designed to block the complement system – a vital part of the innate immune system that acts as the body’s first line of defence against infections. Crovalimab has been engineered to address the medical needs of people living with complement-mediated diseases and overcome some of the challenges of currently available treatment options.
Similar to currently approved C5 inhibitors, crovalimab binds to C5, blocking the last step of the complement cascade. 5 However, crovalimab is also recycled into circulation, enabling rapid and sustained complement inhibition, which may potentially overcome the problem of incomplete C5 inhibition with currently available treatments.4,5,6,7 Crovalimab’s recycling action also enables low dose subcutaneous administration every four weeks, potentially removing the need for regular, time-consuming intravenous infusions. In addition, crovalimab binds to a different C5 binding site from current treatments, which has the potential to provide an effective treatment option for people with specific C5 gene mutations, who do not respond to current therapies. 5 Crovalimab is being investigated in a comprehensive clinical development programme, including five ongoing global phase III studies.8 9, 10, 11,12 Crovalimab is being evaluated in paroxysmal nocturnal haemoglobinuria, atypical haemolytic uraemic syndrome, sickle cell disease, and other complement mediated diseases.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
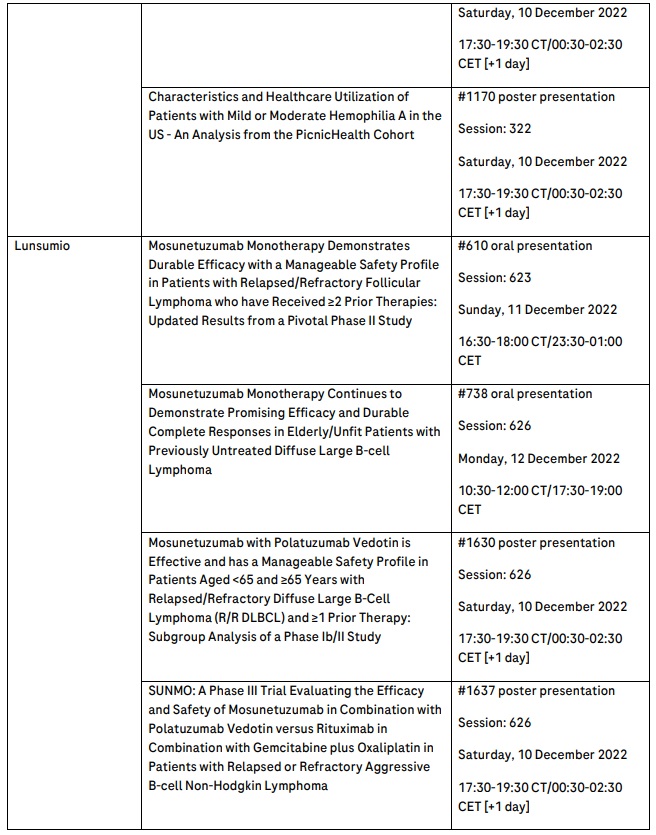
The HAVEN 7 study was designed to further confirm the benefit of preventative treatment (prophylaxis) with Hemlibra from birth in previously untreated or minimally treated infants with severe haemophilia A without inhibitors.
In the study, 77.8% of participants had no bleeding episodes that required treatment.
In addition, real-world efficacy and safety data from the EUHASS database and ATHN 7 study were also presented.
Basel, 11 December 2022 – Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced interim results from the phase III HAVEN 7 study. The study shows Hemlibra® (emicizumab) achieved meaningful bleed control with a favourable safety profile in infants (up to 12 months) with severe haemophilia A, without factor VIII inhibitors: 77.8% of participants did not have any bleeds that required treatment and 42.6% did not have any treated or untreated bleeds at all.1 These results help support the use of Hemlibra in this population, in which it is already approved in many countries around the world. The new data were presented at the 64th American Society of Hematology (ASH) Annual Meeting and Exposition taking place in New Orleans from 10-13 December 2022.
The burden of severe haemophilia A in infants and on their parents and caregivers is significant. The World Federation of Haemophilia treatment guidelines consider the standard of care in haemophilia to be regular prophylaxis initiated at a young age, as studies have shown that early prophylaxis improves long-term outcomes, while reducing the risk of intracranial haemorrhage. 4-6 However, for many infants with haemophilia A, prophylaxis is not started until after the first year of life because of the high treatment burden.7-11 Hemlibra provides a flexible treatment option that can be administered subcutaneously from birth at different dosing frequencies.
“These initial results support the benefit of starting Hemlibra from birth given that early preventative treatment is essential in infants,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Haemophilia can substantially reduce quality of life for those affected, starting at infancy, which is especially distressing for parents and caregivers. We continue to explore Hemlibra's potential benefits to a broad range of people with haemophilia A.”
HAVEN 7 is a phase III, multi-centre, open-label study evaluating the efficacy, safety, pharmacokinetics and pharmacodynamics of Hemlibra in infants with severe haemophilia A without factor VIII inhibitors. The results of this interim analysis, which included data from 54 participants, showed that 77.8% of participants (n=42) did not have any bleeds which required treatment, while 42.6% (n=23) did not have any treated or untreated bleeds at all. There were no treated spontaneous bleeds in any participants, and all treated bleeds were traumatic. A total of 77 bleeds occurred in 31 participants (57.4%); 88.3% were traumatic. Mean model-based annualised bleeding rate (ABR) (95% CI) at the time of interim analysis was 0.4 (0.23–0.65) for treated bleeds. 1
Hemlibra’s safety profile was consistent with previous studies, with no new safety signals observed. Nine people (16.7%) reported a Hemlibra-related adverse event (AE), all of which were local injection site reactions. Eight participants (14.8%) reported 12 serious AEs, unrelated to Hemlibra. There were no deaths, thromboembolic events or cases of thrombotic microangiopathy, reinforcing Hemlibra’s favourable safety profile. No intracranial haemorrhages occurred.1
Primary analysis will be conducted at 52 weeks. The study also has an additional seven-year follow-up period to collect long-term data such as safety and joint health outcomes, further building upon our understanding of the benefit of Hemlibra in this population.
