New two-year Evrysdi data show improvement or maintenance of motor function in people with SMA, a progressive neuromuscular disease that can be fatal
The JEWELFISH study enrolled the broadest and most diverse patient population ever studied in an SMA trial
Longer-term safety data consistent with that previously seen in earlier trials and low study drop-out rate
Evrysdi has proven efficacy in babies, children and adults, with more than 7,000 patients treated to date worldwide
Basel, 12 October 2022- Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced new two-year data from the JEWELFISH study evaluating Evrysdi® (risdiplam) in people with Type 1, 2 or 3 SMA aged 6 months to 60 years at time of enrolment. Patients had been previously treated with other approved or investigational SMA-targeting therapies, including nusinersen (Spinraza(R)) or onasemnogene abeparvovec (Zolgensma(R)). Data showed Evrysdi improved or maintained motor function and led to rapid increases in SMN protein levels which were sustained after 2-years of treatment. These data will be presented at the 27th World Muscle Society (WMS) congress, 11-15 October 2022.
“The consistent safety profile and exploratory efficacy we have seen in the JEWELFISH study, the largest ever conducted in previously treated patients, reinforces Evrysdi as a meaningful treatment option across SMA populations,” said Dr. Claudia Chiriboga, Professor of Neurology and Pediatrics, Department of Neurology, Columbia University Medical Center, New York, USA. “The findings add to our confidence when making treatment decisions for previously-treated patients in need.”
The JEWELFISH study enrolled the broadest and most diverse patient population ever studied in an SMA trial. Of the 174 people enrolled, 36% (n=63) were adults, 63% (n=105) had a Hammersmith Functional Motor Scale Expanded (HFMSE) score of less than 10 at baseline, meaning their disease was very severe, and 83% (n=139) had scoliosis. Forty-four percent (n=76) of those enrolled had previously been treated with nusinersen (Spinraza), 41% (n=71) with olesoxime*, 8% (n=14) with onasemnogene abeparvovec (Zolgensma) and 7% (n=13) with RG7800*.
People with SMA are unable to produce enough survival motor neuron (SMN) protein, leading to debilitating and potentially fatal muscle weakness. The study showed Evrysdi led to a two-fold increase in median SMN protein levels versus baseline after 4 weeks of treatment in all patient groups, irrespective of previous treatment. The SMN protein levels achieved after 4 weeks of treatment were maintained for over two years.
Observed through exploratory efficacy endpoints, the study also suggests maintenance of motor function was sustained at two-years of treatment as measured by change from baseline in Motor Function Measure 32 (MFM-32), Revised Upper Limb Module (RULM) and HFMSE total scores compared to the natural history of SMA in untreated patients. A recent survey conducted by patient advocacy group SMA Europe, more than 96% of people with SMA viewed disease stabilization as progress in terms of their expectations of treatment.
“These important data demonstrate the safety and efficacy of Evrysdi in a broad, real-world population of people previously treated with an SMA-targeting therapy, ” said Levi Garraway, M.D, Ph.D, Roche’s Chief Medical Officer and Head of Global Products. “Those enrolled in JEWELFISH had very severe disease, with over 80% having scoliosis, so maintaining motor function–especially for a progressive disease–can be potentially life-changing.”
The overall adverse event (AE) and serious adverse event (SAE) profiles observed with Evrysdi treatment in JEWELFISH were reflective of underlying disease. The rate of AEs decreased by more than 50% between the first and second 6-month period, and then remained stable thereafter. The rate of SAEs, including pneumonia, decreased throughout the 24-month period, with a total reduction of more than 50% by the second year. The most common AEs (reported in ≥ 12% of all patients:n=173) were pyrexia (24%), upper respiratory tract infection (21%), headache (18%), nasopharyngitis (16%), diarrhoea (14%), nausea (13%) and cough (12%). The most common SAEs (reported in less than >2% of all patients) were pneumonia (3%) respiratory failure (2%), respiratory distress (2%), lower respiratory tract infection (2%) and upper respiratory tract infection (2%). The most common AEs/SAEs were consistent with those observed in treatment-naïve patients in our other three trials. Low rates of discontinuation from the study were observed, with a 5% rate per year over the 24-month period.
Roche leads the clinical development of Evrysdi as part of a collaboration with the SMA Foundation and PTC Therapeutics.
Evrysdi is a survival motor neuron 2 (SMN2) splicing modifier designed to treat SMA caused by mutations in chromosome 5q that lead to SMN protein deficiency. Evrysdi is administered daily at home in liquid form by mouth or by feeding tube.
Evrysdi is designed to treat SMA by increasing and sustaining the production of the SMN protein in the central nervous system (CNS) and peripheral tissues. SMN protein is found throughout the body and is critical for maintaining healthy motor neurons and movement.
Evrysdi was granted PRIME designation by the European Medicines Agency (EMA) in 2018 and Orphan Drug Designation by the U.S. Food and Drug Administration in 2017. In 2021 Evrysdi was awarded Drug Discovery of the Year by the British Pharmacological Society as well as the Society for Medicines Research award for Drug Discovery. Evrysdi is currently approved in 91 countries and the dossier is under review in a further 18 countries.
Evrysdi is currently being evaluated in five multicentre trials in people with SMA:
FIREFISH (NCT02913482) – an open-label, two-part pivotal clinical trial in infants with Type 1 SMA. Part 1 was a dose-escalation study in 21 infants with the primary objective of assessing the safety profile of risdiplam in infants and determining the dose for Part 2. Part 2 is a pivotal, single-arm study of risdiplam in 41 infants with Type 1 SMA treated for 2 years, followed by an open-label extension. Enrolment for Part 2 was completed in November 2018. The primary objective of Part 2 was to assess efficacy as measured by the proportion of infants sitting without support after 12 months of treatment, as assessed by the Gross Motor Scale of the Bayley Scales of Infant and Toddler Development – Third Edition (BSID-III) (defined as sitting without support for 5 seconds). The study met its primary endpoint.
SUNFISH (NCT02908685) – SUNFISH is a two-part, double-blind, placebo controlled pivotal study in people aged 2-25 years with Types 2 or 3 SMA. Part 1 (n=51) determined the dose for the confirmatory Part 2. Part 2 (n=180) evaluated motor function using the total score of Motor Function Measure 32 (MFM-32) at 12 months. MFM-32 is a validated scale used to evaluate fine and gross motor function in people with neurological disorders, including SMA. The study met its primary endpoint.
JEWELFISH (NCT03032172) – an open-label exploratory trial designed to assess the safety, tolerability, pharmacokinetics and pharmacodynamics in people with SMA aged 6 months to 60 years who received other investigational or approved SMA therapies for at least 90 days prior to receiving Evrysdi. The study has completed recruitment (n=174).
RAINBOWFISH (NCT03779334) – an open-label, single-arm, multicentre study, investigating the efficacy, safety, pharmacokinetics, and pharmacodynamics of risdiplam in babies (~n=25), from birth to six weeks of age (at first dose) with genetically diagnosed SMA who are not yet presenting with symptoms. The study is ongoing.
MANATEE (NCT05115110) – a global phase 2/3 clinical study to evaluate the safety and efficacy of GYM329 (RO7204239), an anti-myostatin molecule targeting muscle growth, in combination with Evrysdi for the treatment of SMA in patients 2-10 years of age. The FDA Office of Orphan Products Development granted GYM329 Orphan Drug Designation for the treatment of patients with SMA in December 2021. The study is currently recruiting.
SMA is a severe, progressive neuromuscular disease that can be fatal. It affects approximately one in 10,000 babies and is the leading genetic cause of infant mortality. SMA is caused by a mutation of the survival motor neuron 1 (SMN1) gene, which leads to a deficiency of SMN protein. This protein is found throughout the body and is essential to the function of nerves that control muscles and movement. Without it, nerve cells cannot function correctly, leading to muscle weakness over time. Depending on the type of SMA, an individual’s physical strength and their ability to walk, eat or breathe can be significantly diminished or lost.
