The cobas SARS-CoV-2 Duo test combines the standard qualitative result of a traditional SARS-CoV-2 PCR test with a quantitative result, which measures the viral load of a patient suspected of COVID-19.
Potential benefits of the cobas SARS-CoV-2 Duo test aim to help the healthcare community with contact tracing, patient triage and the approach to medical care.
Since the start of pandemic, Roche has focused on bringing effective diagnostic solutions to healthcare communities worldwide in the fight against COVID-19
Basel, 15 June 2022 – Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that the US Food and Drug Administration (FDA) issued Emergency Use Authorization (EUA) for the cobas SARS-CoV-2 Duo for use on the fully automated cobas 6800/8800 Systems, expanding the Roche COVID-19 portfolio. This is the first automated, real-time RT-PCR assay for the in vitro qualitative and quantitative detection of SARS-CoV-2 RNA in nasal and nasopharyngeal swab specimens. "With the SARS-CoV-2 Duo test, we are now able to detect the COVID-19 virus and simultaneously measure the viral load in an individual,” said Thomas Schinecker, CEO Roche Diagnostics. “The test’s performance suggests that, by earlier and more accurately identifying infected patients, the results may open the path for healthcare providers to more efficiently organise their therapeutic and monitoring interventions.”
The quantitative result is traceable to the World Health Organization (WHO) International Standard for SARS-CoV-2 RNA. The potential benefits from reporting a standardized viral load along with the qualitative result may help clinicians in the assessment and monitoring of infected patients across laboratories and over time.
Roche continues to add a range of diagnostic solutions to our global portfolio to help in the fight against COVID-19. For more information on how Roche is responding to the global COVID-19 pandemic, please visit our COVID-19 response page.
The test will be available in the United States by Q2 2022.
The cobas SARS-CoV-2 Duo test received Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA) on June 14, 2022. The cobas SARS-CoV-2 Duo for use on the cobas® 6800/8800 Systems is an automated real-time RT-PCR assay for the qualitative detection of SARS-CoV-2 RNA in healthcare provider instructed self-collected nasal swab specimens (collected on site), and healthcare provider-collected nasal and nasopharyngeal swab specimens collected from individuals suspected of COVID-19 by their healthcare provider. This assay also performs quantitation of SARS-CoV-2 RNA levels in the collected specimen; however, only the qualitative result of cobas® SARS-CoV-2 Duo is intended for use as an aid in the diagnosis of SARS-CoV-2 infection in patients suspected of COVID-19 by their healthcare provider.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Long-term data at the European Hematology Association (EHA) 2022 congress expands understanding of the impact of Roche medicines in early-stage blood cancers with the goal of providing patients with robust and durable outcomes from their first treatment
Updated data from phase III CLL14 study of Venclexta®/Venclyxto® (venetoclax) plus Gazyva®/Gazyvaro® (obinutuzumab) showed more than 60% of previously untreated people with chronic lymphocytic leukaemia remained in remission five years after starting treatment
Final analysis of phase III GALLIUM study showed meaningful improvement in progression-free survival was maintained with Gazyva/Gazyvaro plus chemotherapy in people with previously untreated follicular lymphoma after eight years of follow-up
Basel, 10 June 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that it is presenting new long-term follow-up results and subanalyses from clinical trials of its approved therapies, as well as data on investigational medicines from its broad blood cancer portfolio, at the European Hematology Association (EHA) 2022 Congress in Vienna. Data include five-year results from the phase III CLL14 study of fixed-duration Venclexta®/Venclyxto® (venetoclax) plus Gazyva®/Gazyvaro® (obinutuzumab) in previously untreated chronic lymphocytic leukaemia (CLL); the final analysis of the phase III GALLIUM study of Gazyva/Gazyvaro plus chemotherapy in people with previously untreated advanced-stage follicular lymphoma (FL); and subanalyses from the phase III POLARIX study of Polivy® (polatuzumab vedotin) in combination with MabThera®/Rituxan® (rituximab) plus cyclophosphamide, doxorubicin and prednisone (R-CHP) in people with previously untreated diffuse large B-cell lymphoma (DLBCL). Roche will also present data from its T-cell engaging bispecific antibody development programmes including Lunsumio® (mosunetuzumab) and glofitamab in patients receiving later lines of therapy for non-Hodgkin lymphoma (NHL) and investigational medicines cevostamab and RG6234 in relapsed or refractory (R/R) multiple myeloma (MM).