Roche also presented data from the European Haemophilia Safety Surveillance (EUHASS) database and the prospective observational ATHN 7 study at ASH 2022. Data from EUHASS, which collects real-world safety data on treatments for inherited disorders, showed the safety profile of Hemlibra in people with haemophilia A was favourable and consistent with clinical trial data. 2 Data from ATHN 7, exploring the efficacy of Hemlibra in women with haemophilia A, showed two of the three female participants had no bleeds; the third had one treated bleed associated with a dental procedure and one untreated bleed associated with menses. Ongoing evaluation is vital to further understand the safety and efficacy profile of Hemlibra in this rare and under-represented population.3
Hemlibra is approved as a treatment for people with haemophilia A with factor VIII inhibitors in more than 110 countries worldwide, and for people without factor VIII inhibitors in more than 100 countries worldwide. It has been studied in one of the largest clinical trial programmes in people with haemophilia A with and without factor VIII inhibitors, including eight phase III studies.
Hemlibra is a bispecific factor IXa- and factor X-directed antibody. It is designed to bring together factor IXa- and factor X, proteins involved in the natural coagulation cascade, and restore the blood clotting process for people with haemophilia A. Hemlibra is a prophylactic (preventative) treatment that can be administered by an injection of a ready-to-use solution under the skin (subcutaneously) once-weekly, every two weeks, or every four weeks (after an initial once-weekly dose for the first four weeks). Hemlibra was created by Chugai Pharmaceutical Co., Ltd. and is being co-developed globally by Chugai, Roche and Genentech. It is marketed in the United States by Genentech as Hemlibra (emicizumab-kxwh), with kxwh as the suffix designated in accordance with Nonproprietary Naming of Biological Products Guidance for Industry issued by the U.S. Food and Drug Administration.
Haemophilia A is an inherited, serious disorder in which a person’s blood does not clot properly, leading to uncontrolled and often spontaneous bleeding. Haemophilia A affects around 900,000 people worldwide, 4,12 approximately 14% and 48% of whom have a moderate or mild form of the disorder,13 respectively. However, the severity of haemophilia A is not always reflective of bleeding behaviour, as some people with non-severe haemophilia may experience symptoms similar to those with severe haemophilia and warrant prophylaxis.14 All severities of haemophilia A can significantly reduce the quality of life for people affected, as well as their family and caregivers. People with haemophilia A either lack or do not have enough of a clotting protein called factor VIII. In a healthy person, when a bleed occurs, factor VIII brings together the clotting factors IXa- and X, which is a critical step in the formation of a blood clot to help stop bleeding. Depending on the severity of their symptoms, people with haemophilia A can bleed frequently, especially into their joints or muscles. 13 These bleeds can present a significant health concern as they often cause pain and can lead to chronic swelling, deformity, reduced mobility and long-term joint damage.15 A serious complication of treatment is the development of inhibitors to factor VIII replacement therapies. Inhibitors are antibodies developed by the body’s immune system that bind to and block the efficacy of replacement factor VIII, making it difficult, if not impossible, to obtain a level of factor VIII sufficient to control bleeding.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera®/Rituxan® (rituximab), Gazyva®/Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie, Hemlibra® (emicizumab) and Lunsumio® (mosunetuzumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies glofitamab, targeting both CD20 and CD3 and cevostamab, targeting both FcRH5 and CD3, Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1, and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The Elecsys® Alzheimer’s disease (AD) CSF assays will be available on the cobas fully automated immunoassay analyzers, enabling patients to get broad access to high quality testing in a timely manner.
Greater accessibility and lower cost means these assays could accelerate the path to diagnosis for people with Alzheimer’s disease.
The assays are easily scalable to meet the imminent demand for Alzheimer’s disease modifying therapies as they become available.
Basel, 8 December 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that its Elecsys® beta-Amyloid (1-42) CSF II (Abeta42) and Elecsys® Phospho-Tau (181P) CSF (pTau181) assays have received U.S. Food and Drug Administration (FDA) 510(k) clearance. The Elecsys AD CSF Abeta42 and pTau181 assays (used as a pTau181/Abeta42 ratio) measure two biomarkers that are hallmarks of Alzheimer’s pathology, beta-amyloid and tau proteins, in adults ages 55 and older being evaluated for the disease.
Currently, the diagnosis of Alzheimer’s is a diagnosis of exclusion, largely based on various cognitive tests, routine laboratory tests and structural imaging (MRI or CT scan). Clinical criteria are currently limited and lead to an accurate diagnosis in only 70-80% of cases.1 To increase diagnosis accuracy, an amyloid positron emission tomography (PET) scan, that measures the build-up of abnormal beta-amyloid protein in the brain, can be used as an adjunct to these evaluations.2. However, the high cost, limited availability, and exposure to radioactivity limit its use. The Elecsys AD CSF assays achieve 90% concordance with the Amyloid PET scan imaging3 and have the potential to provide a more affordable and accessible routine option to confirm the presence of amyloid in the brain. They also offer detection of multiple biomarkers from one draw, with no radiation and potential to detect Alzheimer’s pathology in early stages of disease.4
“Globally, up to 75% of people living with Alzheimer’s disease have not been diagnosed, and those who have, often report a long and complicated process5,” said Thomas Schinecker, CEO of Roche Diagnostics. “The Elecsys AD CSF assays have the potential to guide more people with suspected Alzheimer’s disease towards a diagnosis than ever before. As we are starting to see exciting results for new potential Alzheimer’s treatments, reliable tests that have been clinically validated will be critical in ensuring the right patients are identified and able to benefit from them.”
An early and accurate diagnosis allows those facing Alzheimer's, under the guidance of their physician, an opportunity to take steps to preserve cognitive function, make better care plans, and get involved in clinical research that may lead to new treatments. Research suggests that disease modifying therapies are likely to be most beneficial when used in the earliest stages of the disease. Therefore, early and accurate diagnosis will be even more essential to ensure that people with Alzheimer’s receive the right treatment, at the right time. Due to the large installed base of instruments, the Elecsys AD CSF assays are easily scalable to meet the imminent demand that these therapies will generate as they become available.
Roche’s Elecsys AD CSF assays are already registered in 45 countries worldwide, including those accepting the CE Mark. In July, Roche also announced that the FDA granted Breakthrough Device Designation to its Elecsys® Amyloid Plasma Panel, an innovative, minimally invasive and easily accessible solution which enables the measurement of Alzheimer’s biomarkers from a blood sample. These assays are still in development but once they are available, they could be used to streamline patients toward confirmation of amyloid pathology using the Elecsys AD CSF assays.