Neuroscience is a major focus of research and development at Roche. Our goal is to pursue groundbreaking science to develop new treatments that help improve the lives of people with chronic and potentially devastating diseases.
Roche is investigating more than a dozen medicines for neurological disorders, including multiple sclerosis, spinal muscular atrophy, neuromyelitis optica spectrum disorder, Alzheimer’s disease, Huntington’s disease, Parkinson’s disease and Duchenne muscular dystrophy. Together with our partners, we are committed to pushing the boundaries of scientific understanding to solve some of the most difficult challenges in neuroscience today.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Broad and trusted portfolio of COVID-19 rapid tests feature innovative updates and enhanced performance, building on insights gained throughout the pandemic
All tests now work seamlessly with navify® Pass, Roche’s digital solution that allows individuals and healthcare professionals to immediately store, display, and share COVID-19 vaccination status and test results through a unique data matrix
Roche maintains the capacity to provide tens of millions tests each month to meet sustained high worldwide demand from individuals and healthcare professionals
Basel, 12 October 2022- Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the launch of its next-generation portfolio SARS-CoV-2 rapid antigen tests (“2.0”) for self-testing and professional use in countries accepting the CE Mark. Distribution of the new rapid test portfolio is projected to begin in the coming weeks.
In doing so, Roche builds upon one of the broadest portfolios of COVID-19 rapid testing solutions with three new test configurations that feature enhanced performance through the use of an improved capture antibody and the addition of new digital data sharing capabilities to all tests. Using nasopharyngeal and nasal swab samples, respectively, the tests deliver reliable results in as few as 15 minutes, aiding in the diagnosis of SARS-CoV-2 infection in individuals with or without symptoms consistent with COVID-19.
“The continued evolution of the SARS-CoV-2 virus and COVID-19 pandemic reinforce the need for individuals and healthcare systems to have access to the most reliable rapid testing solutions possible,” said Thomas Schinecker, CEO, Roche Diagnostics. “As a leader in diagnostic innovation, we are proud to be one of the first among our industry to apply insights gained from the past two years toward a next-generation rapid test portfolio. These tests are ready to support global society as we navigate this next phase of the pandemic.”
The three tests set to launch are the following:
Professional use:
SARS-CoV-2 Rapid Antigen Test 2.0 (nasopharyngeal sampling)
SARS-CoV-2 Rapid Antigen Test 2.0 Nasal (nasal sampling)
Self-Test:
SARS-CoV-2 Antigen Self Test Nasal (nasal sampling)
Roche maintains the capacity to provide several tens of millions of each of the tests per month, with the option to quickly build up additional capacity if needed, to address sustained high worldwide demand for rapid testing from governments, healthcare systems and individuals. The next-generation test portfolio will be introduced in partnership with SD Biosensor Inc., with whom Roche has a global distribution agreement and previously launched a range of rapid antigen and antibody tests in more than 50 countries worldwide.
The tests join Roche Diagnostics’s broader COVID-19 portfolio, which includes a wide range of molecular, serological and digital solutions that help diagnose and manage COVID-19 during the initial stages of infection, during the recovery phase, and following the resolution of infection.
Lab testing showed that all three tests can qualitatively detect major variants of concern including Delta and Omicron variants. Emerging variants are continuously monitored.
The SARS-CoV-2 Rapid Antigen Test 2.0 is a rapid chromatographic immunoassay for the qualitative detection of the nucleocapsid protein of SARS-CoV-2 present in human nasopharyngeal swab samples. In a prospective clinical studyA , the SARS-CoV-2 Rapid Antigen 2.0 showed a relative sensitivity of 99.00% (95% CI: 94.55 - 99.97%) and a relative specificity of 99.75% (95% CI: 98.62 - 99.99%). In total, 100 PCR-positive and 402 PCR-negative subjects participated in this study. This included 320 asymptomatic subjects, among whom 34 were positive and 286 were negative; and 182 symptomatic individuals, among whom 66 were positive and 116 were negative. This test is intended as an aid in the diagnosis of SARS‑CoV‑2 infection in individuals with or without symptoms consistent with COVID‑19. This product is intended for professional use in laboratory and near‑patient testing environments. This product is not intended for self‑testing.
The SARS-CoV-2 Rapid Antigen Test 2.0 Nasal is a rapid chromatographic immunoassay for the qualitative detection of the nucleocapsid protein of SARS-CoV-2 present in human nasal swab samples. In prospective clinical studiesA,B, the SARS-CoV-2 Rapid Antigen 2.0 Nasal showed a relative sensitivity of 95.80% (95% CI: 91.09 - 98.44%) and a relative specificity of 100% (95% CI: 99.25 - 100%). In total, 143 PCR-positive and 487 PCR-negative subjects participated in these studies. This included 320 asymptomatic subjects, among whom 34 were positive and 286 were negative; and 310 symptomatic individuals, among whom 109 were positive and 201 were negative. This test is intended as an aid in the diagnosis of SARS‑CoV‑2 infection in individuals with or without symptoms consistent with COVID‑19. This product is intended for professional use in laboratory and near‑patient testing environments. This product is not intended for self‑testing.
The enhanced SARS-CoV-2 Antigen Self Test Nasal is a so-called lateral flow test for the qualitative detection of SARS-CoV-2 nucleocapsid antigen in human nasal samples. This test is used to detect antigens of the SARS-CoV-2 virus in individuals suspected of having COVID-19. It is designed as a self-test for patients. In prospective clinical studiesA, B, the SARS-CoV-2 Antigen Self Test Nasal showed a relative sensitivity of 95.8% (95% CI: 91.09 - 98.44%) and a relative specificity of 100% (95% CI: 99.25 - 100%). In total, 143 PCR-positive and 487 PCR-negative subjects participated in these studies. This included 320 asymptomatic subjects, among whom 34 were positive and 286 were negative; and 310 symptomatic individuals, among whom 109 were positive and 201 were negative. 128 of the subjects were lay-users, sampling and testing themselves or another lay-user, for example their underaged children
An antigen test detects proteins which are structural or functional components of a pathogen and are very specific to that pathogen. In this case, the test would provide a qualitative “yes/no” answer on the presence of the antigen in the patient sample and can be offered as a rapid strip test that is performed by healthcare professionals at the point of care or by individuals at home. If the target antigen (in this case the nucleocapsid protein) is present in sufficient concentrations in the sample, it will bind to specific antibodies and generate a visually detectable signal on the test strip, typically with results ready in 15 minutes.2 A rapid antigen test can reliably detect individuals with a high viral load allowing healthcare professionals to quickly identify those patients at the greatest risk of spreading the infection.
In general, antigen tests have a high specificity, though are not as sensitive as molecular tests that amplify the target DNA or RNA sequence in order to generate a (semi-)quantifiable signal to indicate the presence of the pathogen in a sample. Therefore, to make up for the potential decrease in sensitivity of an antigen test, negative results should be analysed together with additional information, such as the individual’s exposure history, clinical symptoms, and additional test results to help guide the diagnosis and subsequent treatment of the patient.
As a leading healthcare company, we are doing all we can to support countries in their fight against COVID-19 and minimising its impact. That is why we are working with governments, policy makers, healthcare professionals and others to help contain the COVID-19 pandemic and make sure patients continue to receive the tests, treatment and care they need.