“Blood cancers remain challenging to treat at all stages, but by improving frontline treatment options we aim to increase the likelihood of meaningful clinical outcomes for these patients,” said Levi Garraway, M.D., Ph.D., Chief Medical Officer and Head of Global Product Development. “With these new long-term data and other studies of fixed-duration therapies in our portfolio, we are working to lessen the treatment burdens associated with long-term cancer care.”
After a median of 65.4 months following treatment with Venclexta/Venclyxto plus Gazyva/Gazyvaro, results confirm the combination continues to be an effective fixed-duration and chemotherapy-free option for patients with previously untreated CLL and coexisting conditions. The estimated investigator-assessed progression-free survival (PFS) rate at this follow-up was 62.6% with Venclexta/Venclyxto plus Gazyva/Gazyvaro and 27.0% with Gazyva/Gazyvaro plus chlorambucil, and the estimated overall survival (OS) rate was 81.9% versus 77.0% (HR 0.72; 95% CI: 0.48-1.09; p=0.12). In addition, the analysis found that 72.1% of patients in the Venclexta/Venclyxto plus Gazyva/Gazyvaro arm did not require another treatment for CLL in the five years following initial treatment (HR 0.42; 95% CI: 0.31-0.57; p<0.0001). No new safety signals were observed.[1] The CLL14 study is being conducted in cooperation with the German CLL Study Group, headed by Michael Hallek, M.D., University of Cologne.
After eight years of follow-up in people with previously untreated FL, a meaningful improvement in PFS was maintained with Gazyva/Gazyvaro plus chemotherapy, confirming its role as a standard of care for first-line treatment. Seven-year investigator-assessed PFS was significantly improved with Gazyva/Gazyvaro plus chemotherapy (63.4%) compared with MabThera/Rituxan plus chemotherapy (55.7%; HR 0.77; 95% CI: 0.64-0.93; p=0.006). This translated into a longer time to next anti-lymphoma treatment. At seven years, 74.1% of patients receiving Gazyva/Gazyvaro plus chemotherapy had not started new anti-lymphoma therapy compared to 65.4% receiving MabThera/Rituxan plus chemotherapy (HR 0.71; 95% CI: 0.58–0.87; p=0.001). The incidence of serious adverse events was 48.9% with Gazyva/Gazyvaro plus chemotherapy and 43.4% with MabThera/Rituxan plus chemotherapy.
Exploratory subgroup analyses of the phase III POLARIX study of Polivy with R-CHP compared to the current standard of care, MabThera/Rituxan plus cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP), in people with previously untreated DLBCL further support the potential for Polivy to transform the standard of care for people with this aggressive type of lymphoma. One of the datasets being presented is an analysis of study participants from Asia (China, Hong Kong, Japan, South Korea and Taiwan). Among this subgroup, results showed a significant improvement in PFS with Polivy plus R-CHP versus R-CHOP, reducing the risk of disease progression, relapse or death by 36% (HR 0.64; 95% CI: 0.40-1.03). The safety profile was generally comparable for both regimens.
Based on the positive POLARIX results from the overall study population, the European Commission (EC) approved Polivy plus R-CHP in May 2022 for the treatment of adult patients with previously untreated DLBCL.
The pivotal phase II NP30179 expansion study included patients with heavily pre-treated and highly refractory DLBCL and showed fixed-duration glofitamab, an investigational CD20xCD3 T-cell engaging bispecific antibody, induced high and durable complete response (CR) rates. After a median follow-up of 12.6 months, 39.4% of patients (n=61/155) achieved a CR (primary efficacy endpoint) and half of them (51.6%; n=80/155) achieved an overall response (the percentage of patients with a partial or CR; secondary efficacy endpoint), as assessed by an independent review committee. Cytokine release syndrome (CRS) was the most common adverse event, occurring in 63.0% of patients.[4] These data were recently presented at the American Society of Clinical Oncology 2022 Annual Meeting and have been submitted for approval to the European Medicines Agency (EMA). Submissions to additional health authorities worldwide, including the U.S. Food and Drug Administration (FDA), are planned this year.