Alzheimer's disease is characterised by the abnormal accumulation in the brain of amyloid beta and tau proteins, which are biomarkers of Alzheimer’s hallmark pathology. The Elecsys beta-Amyloid (1-42) CSF II and Elecsys Phospho‑Tau (181P) CSF assays accurately and reliably measure the beta‑Amyloid (1‑42) and Phospho‑Tau (181P) concentrations in cerebrospinal fluid (CSF) in adult subjects, ages 55 and older, being evaluated for Alzheimer's disease and other causes of cognitive impairment to generate a pTau181/Abeta42 ratio value. As changes in these biomarkers levels occur at early stages of the disease, the assay can detect Alzheimer’s pathology in earlier stages of disease (e.g. mild cognitive impairment). Elecsys pTau181 and Elecsys Abeta42 assays are both traceable to reference materials ensuring accuracy of the pTau181/Abeta42 ratio results.
The ratio of these biomarkers (pTau181/Abeta42) is consistent with a negative beta‑Amyloid PET scan if the result is less than or equal to the cutoff (negative), and with a positive beta‑Amyloid PET scan if the result is above the ratio cutoff (positive).
Abeta42 and pTau181 assays are intended to be used in addition to other clinical diagnostic evaluations to determine whether a person has Alzheimer’s. A positive pTau181/Abeta42 ratio result in CSF does not establish a diagnosis of Alzheimer's disease.
With more than two decades of scientific research in Alzheimer’s, Roche is working towards a day when we can detect the disease early and stop its progression to preserve what makes people who they are. Today, the company’s Alzheimer’s portfolio spans investigational medicines for different targets, types and stages of the disease. It also includes approved and investigational tools, including digital and blood-based tests and cerebrospinal fluid (CSF) assays, aiming to more effectively detect, diagnose, and monitor the disease. Yet the global challenges of Alzheimer’s go well beyond the capabilities of science, and making a meaningful impact requires collaboration both within the Alzheimer’s community and outside of healthcare. We will continue to work together with numerous partners with the hope we can transform millions of lives.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Phase III GRADUATE studies did not meet their primary endpoints of slowing clinical decline in people with early Alzheimer’s
The level of beta-amyloid removal by gantenerumab was lower than expected
Topline data will be presented at the Clinical Trials on Alzheimer’s Disease (CTAD) Conference
Roche is committed to the Alzheimer’s community and will continue to develop novel diagnostics and potential treatments for Alzheimer’s
Basel, 14 November 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced results from the GRADUATE I and II studies evaluating gantenerumab in people with mild cognitive impairment (MCI) due to Alzheimer’s and mild Alzheimer’s dementia, collectively called early Alzheimer’s disease. The studies did not meet their primary endpoint of slowing clinical decline. Gantenerumab was well tolerated, including the subcutaneous administration.
“So many of our families have been directly affected by Alzheimer’s, so this news is very disappointing to deliver,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We are profoundly grateful to the study participants, their care partners and study sites for their contributions to this research. While the GRADUATE results are not what we hoped, we are proud to have delivered a high quality, clear and comprehensive Alzheimer’s dataset to the field, and we look forward to sharing our learnings with the community as we continue to search for new treatments for this complex disease.”
Study participants treated with gantenerumab showed a slowing of clinical decline in GRADUATE I and GRADUATE II of -0.31 (p=0.0954), and -0.19 (p=0.2998) respectively from baseline score on the Clinical Dementia Rating-Sum of Boxes (CDR-SB), however, neither was statistically significant. This represents a relative reduction in clinical decline of 8% in GRADUATE I and 6% in GRADUATE II compared with placebo. The CDR-SB measures cognitive and functional change across six areas including memory, orientation, judgement and problem solving, community affairs, home and hobbies, and personal care.
The level of beta-amyloid removal, the protein that builds up to make plaques in the brains of people with Alzheimer's disease, was lower than expected. Roche will present topline findings of the GRADUATE I and II studies at the upcoming Clinical Trials on Alzheimer’s Disease (CTAD) Conference on Wednesday, 30 November, 2022 at 16:15 PT.
Amyloid related imaging abnormalities (ARIA) are a common radiological finding associated with amyloid-targeting therapies. The incidence of ARIA-E (oedema or effusion) in the pooled gantenerumab arms was 25%, with the vast majority being asymptomatic and very few leading to treatment discontinuation. The incidence of isolated ARIA-H (haemosiderin) was balanced across the gantenerumab and placebo groups.
Roche remains committed to Alzheimer’s disease, one of the most complex neurological disorders and a major public health challenge. The company is continuing to develop and deliver tests to enable early and accurate Alzheimer’s diagnosis and has a pipeline of investigational medicines for different targets, types and stages of the disease.
The Phase III GRADUATE I and II studies were two global, double-blind, randomised, placebo-controlled clinical trials evaluating the safety and efficacy of the investigational anti-amyloid monoclonal antibody gantenerumab in people with mild cognitive impairment (MCI) due to Alzheimer’s and mild Alzheimer’s dementia over 27 months. 1,965 study participants across 30 countries were randomised 1:1 to receive gantenerumab or placebo by subcutaneous injection titrated to reach a target dose of 510 mg administered every two weeks. The primary endpoint was the change from baseline on the Clinical Dementia Rating-Sum of Boxes (CDR-SB) at 116 weeks. The CDR-SB measures cognitive and functional change across six areas including memory, orientation, judgement and problem solving, community affairs, home and hobbies, and personal care. There were 17 secondary endpoints including change in disease severity assessed using various neuropsychological and functional assessment tools (e.g., MMSE, ADAS-Cog, etc.), assessment of therapeutic levels of gantenerumab, incidence of adverse events, disease biomarkers and scans. A full list is available at clinicaltrials.gov.
Gantenerumab is a fully-human monoclonal IgG1 antibody, an investigational medicine that is subcutaneously administered and designed to target and bind to aggregated forms of beta-amyloid, including oligomers, fibrils and plaques, and activate immune cells in the brain (microglia) to clear amyloid plaques and prevent further accumulation. Gantenerumab was discovered in collaboration with MorphoSys.
With more than two decades of scientific research in Alzheimer’s, Roche is working towards a day when we can detect the disease early and stop its progression to preserve what makes people who they are. Today, the company’s Alzheimer’s portfolio spans investigational medicines for different targets, types and stages of the disease. It also includes approved and investigational tools, including digital and blood-based tests and cerebrospinal fluid (CSF) assays, aiming to more effectively detect, diagnose, and monitor the disease. Yet the global challenges of Alzheimer’s go well beyond the capabilities of science, and making a meaningful impact requires collaboration both within the Alzheimer’s community and outside of healthcare. We will continue to work together with numerous partners with the hope we can transform millions of lives.