The pandemic has profoundly raised awareness of the role diagnostics play in COVID-19 diagnosis, treatment development and disease management. Roche has developed and launched more than 20 COVID-19 diagnostics solutions, including polymerase chain reaction (PCR) and rapid antigen and antibody tests. Our solutions serve the entire diagnostic continuum, from high-throughput laboratories to point-of-care and home self-testing, and cover all currently known variants. To help meet global demand, we have supplied more than 1.5 billion tests for COVID-19 since March 2020.
Roche continues to evaluate its existing therapeutic portfolio and is researching future options to help benefit patients with COVID-19. Our IL-6 inhibitor Actemra®/RoActemra® (tocilizumab) has been approved for patients hospitalised with severe COVID-19 in more than 30 countries including the European Union and is authorised for emergency use in the United States. The World Health Organization has prequalified Actemra for use in patients with severe COVID-19, facilitating its availability in low- and middle-income countries. In addition, we have been improving access to Actemra by introducing an international differentiated pricing strategy, providing the medicine at cost for use in low- and middle-income countries and non asserting patents in these regions during the pandemic.
We have also been partnering with Regeneron to jointly develop the antibody combination Ronapreve™ (casirivimab and imdevimab, known as REGEN-COV™ in the US). It has been approved in multiple territories including the European Union, Japan, and Switzerland and authorised for emergency or temporary pandemic use in many countries including the US. The antibody combination has been made available to patients in more than 60 countries, across many geographies including low and middle income countries. As the virus continues to evolve, we are constantly monitoring Ronapreve’s activity against emerging variants of concern, and will share results with health authorities as soon as possible.
Our utmost goal remains to be a trusted partner who acts with urgency to save and improve the lives of patients with COVID-19 and to reduce its burden on society.
SD Biosensor is a global in-vitro diagnostic company focused on the development of immunoassay and molecular diagnostic products at the POC. Founded in 2010, SD Biosensor has continued to research and develop products that can aid in the fast and accurate diagnosis of patients across the testing journey. Through these innovative products, they are striving to become a leading global in vitro diagnostics company.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The PRAME (EPR20330) Antibody evaluates PRAME protein expression from patients with suspected melanoma.
Understanding if the PRAME protein is expressed helps enable more informed clinical decisions and may improve patient outcomes.
The PRAME (EPR20330) Antibody is the latest addition to Roche’s robust dermatology portfolio with more than 50 dermatology biomarkers designed to meet a broad range of diagnostic needs.
Basel, 11 October 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the launch of Anti-PRAME (EPR 20330) Rabbit Monoclonal Primary Antibody to identify PRAME protein expression in tissue samples from patients with suspected melanoma.
Because the PRAME (PReferentially expressed Antigen in MElanoma) protein is expressed in most melanomas, the PRAME (EPR20330) Antibody* is used as an aid to differentiate between benign and malignant lesions to help improve diagnostic decisions. If PRAME expression is detected, this suggests that the lesion is malignant.
“Every four minutes, one person dies from skin cancer. However, when detected early, localised melanoma is highly curable with a simple surgical excision,” said Thomas Schinecker, CEO of Roche Diagnostics. “Identifying this critical biomarker helps clinicians determine if their patient has melanoma. We’re proud to add the PRAME assay to our already comprehensive test menu of dermatology biomarkers that help inform a patient’s diagnosis and treatment.”
Melanoma is an aggressive skin cancer that originates from melanocytes, which are cells in the skin and eyes that produce and contain melanin. Melanoma develops when unrepaired DNA damage to melanocytes triggers mutations that cause the melanocytes to grow rapidly and form malignant tumours. When detected early, the 5-year survival rate is 99 percent.1
The first test that is used to evaluate a potential melanocytic lesion is a hematoxylin and eosin (H&E) stain of the patient’s tissue sample. However, not all melanomas are easily diagnosed using H&E alone. Studies suggest that detection of PRAME expression by immunohistochemistry (IHC) complements findings from routinely used tests and enables more informed clinical decisions and improved patient outcomes.2-4 In particularly challenging cases, use of PRAME IHC may highlight abnormal cells expressing the PRAME protein and provide more confidence in diagnosis of melanoma.
More information about PRAME will be presented at the International Academy of Pathology 2022 World Congress in Sydney, Australia on 14 October 2022 in a session titled “The diagnostic approach to melanocytic lesions”.
The PRAME (EPR20330) Antibody is fully automated on the full line of Roche BenchMark IHC/ISH instruments. This antibody is validated for use using OptiView DAB IHC Detection, ultraView Universal DAB Detection & ultraView Universal Alkaline Phosphatase Red Detection Kits giving added flexibility to laboratories and pathologists' preferences.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
New positive data from Evrysdi, a treatment for spinal muscular atrophy (SMA), a progressive neuromuscular disease that can be fatal
Data from the gene therapy programme for Duchenne muscular dystrophy (DMD), a progressive disease that leads to premature death, reinforce confidence in the most advanced Phase 3 study currently underway
Study designs of two new trials in generalised myasthenia gravis (gMG), a rare chronic autoimmune disease, and facioscapulohumeral muscular dystrophy (FSHD), a genetic muscle disorder
Basel, 05 October 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that new data from its industry-leading neuromuscular portfolio will be presented at the World Muscle Society (WMS) congress, 11th-15th October 2022. These data demonstrate Roche’s commitment to advancing clinical understanding and supporting the development of treatments for people living with neuromuscular disorders.
“The continued expansion of our neuromuscular portfolio demonstrates our commitment to developing innovative medicines for a range of neurological disorders,” said Levi Garraway, M.D., Ph. D., Roche’s Chief Medical Officer and Head of Global Product Development. “Through our ongoing research and development and our strong partnerships with patient communities, we continue to address the needs of people with rare neuromuscular conditions.”
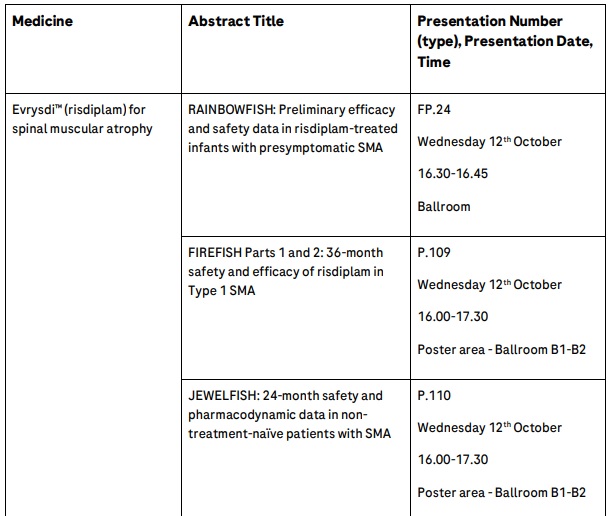
SMA is a severe, progressive neuromuscular disease that can be fatal. It is the leading genetic cause of infant mortality, affecting approximately one in 10,000 babies. For the first time, Roche will present new 2-year exploratory efficacy data from the JEWELFISH study in a broad range of patients previously treated with an SMA-targeting therapy, including nusinersen (SpinrazaR) or onasemnogene abeparvovec (ZolgensmaR).
Additional data from Evrysdi’s comprehensive clinical development programme will also be presented, including:
Preliminary efficacy and safety data from the RAINBOWFISH study, in pre-symptomatic babies from birth to 6 weeks of age (at first dose), which showed that all babies treated with Evrysdi for one-year or more were alive without permanent ventilation, maintained swallowing and feeding abilities, and had not required hospitalisation.
Three-year data from the SUNFISH study, further highlighting the long-term efficacy and safety profile of Evrysdi in a broad population of children, teenagers and adults with SMA.
Three-year pooled safety and efficacy data from Part 1 and Part 2 of the FIREFISH study which showed that after three years of treatment at the pivotal dose (n=58), 84% of infants were alive and did not require permanent ventilation. Overall, babies maintained or improved their motor skills in terms of developmental milestones and motor function between Month 24 and Month 36.
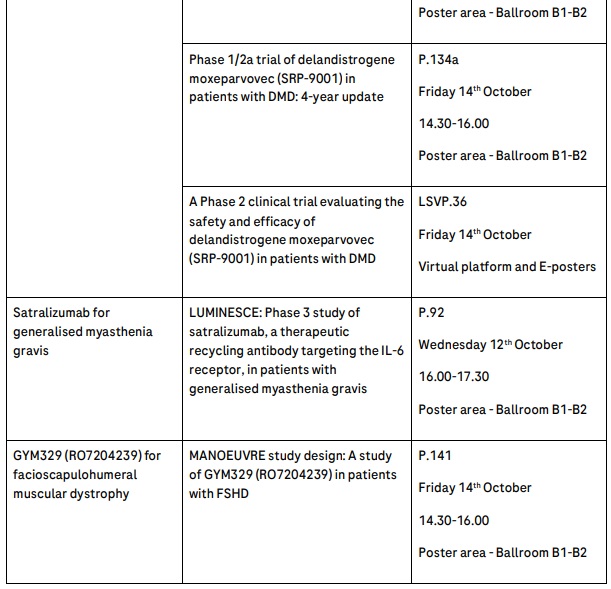
DMD is a rare X-linked, progressive neuromuscular disease caused by mutations in the DMD gene that disrupts the production of functional dystrophin protein, leading to a loss of muscle function and premature death. It is one of the most common fatal genetic disorders, affecting approximately one in every 3,500 to 5,000 male births worldwide. Results from three ongoing clinical trials of gene therapy delandistrogene moxeparvovec in DMD will be presented:
Long-term, four-year data from Study 101, an open-label Phase 1/2a study evaluating the safety of delandistrogene moxeparvovec in four ambulatory participants aged between 4-8 years old with DMD. The enduring response and safety observed in the study support the continuation of clinical trials assessing delandistrogene moxeparvovec using single dose gene therapy in patients with DMD.
One-year data from ENDEAVOR, an open-label Phase 1b study evaluating the expression and safety of commercially representative delandistrogene moxeparvovec in four cohorts of DMD patients, representing different stages of disease progression. One-year safety and functional data and 12-week expression data from Cohort 1 will be presented. These data reinforce our confidence in the probability of success of EMBARK, our first Phase 3 study using intended commercial process material, which is now fully enrolled.
Integrated analyses comparing data from patients treated with delandistrogene moxeparvovec with an external comparator cohort will be presented, including collective safety data from Study 101, Study 102 and ENDEAVOR.
Myasthenia gravis is a rare chronic autoimmune, neuromuscular disease that causes weakness in skeletal muscles. It most commonly affects the muscles that control the eyes and eyelids, facial expressions, chewing, swallowing and speaking as well as limb musculature and muscles of respiration.
Roche will present the study design and methodology for LUMINESCE, a Phase 3 randomised, double-blind, placebo-controlled, multicentre study evaluating the efficacy and safety of satralizumab versus placebo on function in daily life in people with gMG. The LUMINESCE study is enrolling a large and broad population of 240 people with gMG, aged 12 years and older, including patients with various disease-causing autoantibodies.
FSHD is a rare autosomal dominant genetic disorder characterised by progressive weakening of the skeletal muscles in the face, shoulders, arms, trunk and limbs. There is currently no approved therapy for this disease.
Roche will present the study design for MANOEUVRE, a multi-center, randomised, placebo-controlled, double-blind, Phase 2 study, that will investigate the effect of GYM329 (RO7204239), an investigational anti-myostatin antibody, in patients with FSHD.
The full range of data from Roche’s clinical development programme in neuromuscular disease being presented at WMS 2022 include:

Full session details and data presentations listing for the WMS 2022 Hybrid Congress can be found at the meeting website: www.worldmusclesociety.org
Follow Roche on Twitter via @Roche and keep up to date with WMS 2022 Congress news and updates by using the hashtag #RocheAtWMS2022, #WMS and #neuromuscular
Evrysdi is a survival motor neuron 2 (SMN2) splicing modifier designed to treat SMA caused by mutations in chromosome 5q that lead to survival motor neuron (SMN) protein deficiency. Evrysdi is administered daily at home in liquid form by mouth or by feeding tube.
Evrysdi is designed to treat SMA by increasing and sustaining the production of the SMN protein in the central nervous system (CNS) and peripheral tissues. SMN protein is found throughout the body and is critical for maintaining healthy motor neurons and movement.
Evrysdi was granted PRIME designation by the European Medicines Agency (EMA) in 2018 and Orphan Drug Designation by the U.S Food and Drug Administration in 2017. In 2021, Evrysdi was awarded Drug Discovery of the Year by the British Pharmacological Society as well as the Society for Medicines Research award for Drug Discovery. Evrysdi is currently approved in 91 countries and the dossier is under review in a further 18 countries. To date, more than 7,000 adults, children and babies have been treated with Evrysdi worldwide.
Delandistrogene moxeparvovec (SRP-9001; rAAVrh74.MHCK7.micro-dystrophin) is an investigational gene therapy aimed to deliver the micro-dystrophin-encoding transgene directly to the skeletal and cardiac muscle for the targeted production of the SRP-9001-dystrophin protein to enable a durable clinical response. In December 2019, Roche partnered with Sarepta to combine Roche’s global reach and drug development and regulatory expertise to accelerate access to delandistrogene moxeparvovec for patients
ENSPRYNG, which was designed by Chugai, a member of the Roche Group, is a humanised monoclonal antibody that targets interleukin-6 (IL-6) receptor activity. ENSPRYNG was designed using novel recycling antibody technology which, compared to conventional technology, allows for longer duration of the antibody and subcutaneous dosing every four weeks.
Positive Phase III results for ENSPRYNG, as both monotherapy and in combination with baseline immunosuppressive therapy, demonstrate that IL-6 inhibition is an effective therapeutic approach for neuromyelitis optica spectrum disorder ( NMOSD). ENSPRYNG is currently approved for NMOSD in 72 countries with further applications under review with numerous regulators. Roche continues to investigate ENSPRYNG in further indications including generalised myasthenia gravis (gMG), Myelin Oligodendrocyte Glycoprotein Antibody-associated Disease (MOGAD) and Autoimmune Encephalitis (AIE).
ENSPRYNG has been designated as an orphan drug for gMG, MOGAD, AIE (NMDAR) and NMOSD in the United States. ENSPRYNG has also been designated as an orphan drug for NMOSD in Europe, Russia and Japan. In addition, it was granted Breakthrough Therapy Designation for the treatment of NMOSD by the FDA in December 2018.
GYM329 is an investigational anti-myostatin antibody that is designed to increase muscle growth by binding to inhibiting myostatin, a natural negative regulator of muscle growth. By binding to the protein, GYM329 was created with the aim of blocking the function of myostatin, therefore helping muscles to grow in size and strength.
Neuroscience is a major focus of research and development at Roche. Our goal is to pursue groundbreaking science to develop new treatments that help improve the lives of people with chronic and potentially devastating diseases.
Roche has both approved and investigational medicines across multiple sclerosis, spinal muscular atrophy, neuromyelitis optica spectrum disorder, myasthenia gravis, Alzheimer’s disease, Huntington’s disease, Parkinson’s disease and Duchenne muscular dystrophy. Together with our partners, we are committed to pushing the boundaries of scientific understanding to solve some of the most difficult challenges in neuroscience today.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Approximately half of all patients with metastatic breast cancer (mBC) express low levels of HER2. These patients with HER2 low status may now be eligible for a targeted treatment, which could significantly improve their outcomes
The PATHWAY anti-HER2 (4B5) test is the only FDA approved companion diagnostic indicated as an aid in the assessment of HER2 low status in metastatic breast cancer patients. These patients may now consider ENHERTU as a treatment option based on the results of the Phase 3 DESTINY-Breast04 trial.