Neuroscience is a major focus of research and development at Roche. Our goal is to pursue groundbreaking science to develop new treatments that help improve the lives of people with chronic and potentially devastating diseases.
Roche has both approved and investigational medicines across multiple sclerosis, spinal muscular atrophy, neuromyelitis optica spectrum disorder, myasthenia gravis, Alzheimer’s disease, Huntington’s disease, Parkinson’s disease and Duchenne muscular dystrophy. Together with our partners, we are committed to pushing the boundaries of scientific understanding to solve some of the most difficult challenges in neuroscience.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Interim data from phase III HAVEN 7 study reinforce Hemlibra’s efficacy and safety in infants with severe haemophilia A without factor VIII inhibitors1
New and updated data support use of Polivy in diffuse large B-cell lymphoma, including its potential as a treatment option for previously untreated patients
New and updated data for innovative CD20xCD3 T-cell engaging bispecific antibodies Lunsumio and glofitamab further enhance their potential as effective, off-the-shelf, fixed-duration treatment options for people with lymphoma
First phase III data for crovalimab show the co-primary efficacy endpoints were met, with subcutaneous injections achieving disease control in people with paroxysmal nocturnal haemoglobinuria as shown in COMMODORE 3 study in China
Basel, 3 November 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that it will present new data from its industry-leading haematology portfolio at the 64th American Society of Hematology (ASH) Annual Meeting from 10-13 December 2022. The data to be presented span numerous blood diseases, including haemophilia A, paroxysmal nocturnal haemoglobinuria (PNH), and various types of blood cancers, including non-Hodgkin lymphoma (NHL) and multiple myeloma. Roche’s approved and investigational medicines will be featured in more than 50 abstracts, including more than 15 oral presentations.
“We continually strive to improve patient outcomes by exploring new treatment options across blood disorders, such as lymphomas and rare blood diseases, where unmet needs remain high,” said Levi Garraway, M.D., Ph.D., Chief Medical Officer and Head of Global Product Development. “The data we are presenting reinforce our ongoing commitment to redefining treatment paradigms, improving on existing standards of care and addressing a diversity of patient and healthcare system needs.”
With 25 years of expertise in blood diseases, Roche has developed new medicines that changed the standard of care in several blood disorders with high unmet need. The data at this year’s meeting exemplify Roche’s commitment to investing in its current portfolio to further improve patient outcomes.
Interim data from the phase III HAVEN 7 study reinforce the efficacy and safety of Hemlibra® (emicizumab) in infants with severe haemophilia A without factor VIII inhibitors.1 For this population, early prophylaxis may prevent long-term damage to joints and muscles and potentially reduce the risk of intracranial haemorrhage, which can be life-threatening.
New data evaluating Polivy® (polatuzumab vedotin) that underscore the potential impact of this treatment for the diffuse large B-cell lymphoma (DLBCL) patient community will be shared at the meeting. Health-related quality of life (HRQoL) data from the phase III POLARIX study will be presented, highlighting the potential impact of Polivy in combination with MabThera®/Rituxan® (rituximab), cyclophosphamide, doxorubicin and prednisone (R-CHP) on reducing the need for subsequent treatments in people with previously untreated DLBCL, a population where multiple subsequent treatments can be a significant treatment burden.2 Based on data from the POLARIX study, this Polivy combination has been approved in the EU and recently, Japan, for the treatment of adult patients with previously untreated DLBCL.
Roche is presenting updated data from the broadest and most comprehensive CD20xCD3 bispecific antibody development programme in the industry. This aims to provide off-the-shelf, fixed-duration treatment options, which address the unique and diverse needs of people with blood cancers. Data include updated analyses for Lunsumio® (mosunetuzumab), the first CD20xCD3 T-cell engaging bispecific antibody approved by the European Commission to treat follicular lymphoma (FL), and glofitamab, for which data have been submitted for approval to the European Medicines Agency, and submissions to additional health authorities worldwide, including the U.S. Food and Drug Administration (FDA), are ongoing.
An updated analysis from the pivotal phase II GO29781 study of Lunsumio in people with relapsed or refractory (R/R) FL after two or more prior therapies will show continued durable responses across multiple key efficacy endpoints in addition to offering the potential to be administered in an outpatient setting.3 In addition, studies evaluating Lunsumio as a monotherapy and in novel combinations for the treatment of DLBCL in earlier lines of treatment will be presented, highlighting the potential of Lunsumio in other settings.
Updated results from the phase II NP30179 study will show a fixed course of glofitamab monotherapy can deliver durable complete responses in people with heavily pre-treated aggressive lymphomas.6,7 Results from the pivotal R/R DLBCL cohort indicate patients can maintain durable responses following fixed-duration treatment with glofitamab, potentially allowing them to benefit from a treatment-free period
Roche is applying its scientific expertise to expand its haematology clinical development programme by exploring additional blood diseases and bringing innovations that address the various needs of patients in areas of high unmet need.
The first phase III clinical data for crovalimab from the COMMODORE 3 study in China, will be presented at ASH. These data demonstrate that crovalimab met the co-primary efficacy endpoints, suggesting that crovalimab is efficacious and well-tolerated in people with PNH, a rare and life-threatening blood condition, where healthy red blood cells are targeted and destroyed by the body’s complement system.8 There are currently no effective treatment options for PNH broadly available in China.
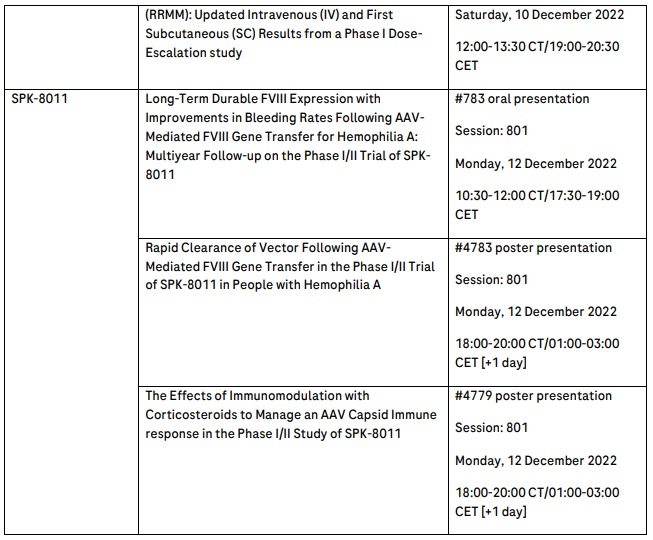
Spark Therapeutics, a member of the Roche Group, will share updated long-term follow-up data from the ongoing phase I/II clinical trial of SPK-8011, an investigational AAV-based gene therapy being developed for the treatment of haemophilia A.10 The acquisition of Spark Therapeutics brought new capabilities in haemophilia A to address the high unmet medical need for people living with this disease and endeavour to create additional benefit beyond current treatment options.