Basel, 4 October 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that the US Food and Drug Administration (FDA) approved the PATHWAY anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody* to identify metastatic breast cancer patients with low HER2 expression for whom ENHERTU® (fam-trastuzumab deruxtecan-nxki) may be considered as a targeted treatment. ENHERTU is a specifically engineered HER2-directed antibody drug conjugate (ADC) being jointly developed and commercialised by AstraZeneca and Daiichi Sankyo.
HER2 is a receptor protein that helps cancer cells grow quickly. To determine a patient’s HER2 status, pathologists evaluate, or score, the level of HER2 receptor protein expressed in breast cancer tissue samples. If a patient's tumour expresses high levels of HER2, the patient is identified as HER2-positive and may be considered for HER2-targeted treatment. However, half of all patients with metastatic breast cancer express low levels of HER2 which historically classified them as HER2-negative.
The PATHWAY anti-HER2 (4B5) test now includes a scoring algorithm that helps pathologists to identify “low expressors” of HER2, assigning a HER2 low status to this group of patients. With this lower cutoff, the test is able to identify patients who may benefit from ENHERTU as a treatment option.
“Roche is proud to lead the way in HER2 diagnostics through critical innovations that support the identification of patients who may benefit from novel HER2-targeted therapies,” said Thomas Schinecker, CEO of Roche Diagnostics. “Previously, metastatic breast cancer patients with a lower level of HER2 expression were considered to be part of the HER2-negative population and had no HER2-targeted treatment options. Now, they may be eligible for a HER2-targeted therapy, significantly increasing the number of patients who could have improved outcomes.”
The PATHWAY anti-HER2 (4B5) test was used as part of the DESTINY-Breast04 trial sponsored by AstraZeneca and Daiichi Sankyo to identify patients whose tumours expressed low levels of HER2 protein. The trial reported a 50% reduction in the risk of disease recurrence or death and an overall gain of six months over standard of care in patients treated with ENHERTU whose tumours had low levels of HER2 expression.
Breast cancer has surpassed lung cancer as the most commonly diagnosed cancer, with an estimated 2.3 million new cases diagnosed worldwide each year. More than 620,000 people die from breast cancer every year.4,5
The FDA approval of the new HER2 low indication expands on the intended use for Roche’s proven, on-market PATHWAY anti-HER2 (4B5) test, delivering timely, clear and confident results. The launch exemplifies Roche’s commitment to continuing to innovate integrated, high medical value solutions that help to advance personalised healthcare.
Roche’s pre-diluted PATHWAY anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody, used in combination with the fully automated BenchMark IHC/ISH slide staining instrument, standardises all immunohistochemistry (IHC) processes from baking through staining, and reduces the possibility of human error.5 It also minimises inherent variability resulting from individual reagent dilution and other processes found in manual and semi-automated IHC methods. The Roche HER2 (4B5) clone achieves consistently high proficiency assessment scores compared to other clones6 and demonstrates high concordance with HER2 FISH7,8, empowering laboratories to employ the most widely adopted and reliable HER2-IHC primary antibody.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Approximately half of all patients with metastatic breast cancer (mBC) express low levels of HER2. These patients with HER2 low status may now be eligible for a targeted treatment, which could significantly improve their outcomes
The PATHWAY anti-HER2 (4B5) test is the only FDA approved companion diagnostic indicated as an aid in the assessment of HER2 low status in metastatic breast cancer patients. These patients may now consider ENHERTU as a treatment option based on the results of the Phase 3 DESTINY-Breast04 trial.
Basel, 4 October 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that the US Food and Drug Administration (FDA) approved the PATHWAY anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody* to identify metastatic breast cancer patients with low HER2 expression for whom ENHERTU® (fam-trastuzumab deruxtecan-nxki) may be considered as a targeted treatment. ENHERTU is a specifically engineered HER2-directed antibody drug conjugate (ADC) being jointly developed and commercialised by AstraZeneca and Daiichi Sankyo.
HER2 is a receptor protein that helps cancer cells grow quickly. To determine a patient’s HER2 status, pathologists evaluate, or score, the level of HER2 receptor protein expressed in breast cancer tissue samples. If a patient's tumour expresses high levels of HER2, the patient is identified as HER2-positive and may be considered for HER2-targeted treatment. However, half of all patients with metastatic breast cancer express low levels of HER2 which historically classified them as HER2-negative.
The PATHWAY anti-HER2 (4B5) test now includes a scoring algorithm that helps pathologists to identify “low expressors” of HER2, assigning a HER2 low status to this group of patients. With this lower cutoff, the test is able to identify patients who may benefit from ENHERTU as a treatment option.
“Roche is proud to lead the way in HER2 diagnostics through critical innovations that support the identification of patients who may benefit from novel HER2-targeted therapies,” said Thomas Schinecker, CEO of Roche Diagnostics. “Previously, metastatic breast cancer patients with a lower level of HER2 expression were considered to be part of the HER2-negative population and had no HER2-targeted treatment options. Now, they may be eligible for a HER2-targeted therapy, significantly increasing the number of patients who could have improved outcomes.”
The PATHWAY anti-HER2 (4B5) test was used as part of the DESTINY-Breast04 trial sponsored by AstraZeneca and Daiichi Sankyo to identify patients whose tumours expressed low levels of HER2 protein. The trial reported a 50% reduction in the risk of disease recurrence or death and an overall gain of six months over standard of care in patients treated with ENHERTU whose tumours had low levels of HER2 expression.
Breast cancer has surpassed lung cancer as the most commonly diagnosed cancer, with an estimated 2.3 million new cases diagnosed worldwide each year. More than 620,000 people die from breast cancer every year.4,5
The FDA approval of the new HER2 low indication expands on the intended use for Roche’s proven, on-market PATHWAY anti-HER2 (4B5) test, delivering timely, clear and confident results. The launch exemplifies Roche’s commitment to continuing to innovate integrated, high medical value solutions that help to advance personalised healthcare.
Roche’s pre-diluted PATHWAY anti-HER2/neu (4B5) Rabbit Monoclonal Primary Antibody, used in combination with the fully automated BenchMark IHC/ISH slide staining instrument, standardises all immunohistochemistry (IHC) processes from baking through staining, and reduces the possibility of human error.5 It also minimises inherent variability resulting from individual reagent dilution and other processes found in manual and semi-automated IHC methods. The Roche HER2 (4B5) clone achieves consistently high proficiency assessment scores compared to other clones6 and demonstrates high concordance with HER2 FISH7,8, empowering laboratories to employ the most widely adopted and reliable HER2-IHC primary antibody.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Phase III data that showed people with nAMD and DME treated with Vabysmo up to every four months achieved similar outcomes compared to receiving treatment every two months with aflibercept
In addition, patients treated with Vabysmo received up to 33% fewer median number of injections compared to aflibercept
Reducing the number of eye injections over time could offer a less burdensome treatment schedule for individuals, their caregivers and healthcare systems
Vabysmo simultaneously targets and inhibits two disease pathways involving Ang-2 and VEGF-A linked to a number of vision-threatening retinal conditions
Basel, 19 September 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the European Commission (EC) approved Vabysmo® (faricimab) for the treatment of neovascular or ‘wet’ age-related macular degeneration (nAMD) and visual impairment due to diabetic macular edema (DME). These retinal conditions are two of the leading causes of vision loss worldwide, affecting more than 40 million people 1,2,3,4
“Many people with nAMD and DME struggle to keep up with the monthly eye injections and physician visits, often associated with current standards of care, and unfortunately their vision may suffer as a result of undertreatment,” said Prof Ramin Tadayoni, head of the ophthalmology department, Lariboisière, Saint-Louis and Rothschild Hospitals, Paris, France, and European Society of Retina Specialists (EURETINA) president elect. “For people in Europe living with these conditions, today’s approval offers the first new mechanism of action in over a decade; one which could improve and protect their vision with fewer injections over time.”