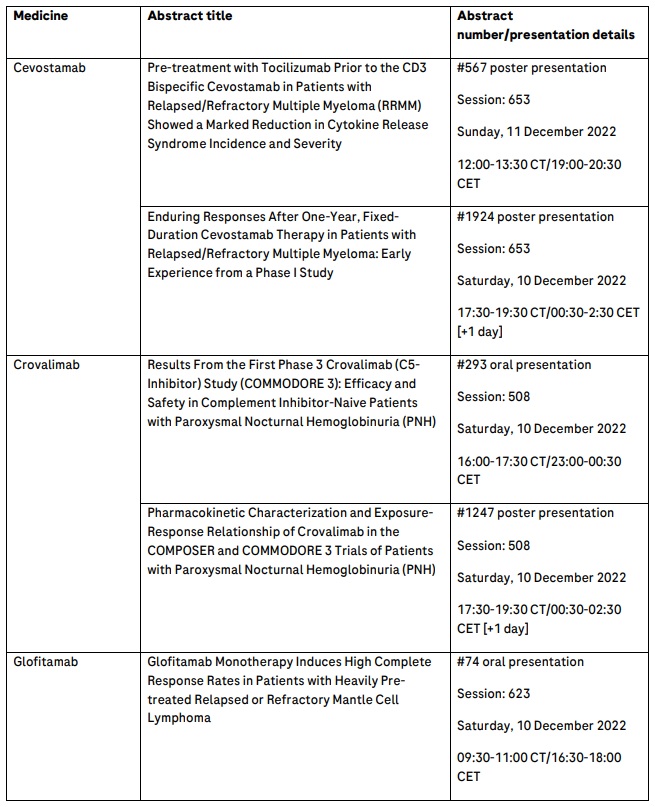
Positive data on cevostamab will be presented at ASH, including data from the phase I GO39775 study, which suggest that patients with heavily pre-treated multiple myeloma can maintain durable responses with fixed-duration cevostamab.11 Additionally, phase I data from Roche’s GPRC5DxCD3 T-cell engaging bispecific antibody, RG6234, showing encouraging preliminary activity in people with R/R multiple myeloma, will be presented.12 With this pipeline, Roche is committed to advancing treatments for multiple myeloma, which remains an incurable disease characterised by multiple relapses.
Further information on the key abstracts featuring Roche medicines that will be presented at ASH can be found in the table below.
Follow Roche on Twitter via @Roche and on LinkedIn and keep up to date with ASH Annual Meeting news and updates by using the hashtag #ASH22.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera®/Rituxan® (rituximab), Gazyva®/Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie, Hemlibra® (emicizumab) and Lunsumio® (mosunetuzumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies glofitamab, targeting both CD20 and CD3 and cevostamab, targeting both FcRH5 and CD3, Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1, and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
We believe gene therapy has the potential to revolutionise medicine and improve the lives of patients with genetic and other serious diseases. Pairing Roche’s long-standing commitment to developing medicines in haemophilia with Spark Therapeutics’ proven gene therapy expertise brings together the best team of collaborators researching gene therapies in haemophilia A.
It is our aligned objective to develop gene therapies for haemophilia A that, with the lowest effective dose and the optimal immunomodulatory regimen, demonstrate safety, predictability, efficacy, and durability for patients.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
OCREVUS data will show significant benefit on slowing disease activity and progression in patients with treatment-naive early-stage relapsing-remitting multiple sclerosis (RRMS)
Largest pregnancy safety data across anti-CD20 medicines for OCREVUS in multiple sclerosis (MS)
Nine-year safety data for OCREVUS reinforces its favourable benefit-risk profile
New research demonstrates impact of misdiagnosis and delay of starting treatment in NMOSD
Basel, 19 October 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that new OCREVUS® (ocrelizumab) data and continued research into neuromyelitis optica spectrum disorder (NMOSD) will be presented at the 38th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) from 26-28 October 2022. These data include 35 abstracts, highlighting disease activity and progression results in early-stage RRMS, pregnancy outcomes from more than 2,000 women with MS and long-term safety data for OCREVUS, as well as global NMOSD data exploring impact of delayed treatment, clinical characterization of disease severity and stability, and accurate identification of people living with NMOSD through healthcare claims-based algorithms. Finally, the design of a Phase III study evaluating the efficacy and safety of satralizumab in Myelin Oligodendrocyte Glycoprotein Antibody-associated Disease (MOGAD), a rare, chronic and debilitating autoimmune disease primarily affecting the optic nerve, brain and spinal cord, will be presented.
“Our aim is to enable people living with MS and NMOSD to maintain life to the fullest. With over 250,000 people treated with OCREVUS, we continue to see significant reductions in MS disease progression balanced with favourable safety,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We are also focused on remaining unmet needs – such as earlier diagnosis and treatment – which is critical to ensure patients are receiving the most appropriate treatment.”
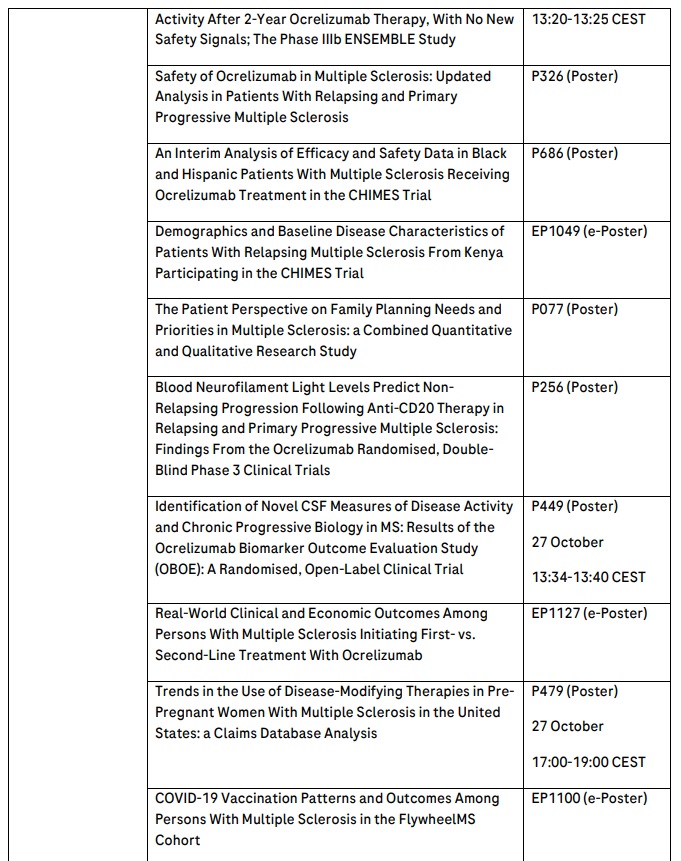
Roche will present 29 MS abstracts, including data from a two-year interim analysis of treatment-naive, early-stage patients with RRMS from the open label Phase IIIb ENSEMBLE study that will show the positive impact on disease activity and progression when newly diagnosed patients are treated with OCREVUS, and outcomes from the largest cumulative pregnancy dataset for an anti-CD20 MS medicine in more than 2,000 women treated with OCREVUS. Long-term data from all OCREVUS clinical trials in relapsing MS (RMS) and primary progressive MS (PPMS) over nine years will reinforce the consistently favourable benefit-risk profile of OCREVUS.
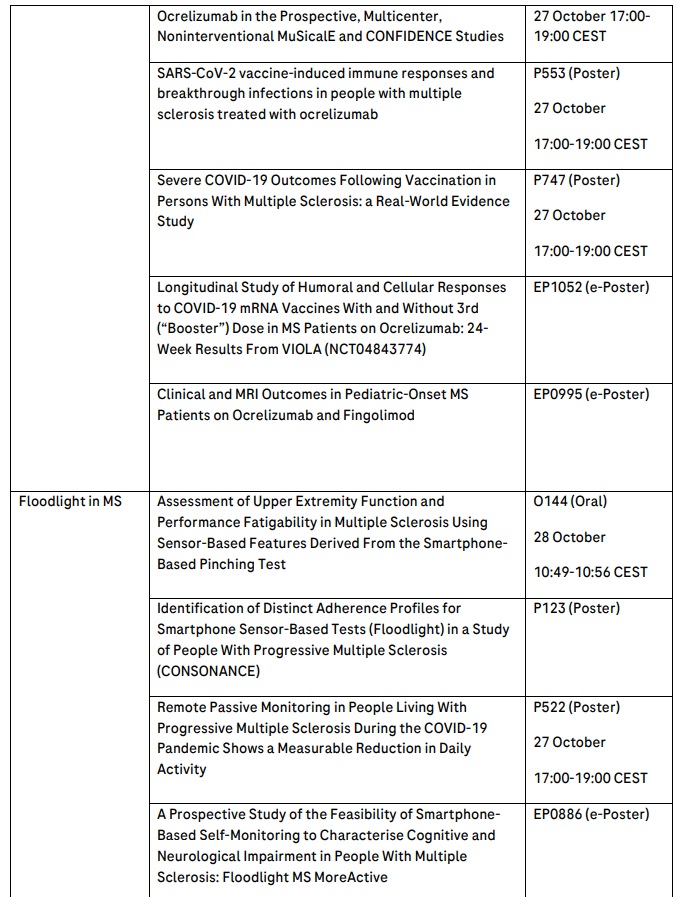
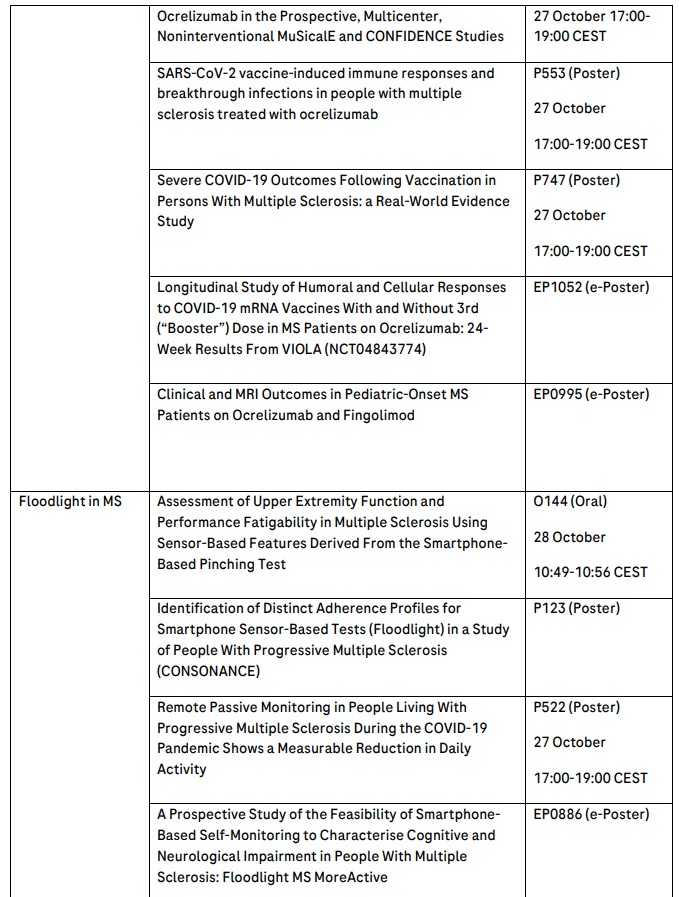
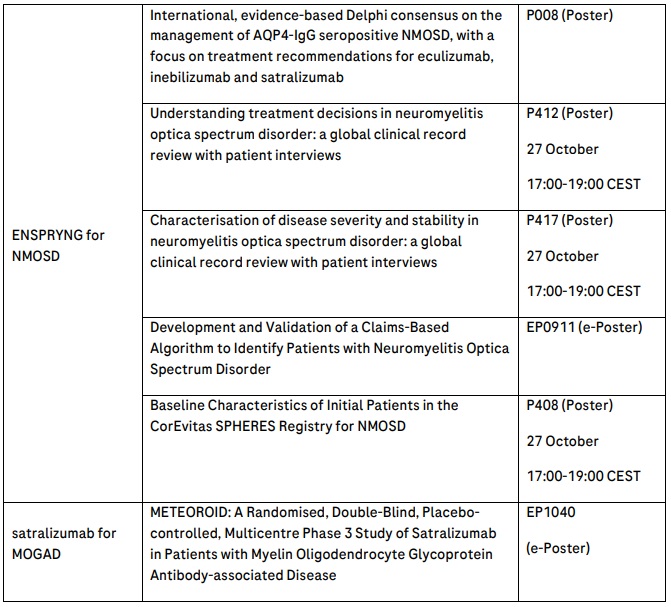
Roche will present five NMOSD abstracts, including the development and testing of a healthcare claims-based algorithm to identify people living with NMOSD. Misdiagnosis of NMOSD is common and associated with a delay in initiating maintenance therapy. This was highlighted in a study looking to develop a clearer understanding of patient characteristics, relapse severity and other drivers of treatment choice.