Vabysmo is the only injectable eye medicine approved in Europe with phase III studies supporting treatment at intervals of up to four months for people living with nAMD and DME.5,6,7 With the potential to require fewer eye injections over time, while also improving and maintaining vision and anatomy, Vabysmo could offer a less burdensome treatment schedule for individuals, their caregivers and healthcare systems 6,7,8,9
“The approval of Vabysmo in Europe is the result of years of pioneering research from Roche ophthalmologists and scientists, who are deeply committed to improving outcomes for people with retinal conditions,” said Levi Garraway, M.D., PhD., Roche’s Chief Medical Officer and Head of Global Product Development. “We are delighted to offer people in Europe this first-of-its-kind treatment option and are working to bring Vabysmo to people with nAMD and DME as soon as possible.”
Today’s approval is based on results across four phase III studies in two indications, involving 3,220 patients: TENAYA and LUCERNE in nAMD at year one, and YOSEMITE and RHINE in DME up to two years. The studies showed that people treated with Vabysmo, given at intervals of up to four months, achieved similar vision gains and anatomical improvements compared to aflibercept given every two months.6,7,8 The totality of data across all four studies at two years showed that more than 60% of people treated with Vabysmo were able to extend treatment to every four months, while improving and maintaining vision. Additionally, up to two years, people with nAMD and DME treated with Vabysmo received 33% (10 vs. 15) and 21% (11 vs. 14) fewer median number of injections compared to aflibercept, respectively.6,9
Vabysmo, a bispecific antibody, is uniquely engineered to target and inhibit two disease pathways, linked to a number of vision-threatening retinal conditions, by neutralising angiopoietin-2 (Ang-2) and vascular endothelial growth factor-A (VEGF-A), to restore vascular stability. By independently blocking both pathways involving Ang-2 and VEGF-A, Vabysmo is designed to stabilise blood vessels and thereby reduce inflammation, leakage and abnormal vessel growth (neovascularisation) more than inhibition of VEGF-A alone.7 This sustained blood vessel stabilisation may improve disease control, vision and anatomical outcomes for longer.7,8
Vabysmo is now approved in the European Union and nine other countries around the world, including the US, Japan, and the UK, for people living with nAMD and DME, and submissions to other regulatory authorities are ongoing.5,10,11,12 Globally, more than 100,000 Vabysmo doses have been distributed for treatment of these conditions to date.13 Roche also continues to explore areas where Vabysmo has the potential to deliver additional benefits to patients, including retinal vein occlusion.
Roche has a robust phase III clinical development programme for Vabysmo. The programme includes AVONELLE-X, an extension study of TENAYA and LUCERNE, evaluating the long-term safety and tolerability of Vabysmo in neovascular or ‘wet’ age-related macular degeneration (nAMD), and RHONE-X, an extension study of YOSEMITE and RHINE, evaluating the long-term safety and tolerability of Vabysmo in diabetic macular edema (DME).14,15 Additionally, the BALATON and COMINO trials are underway, evaluating the efficacy and safety of Vabysmo in people with macular edema following retinal vein occlusion.16,17 Roche has also initiated the phase IV ELEVATUM study of Vabysmo in underrepresented patient populations with DME.
TENAYA (NCT03823287) and LUCERNE (NCT03823300) were two identical, randomised, multicentre, double-masked, global phase III studies evaluating the efficacy and safety of Vabysmo® (faricimab) compared to aflibercept in 1,329 people living with neovascular or ‘wet’ age-related macular degeneration (671 in TENAYA and 658 in LUCERNE).
Both studies met their primary endpoint, with Vabysmo given at intervals of up to every four months consistently shown to offer visual acuity gains and anatomical improvements that were non-inferior to aflibercept given every two months. A secondary endpoint in both studies measured the proportion of people in the Vabysmo arm that were treated on dosing schedules of every three or four months during the first year. Importantly, 46% (n=144/315) of those receiving Vabysmo in TENAYA and 45% (n=142/316) in LUCERNE were able to be treated every four months in the first year, and an additional 34% (n=107/315) and 33% (n=104/316), respectively, were able to be treated every three months. Combined, nearly 80% of people receiving Vabysmo were able to go three months or longer between treatments during the first year.
At two years, vision improvements were comparable across both treatment arms. In TENAYA, the average vision gains from baseline at two years were +3.7 eye chart letters in the Vabysmo arm and +3.3 letters in the aflibercept arm. In LUCERNE, the average vision gains from baseline at two years were +5.0 letters in the Vabysmo arm and +5.2 letters in the aflibercept arm. Furthermore, 59% (n=160/271) of Vabysmo patients in TENAYA and 67% (n=192/287) in LUCERNE achieved four-month dosing at two years. This is an increase over one-year results, which showed 46% (n=144/315) of Vabysmo patients in TENAYA and 45% (n=142/316) in LUCERNE achieved four-month dosing. An additional 15% (n=41/271) of Vabysmo patients in TENAYA and 14% (n=41/287) in LUCERNE achieved three-month dosing at two years. Combined, approximately 80% of Vabysmo patients were able to go three months or longer between treatments at the end of the second year.
Vabysmo was generally well tolerated in both studies, with a favourable benefit-risk profile. In TENAYA and LUCERNE, the most common adverse reactions (≥3% of people) included cataract, conjunctival haemorrhage, vitreous floaters, retinal pigment epithelial tears, increase of intraocular pressure and eye pain. Safety results were consistent across study arms.
Two-year data from TENAYA and LUCERNE were presented at the 2022 American Society of Retina Specialists Annual Scientific Meeting. These data will be submitted to the European Medicines Agency in due course.
YOSEMITE (NCT03622580) and RHINE (NCT03622593) were two identical, randomised, multicentre, double-masked, global phase III studies evaluating the efficacy and safety of Vabysmo® (faricimab) compared to aflibercept in 1,891 people with visual impairment due to diabetic macular edema (940 in YOSEMITE and 951 in RHINE).
Both studies met their primary endpoint, with Vabysmo given at intervals of up to every four months consistently shown to offer visual acuity gains and anatomical improvements that were non-inferior to aflibercept given every two months. A secondary endpoint in both studies measured the proportion of people in the Vabysmo treat-and-extend arm that achieved dosing schedules of every three or four months. Importantly, 53% (n=151/286) of those in the Vabysmo treat-and-extend arm in YOSEMITE and 51% (n=157/308) in RHINE achieved four-month dosing at the end of the first year, and an additional 21% (n=60/286) and 20% (n=63/308), respectively, achieved three-month dosing. At two years, the number of people in the Vabysmo treat-and-extend arm achieving four-month dosing increased to 60% (n=162/270) in YOSEMITE and 64% (n=185/287) in RHINE. An additional 18% (n=49/270) of people in YOSEMITE and 14% (n=39/287) in RHINE achieved three-month dosing. Combined, almost 80% of people in the Vabysmo treat-and-extend arm were able to go three months or longer between treatments at the end of the second year.
Vabysmo was generally well tolerated in both studies, with a favourable benefit-risk profile. In YOSEMITE and RHINE, the most common adverse reactions (≥3% of people) included cataract, conjunctival haemorrhage, vitreous floaters, increase of intraocular pressure and eye pain. Safety results were consistent across study arms.