The development of validated consensus statements on AQP4-IgG seropositive NMOSD management will also be presented with a focus on treatment recommendations including satralizumab; these statements aim to optimise patient outcomes through informed treatment decision making. The characterisation of disease severity and stability in NMOSD will also be presented, with the aim of integrating these in worldwide NMOSD clinical practice.
Roche will also present the study design from a Phase III study that will evaluate the efficacy and safety of satralizumab in MOGAD.
Follow Roche on Twitter via @Roche and keep up to date with ECTRIMS 2022 news and updates by using the hashtag #ECTRIMS2022.


OCREVUS is the first and only therapy approved for both RMS (including RRMS and active, or relapsing, secondary progressive MS [SPMS], in addition to clinically isolated syndrome [CIS] in the U.S.) and PPMS. OCREVUS is a humanised monoclonal antibody designed to target CD20-positive B cells, a specific type of immune cell thought to be a key contributor to myelin (nerve cell insulation and support) and axonal (nerve cell) damage. This nerve cell damage can lead to disability in people with MS. Based on preclinical studies, OCREVUS binds to CD20 cell surface proteins expressed on certain B cells, but not on stem cells or plasma cells, suggesting that important functions of the immune system may be preserved. OCREVUS is administered by intravenous infusion every six months. The initial dose is given as two 300 mg infusions given two weeks apart. Subsequent doses are given as single 600 mg infusions.
ENSPRYNG, which was designed by Chugai, a member of the Roche Group, is a humanised monoclonal antibody that targets interleukin-6 (IL-6) receptor activity. ENSPRYNG was designed using novel recycling antibody technology which, compared to conventional technology, allows for longer duration of the antibody and subcutaneous dosing every four weeks.
Positive Phase III results for ENSPRYNG, as both monotherapy and in combination with baseline immunosuppressive therapy, demonstrate that IL-6 inhibition is an effective therapeutic approach for neuromyelitis optica spectrum disorder (NMOSD). ENSPRYNG is currently approved for NMOSD in 72 countries with further applications under review with numerous regulators. Roche continues to investigate ENSPRYNG in further indications including generalised myasthenia gravis (gMG), Myelin Oligodendrocyte Glycoprotein Antibody-associated Disease (MOGAD) and Autoimmune Encephalitis (AIE).
ENSPRYNG was granted Breakthrough Therapy Designation for the treatment of NMOSD by the FDA in December 2018 and designated as an orphan drug for NMOSD in the United States, Europe, Russia and Japan.
In addition, it has been designated as an orphan drug for gMG, MOGAD and AIE (NMDAR).
Neuroscience is a major focus of research and development at Roche. Our goal is to pursue ground-breaking science to develop new treatments that help improve the lives of people with chronic and potentially devastating diseases.
Roche has both approved and investigational medicines across multiple sclerosis, spinal muscular atrophy, neuromyelitis optica spectrum disorder, myasthenia gravis, Alzheimer’s disease, Huntington’s disease, Parkinson’s disease and Duchenne muscular dystrophy. Together with our partners, we are committed to pushing the boundaries of scientific understanding to solve some of the most difficult challenges in neuroscience today.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Group sales up 2%1 at constant exchange rates (CER) and 1% in Swiss francs; as expected, significantly lower COVID-19-related sales in both divisions in the third quarter
Sales in the Pharmaceuticals Division at the previous year’s level with significantly lower sales of COVID-19-related products (Ronapreve and Actemra/RoActemra) and losses to biosimilars, offset by strong growth of newer medicines
Sales in the Diagnostics Division rise 6%; base business remains strong; as expected, demand for COVID-19 tests sharply down in third quarter
Highlights in the third quarter:
EU approval for Vabysmo (severe eye diseases)
US approvals for Xofluza (influenza in children)
Launch of a digital PCR diagnostic platform and of COVID-19, skin and breast cancer tests
“Breakthrough Device Designation” granted for Alzheimer’s blood tests
Outlook for 2022 confirmed
Severin Schwan, CEO of Roche: “Group sales grew 2% despite the expected sharp decline in COVID-19-related products in both divisions in the third quarter. The demand for our newer medicines for multiple sclerosis, haemophilia, spinal muscular atrophy and cancer remains high. I am particularly pleased that so many patients with severe eye disease have already accessed our new medicine Vabysmo and started treatment. Likewise, our Diagnostics Division base business continued to grow strongly. Based on our current assessment, we confirm the outlook for the full year.”
![[Ad hoc announcement pursuant to Art. 53 LR] Roche records solid results for the first nine months of 2022](http://lswlifescienceworld.com/wp-content/uploads/2025/06/lsw-bulletin-oct18-2022-1.jpg)
Roche expects stable sales or sales growth in the low-single-digit range (at constant exchange rates). Core earnings per share are targeted to grow in the low- to mid-single-digit range (at constant exchange rates). Roche expects to increase its dividend in Swiss francs further.
In the first nine months, the Roche Group generated sales growth of 2% (1% in CHF) to CHF 47 billion.
As expected, the third quarter of 2022 was particularly challenging due to base effects, as the demand for COVID-19 medicines and tests was exceptionally high in the same quarter of 2021.
Sales of the Pharmaceuticals Division were at the same level as in the previous year, at CHF 33.2 billion.
The newer medicines Ocrevus (multiple sclerosis), Hemlibra (haemophilia), Evrysdi (spinal muscular atrophy) and Phesgo (breast cancer) continued their strong growth. The eye medicine Vabysmo, which was only launched at the beginning of the year, is now also one of the strongest growth drivers. These top-five medicines alone generated additional sales totalling CHF 2.2 billion.
As expected, sales growth was offset by the biosimilars-related decline in sales, especially for the established cancer medicines Avastin, MabThera/Rituxan and Herceptin. Sales of Actemra/RoActemra and Ronapreve (COVID-19) were also significantly lower than in the same period of 2021, with a decline of roughly CHF 1 billion, as the pandemic continued to weaken in many countries in 2022.
Sales in the United States decreased slightly with 1%. Growth of Ocrevus, Hemlibra, Vabysmo, Xolair and Tecentriq partially offset lower sales of Actemra/RoActemra, Avastin, Herceptin, MabThera/Rituxan, Lucentis and Esbriet.
In Europe, sales were also slightly down by 1%. The main reason for this was the decline in sales of the COVID-19 medicine Ronapreve compared to the previous year. Excluding this base effect, sales in Europe grew by 6%.
Sales were up in Japan (+7%), mainly due to supply of Ronapreve to the government, followed by sales growth of Evrysdi, Hemlibra, Polivy and Enspryng. This more than offset the impact of government price cuts and biosimilars.
Sales in the International region were stable. The impact of biosimilars and decline in sales of Actemra/RoActemra was compensated by Perjeta, Kadcyla, Hemlibra and Ocrevus sales.
The Diagnostics Division generated a 6% increase in sales to CHF 13.8 billion. The division’s base business continued its strong sales growth across all regions (+6%), with the largest contributions coming from the Europe, Middle East and Africa (EMEA) and Asia-Pacific regions. Immunodiagnostic products were the main growth drivers.
As expected, the demand for COVID-19 tests declined sharply in the third quarter of 2022 (CHF 0.6 billion versus CHF 1.0 billion in the same period last year).
Sales in the Asia-Pacific and North America regions increased strongly, by 28% and 20%, respectively. The 13% decline in sales in the EMEA region is primarily due to the lower demand for COVID-19 tests.
Ophthalmology
In September, the European Commission (EC) approved Vabysmo, the first bispecific antibody for the eye. Vabysmo simultaneously targets and inhibits two disease pathways that drive neovascular or ‘wet’ age-related macular degeneration (nAMD) and diabetic macular oedema (DME) – two of the leading causes of vision loss. With the potential to require fewer eye injections over time, while also improving and maintaining vision, Vabysmo could offer a less burdensome treatment schedule for individuals, their caregivers and healthcare systems.
Oncology In August, the FDA accepted the supplemental Biologics License Application (sBLA) for the Polivy combination therapy for previously untreated diffuse large B-cell lymphoma (DLBCL), an aggressive form of blood cancer. In four out of ten people with DLBCL, the cancer returns after initial treatment, at which point treatment options are limited and survival is short. Later in August, the Japanese health authorities granted approval for this Polivy combination in previously untreated DLBCL.
Also in August, Roche announced that the IMscin001 study met its co-primary endpoints. The study evaluated a subcutaneous formulation of cancer immunotherapy Tecentriq in people with advanced non-small cell lung cancer. Administered under the skin, this new formulation reduces the time spent receiving treatment to a matter of minutes, compared with up to an hour for an infusion.
Neuroscience
In October, Roche presented new data from its expanding neuromuscular disease portfolio. New two-year Evrysdi data show improvement or maintenance of motor function in people with spinal muscular atrophy (SMA). SMA is a severe, progressive neuromuscular disease that can be fatal. It is the leading genetic cause of infant mortality.
In addition, data from the gene therapy EMBARK study for Duchenne muscular dystrophy (DMD), the most advanced phase III study currently underway, reinforces our confidence in the programme. DMD is a rare, progressive neuromuscular disease, leading to a loss of muscle function and premature death.
Virology
In August, the FDA approved Xofluza for the treatment of acute uncomplicated influenza in otherwise healthy children aged five years and older. Historically, school-aged children have played a significant role in the community transmission of influenza. This approval marks the first single-dose oral influenza medicine approved in the US for children. Additionally, the FDA approved Xofluza for the prevention of influenza in children following contact with someone with influenza.
Pharmaceuticals: key development milestones in the third quarter of 2022
![[Ad hoc announcement pursuant to Art. 53 LR] Roche records solid results for the first nine months of 2022 -1](http://lswlifescienceworld.com/wp-content/uploads/2025/06/lsw-bulletin-oct18-2022-2.jpg)
In July, the FDA granted Breakthrough Device Designation to the Elecsys Amyloid Plasma Panel, an innovative new solution that enables Alzheimer’s disease to be detected earlier. This panel measures two blood-based biomarkers, called pTau 181 and ApoE4. It has the potential to streamline a patient’s journey and increase the speed of diagnosis, giving people living with Alzheimer’s disease and their caregivers more time to plan and prepare for the future.
In August, Roche launched the Digital LightCycler System. This next-generation digital PCR system helps clinical researchers better understand the nature of a patient’s cancer, genetic disease or infection. It has the potential to find and quantify ultra-rare, hard-to-detect disease mutations, leading to early diagnosis and therapy decisions.
Every four minutes, one person dies from skin cancer. However, when detected early, localised melanoma is curable with a simple surgical excision. The newly launched PRAME (EPR20330) Antibody test evaluates PRAME protein expression from patients with suspected melanoma. Identifying this critical biomarker helps clinicians determine if their patient has melanoma.
Approximately half of all patients with metastatic breast cancer express low levels of HER2 receptor protein. The newly launched PATHWAY anti-HER2 (4B5) test is the only FDA-approved companion diagnostic indicated as an aid in the assessment of HER2 low status in these patients. Historically these patients have simply been classified as HER2-negative, leaving them with few treatment options. They may now be eligible for targeted therapy.
With the launch of additional COVID-19 tests, Roche also reinforced its position as a world-leading supplier of COVID-19 diagnostics:
The Elecsys IGRA SARS-CoV-2 test can help provide a deeper understanding of a person’s immune response to SARS-CoV-2 infection or vaccination. It will serve as an additional tool to make better-informed decisions around care, sanitary measures and treatment options – particularly important for at-risk patient groups.
(The test was launched in countries that accept the CE Mark.)
The next generation portfolio of SARS-CoV-2 Rapid Antigen tests (“2.0”) for self-testing and professional use feature innovative updates and enhanced performance, building on insights gained throughout the pandemic.
(These tests are used under the CE mark.)
Based on continuous analysis performed since the onset of the pandemic, all Roche molecular tests detect all SARS-CoV-2 variants.
![[Ad hoc announcement pursuant to Art. 53 LR] Roche records solid results for the first nine months of 2022 -2](http://lswlifescienceworld.com/wp-content/uploads/2025/06/lsw-bulletin-oct18-2022-3.jpg)
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