Age-related macular degeneration (AMD) is a condition that affects the part of the eye that provides sharp, central vision needed for activities like reading.1,19 Neovascular or ‘wet’ AMD (nAMD) is an advanced form of the disease that can cause rapid and severe vision loss if left untreated.20,21 It develops when new and abnormal blood vessels grow uncontrolled under the macula, causing swelling, bleeding and/or fibrosis.21 Worldwide, around 20 million people are living with nAMD – the leading cause of vision loss in people over the age of 60 – and the condition will affect even more people around the world as the global population ages.
Affecting around 21 million people globally, diabetic macular edema (DME) is a vision-threatening retinal condition associated with blindness and decreased quality of life when left untreated.3,23 DME occurs when damaged blood vessels leak into and cause swelling in the macula – the central area of the retina responsible for the sharp vision needed for reading and driving.19,24 The number of people with DME is expected to grow as the prevalence of diabetes increases.
Vabysmo is the first bispecific antibody approved for the eye. It targets and inhibits two disease pathways linked to a number of vision-threatening retinal conditions, by neutralising angiopoietin-2 (Ang-2) and vascular endothelial growth factor-A (VEGF-A). Ang-2 and VEGF-A contribute to vision loss by destabilising blood vessels, causing new leaky blood vessels to form and increasing inflammation. By blocking pathways involving Ang-2 and VEGF-A, Vabysmo is designed to stabilise blood vessels.
Roche is focused on saving people’s eyesight from the leading causes of vision loss through pioneering therapies. Through our innovation in the scientific discovery of new potential drug targets, personalised healthcare, molecular engineering, biomarkers and continuous drug delivery, we strive to design the right therapies for the right patients.
We have the broadest retina pipeline in ophthalmology, which is led by science and informed by insights from people with eye diseases. Our pipeline includes gene therapies and treatments for geographic atrophy and other vision-threatening diseases, including rare and inherited conditions.
Applying our extensive experience, we have already brought breakthrough ophthalmic treatments to people living with vision loss. Susvimo™ (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant is the first U.S. Food and Drug Administration-approved refillable eye implant for neovascular or ‘wet’ age-related macular degeneration that continuously delivers a customised formulation of ranibizumab over a certain period of months.26 Vabysmo® (faricimab) is the first bispecific antibody approved for the eye, which targets two disease pathways that drive retinal conditions.8,10 Lucentis®* (ranibizumab injection) is the first treatment approved to improve vision in people with certain retinal conditions.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The Digital LightCycler System is a next-generation digital PCR system that helps clinical researchers better understand the nature of a patient’s cancer, genetic disease or infection.
The new system has the potential to find and quantify ultra-rare, hard to detect mutations, leading to early diagnosis and treatment strategies.
The Digital LightCycler System is designed for laboratories performing highly sensitive and precise DNA and RNA analysis in areas such as oncology and infectious diseases.
Basel, 23 August 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the launch of the Digital LightCyclerⓇ System, Roche’s first digital polymerase chain reaction ( PCR) system. This next-generation system detects disease and is designed to accurately quantify trace amounts of specific DNA and RNA targets not typically detectable by conventional PCR methods.
The Digital LightCycler System will allow clinical researchers to divide DNA and RNA from an already extracted clinical sample into as many as 100,000 microscopic individual reactions. The system can then perform PCR and produce highly sophisticated data analysis on the results.
"Understanding the hidden characteristics of serious diseases is fundamental for ensuring that the most effective treatment is selected for each patient," said Thomas Schinecker, CEO of Roche Diagnostics. “The Digital LightCycler System will support clinical researchers and laboratories in identifying rare and emerging disease mutations. This can be instrumental in early diagnosis and therapy decisions."
For areas like oncology and infectious disease, the system provides new opportunities for researchers beyond what traditional PCR technology can achieve. This includes ultra rare targets, accurately quantifying how much of the target DNA or RNA there is at a molecular level, and other high precision analysis.
Roche has made significant contributions to establish and advance PCR technology over several decades. In 1991, Roche saw the potential in PCR and invested in refining the science for use in molecular diagnostics to detect pathogens and genetic code changes that lead to diseases.
The unique capabilities of the Digital LightCycler System complement the Roche suite of widely available PCR solutions. The launch of this new system adds to this PCR ecosystem, and builds on the company’s founding legacy in PCR. Both solutions will be offered by Roche, providing a suite of molecular solutions to the healthcare community.
The system will be available in 15 countries worldwide in 2022 with plans to launch into more countries in the near future. It will be CE-marked and has an FDA 510(k) exempt status.
The Digital LightCycler System is the first digital PCR (dPCR) system from Roche and is intended for in-vitro diagnostic (IVD) testing. It performs absolute quantification of DNA and RNA targets using PCR technology. The Digital LightCycler System provides flexibility for laboratories through three unique reaction plates allowing customisation of the sample volumes used, the number of times a sample is partitioned (divided), and how many different tests can be run on a single sample (known as multiplexing). The system also features an easy workflow, moving from sample to results efficiently.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
First new treatment regimen in more than 20 years to significantly improve outcomes in people with this fast-growing type of lymphoma
Application is based on pivotal data from the phase III POLARIX study showing Polivy plus R-CHP significantly reduced the risk of disease progression, relapse or death with comparable safety versus the standard of care, R-CHOP
Various combination studies with Polivy and the company's CD20xCD3 bispecifics in diffuse large B-cell lymphoma are ongoing
Basel, 16 August 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the U.S. Food and Drug Administration (FDA) has accepted the company’s supplemental Biologics License Application (sBLA) for Polivy® (polatuzumab vedotin-piiq) in combination with Rituxan® (rituximab) plus cyclophosphamide, doxorubicin and prednisone (R-CHP) for the treatment of people with previously untreated diffuse large B-cell lymphoma (DLBCL). The FDA is expected to make a decision on approval by 2 April 2023.
“The POLARIX study results suggest that Polivy plus R-CHP could transform the treatment of this aggressive malignancy, and we are working with the FDA to bring this combination to newly diagnosed DLBCL patients as soon as possible,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We hope it will become the new standard of care for the first-line treatment of DLBCL, potentially reducing the need for subsequent treatments and limiting patient burden.”
DLBCL is an aggressive blood cancer. Although DLBCL often responds to initial treatment, it is not cured with the current standard of care in four out of 10 people. Most relapses occur within two years of starting treatment and the majority of those who require subsequent lines of therapy have poor outcomes.
The sBLA is based on results from the pivotal phase III POLARIX trial, which is the first in two decades to show a clinically meaningful improvement in progression-free survival (PFS) compared to the current standard of care Rituxan plus cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP). The risk of disease progression, relapse or death was reduced by 27% with Polivy plus R-CHP compared with R-CHOP after a median follow-up of 28.2 months (hazard ratio [HR] 0.73; 95% confidence interval [CI]: 0.57–0.95; p<0.02). Safety outcomes were consistent with those seen in previous clinical trials, and the safety profile was comparable for Polivy plus R-CHP versus R-CHOP, including rates of Grade 3-4 adverse events (AEs; 57.7% versus 57.5%), serious AEs (34.0% versus 30.6%), Grade 5 AEs (3.0% versus 2.3%), and AEs leading to dose reduction (9.2% versus 13.0%).
Based on pivotal data from the POLARIX study, the European Commission approved Polivy in combination with R-CHP in May 2022 for the treatment of adult patients with previously untreated DLBCL. Polivy is currently approved as a readily available, fixed-duration treatment option for relapsed or refractory (R/R) DLBCL in combination with bendamustine and Mabthera/Rituxan in more than 70 countries worldwide, including in the EU and in the United States.
Roche continues to explore areas of unmet need where Polivy has the potential to deliver additional benefit, including in ongoing studies investigating combinations of Polivy with the company’s CD20xCD3 T-cell engaging bispecific antibodies Lunsumio® (mosunetuzumab) and glofitamab, and with Rituxan in combination with gemcitabine and oxaliplatin in the phase III POLARGO study.
POLARIX [NCT03274492] is an international phase III, randomised, double-blind, placebo- controlled study evaluating the efficacy, safety and pharmacokinetics of Polivy® (polatuzumab vedotin-piiq) plus Rituxan® (rituximab), cyclophosphamide, doxorubicin, and prednisone (R-CHP) versus rituximab, cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP) in people with previously untreated diffuse large B-cell lymphoma. Eight-hundred and seventy-nine patients were randomised 1:1 to receive either Polivy plus R- CHP plus a vincristine placebo for six cycles, followed by rituximab for two cycles; or R-CHOP plus a Polivy placebo for six cycles, followed by two cycles of rituximab. The primary outcome measure is progression-free survival (PFS) as assessed by the investigator using the Lugano Response Criteria for malignant lymphoma. POLARIX is being conducted in collaboration with The Lymphoma Study Association (LYSA) and The Lymphoma Academic Research Organisation (LYSARC). Other clinical investigators from around the world also participated in the trial.
DLBCL is the most common form of non-Hodgkin lymphoma (NHL), accounting for about one in three cases of NHL.[1] DLBCL is an aggressive (fast-growing) type of NHL. [1] While it is generally responsive to treatment in the frontline, as many as 40% of people will relapse or have refractory disease, at which time salvage therapy options are limited and survival is short.[2,3] Approximately 150,000 people worldwide are estimated to be diagnosed with DLBCL each year.
Polivy is a first-in-class anti-CD79b antibody-drug conjugate (ADC). The CD79b protein is expressed specifically in the majority of B-cells, an immune cell impacted in some types of non-Hodgkin lymphoma (NHL), making it a promising target for the development of new therapies. Polivy binds to cancer cells such as CD79b and destroys these B-cells through the delivery of an anti-cancer agent, which is thought to minimise the effects on normal cells. Polivy is being developed by Roche using Seagen ADC technology and is currently being investigated for the treatment of several types of NHL. Polivy is currently marketed in the EU for the treatment of relapsed or refractory diffuse large B-cell lymphoma.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of hematological diseases. Our approved medicines include MabThera/Rituxan® (rituximab), Gazyvaro/Gazyva® (obinutuzumab), Polivy® (polatuzumab vedotin-piiq), Venclexta/Venclyxto® (venetoclax) in collaboration with AbbVie, and Hemlibra® (emicizumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies, glofitamab and Lunsumio®( mosunetuzumab), targeting both CD20 and CD3, and cevostamab, targeting both FcRH5 and CD3; Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1 and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
IMscin001 study showed non-inferior levels of cancer immunotherapy Tecentriq in the blood, when injected subcutaneously, compared to intravenous infusion, in people with advanced non-small cell lung cancer
Administered under the skin, the subcutaneous formulation reduces time spent receiving treatment to just minutes, compared with up to an hour for IV infusion
Data will be submitted to health authorities globally, including the US Food and Drug Administration and European Medicines Agency
Basel, 2 August 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the Phase III IMscin001 study evaluating a subcutaneous formulation of Tecentriq® (atezolizumab) met its co-primary endpoints. The study showed non-inferior levels of Tecentriq in the blood (pharmacokinetics), when injected subcutaneously, compared with intravenous (IV) infusion, in cancer immunotherapy-naïve patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) for whom prior platinum therapy has failed. The safety profile of the subcutaneous formulation was consistent with that of IV Tecentriq.
Administering Tecentriq subcutaneously (injecting the medicine under the skin) reduces the treatment time to 3-8 minutes per injection, compared with 30-60 minutes for standard IV infusion.
“By reducing the administration time, this new Tecentriq formulation could help save time for patients and healthcare systems,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We are excited by the potential of bringing a subcutaneous cancer immunotherapy to patients globally, delivering on our commitment to improve the treatment experience for patients.”
Multiple oncology studies suggest that the majority of cancer patients generally prefer to receive treatment subcutaneously due to reduced pain and discomfort, ease of administration and shorter duration of treatment, compared to IV infusion.
Roche will share detailed findings of the IMscin001 study at an upcoming medical meeting and submit them for regulatory approval to health authorities globally, including the US Food and Drug Administration (FDA) and European Medicines Agency (EMA).
IMscin001 is a Phase Ib/III, global, multicentre, randomised study evaluating the pharmacokinetics, safety and efficacy of the subcutaneous formulation of Tecentriq, compared with IV Tecentriq, in patients with previously treated locally advanced or metastatic NSCLC for whom prior platinum therapy has failed. The study enrolled 371 patients. The co-primary endpoints of the study are minimum levels of Tecentriq in the blood during a given dosing interval on the basis of established pharmacokinetic measurements; observed serum Ctrough and model-predicted area under the curve (AUC). Secondary endpoints include safety, immunogenicity, patient-reported outcomes and efficacy.
The investigational subcutaneous formulation combines Tecentriq with Halozyme Therapeutics’ Enhanze® drug delivery technology.
Tecentriq is a monoclonal antibody designed to bind with a protein called programmed death ligand-1 (PD-L1), which is expressed on tumour cells and tumour-infiltrating immune cells, blocking its interactions with both PD-1 and B7.1 receptors. By inhibiting PD-L1, Tecentriq may enable the activation of T-cells. Tecentriq is a cancer immunotherapy that has the potential to be used as a foundational combination partner with other immunotherapies, targeted medicines and various chemotherapies across a broad range of cancers.
The Enhanze drug delivery technology is based on a proprietary recombinant human hyaluronidase PH20 (rHuPH20), an enzyme that locally and temporarily degrades hyaluronan – a glycosaminoglycan or chain of natural sugars in the body – in the subcutaneous space. This increases the permeability of the tissue under the skin, allowing space for Tecentriq to enter, and enables the subcutaneous formulation to be rapidly dispersed and absorbed into the bloodstream.
IV Tecentriq is approved for some of the most aggressive and difficult-to-treat forms of cancer. IV Tecentriq was the first cancer immunotherapy approved for the treatment of a certain type of early-stage NSCLC, small cell lung cancer (SCLC) and hepatocellular carcinoma (HCC). IV Tecentriq is also approved in countries around the world, either alone or in combination with targeted therapies and/or chemotherapies, for various forms of metastatic NSCLC, certain types of metastatic urothelial cancer, PD-L1-positive metastatic triple-negative breast cancer and BRAF V600 mutation-positive advanced melanoma.
Roche’s rigorous pursuit of groundbreaking science has contributed to major therapeutic and diagnostic advances in oncology over the last 50 years, and today, realising the full potential of cancer immunotherapy is a major area of focus. With over 20 molecules in development, Roche is investigating the potential benefits of immunotherapy alone, and in combination with chemotherapy, targeted therapies or other immunotherapies with the goal of providing each person with a treatment tailored to harness their own unique immune system to attack their cancer. Our scientific expertise, coupled with an innovative pipeline and extensive partnerships, gives us the confidence to continue pursuing the vision of finding a cure for cancer by ensuring the right treatment for the right patient at the right time.
In addition to Roche’s approved PD-L1 checkpoint inhibitor, Tecentriq® (atezolizumab), Roche’s broad cancer immunotherapy pipeline includes other checkpoint inhibitors, such as tiragolumab, a novel cancer immunotherapy designed to bind to TIGIT, individualised neoantigen therapies and T-cell bispecific antibodies.
To learn more about Roche’s scientific-led approach to cancer immunotherapy, please follow this link: http://www.roche.com/research_and_development/what_we_are_working_on/oncology/cancer-immunotherapy.htm
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