An exploratory subgroup analysis showed Lunsumio could be an efficacious and tolerable option in patients aged <65 and ≥65 years who had R/R FL and had received two or more prior therapies. Patients ≥65 years old achieved a higher objective response rate (ORR) than those <65 years old (87.0% vs 77.0%, respectively). Lower rates of CRS and serious adverse events were observed in patients ≥65 years old (37%) compared to those <65 years old (52%).[5] Additional data from the GO29781 study showed that retreatment with Lunsumio in patients who achieved a CR but whose disease subsequently progressed was effective and the safety of retreatment was consistent with initial treatment.[6]
The EC recently approved Lunsumio for the treatment of people with R/R FL who have received at least two prior systemic therapies.
The data being presented at EHA, as well as phase III studies currently underway, will expand the understanding of glofitamab and Lunsumio and their impact in both later and earlier lines of treatment, with the aim of providing robust and durable treatment outcomes for people with different types of lymphomas.
In line with Roche’s commitment to improving outcomes and personalising care for people with blood cancer, the company has expanded beyond lymphoma and leukaemia, evaluating two investigational medicines in MM. This is the third most common type of blood cancer, diagnosed in more than 170,000 people around the world each year and involves plasma cells (antibody-producing cells in the bone marrow).[7,8] Although advances in treatment have improved outcomes, MM remains an incurable disease characterised by multiple relapses, with an overall five-year survival rate of about 55%.[9] Roche is presenting data at EHA on cevostamab, an investigational FcRH5xCD3 T-cell engaging bispecific antibody that is being evaluated as a monotherapy and in combination with other medicines to treat people with R/R MM, and on RG6234, a novel GPRC5DxCD3 T-cell engaging bispecific antibody that is being studied in a phase I trial in people with R/R MM. While early, the clinical activity and safety profiles observed with these molecules look encouraging and support further exploration.
Venclexta/Venclyxto is a first-in-class targeted medicine designed to selectively bind and inhibit the B-cell lymphoma-2 (BCL-2) protein. In some blood cancers and other tumours, BCL-2 builds up and prevents cancer cells from dying or self-destructing, a process called apoptosis. Venclexta/Venclyxto blocks the BCL-2 protein and works to help restore the process of apoptosis.
Venclexta/Venclyxto is being developed by AbbVie and Roche. It is jointly commercialised by AbbVie and Genentech, a member of the Roche Group, in the US and commercialised by AbbVie outside of the US. Together, the companies are committed to research with Venclexta/Venclyxto, which is currently being studied in clinical trials across several types of blood cancers.
In the US, Venclexta has been granted six Breakthrough Therapy Designations by the U.S. Food and Drug Administration: one for previously untreated chronic lymphocytic leukaemia (CLL), two for relapsed or refractory CLL, two for previously untreated acute myeloid leukaemia, and one for myelodysplastic syndromes.
Gazyva/Gazyvaro is an engineered monoclonal antibody designed to attach to CD20, a protein found only on certain types of B-cells. It is thought to work by attacking targeted cells both directly and together with the body's immune system. Gazyva/Gazyvaro is part of a collaboration between Roche and Biogen.
In the US, Europe and multiple other countries, Gazyva/Gazyvaro is currently approved in combination with chlorambucil for patients with previously untreated chronic lymphocytic leukaemia (CLL). It is also approved in combination with bendamustine, followed by Gazyva/Gazyvaro maintenance for the treatment of follicular lymphoma (FL) patients who did not respond to a MabThera®/Rituxan® (rituximab)-containing regimen, or whose FL returned after such treatment and in combination with chemotherapy for previously untreated advanced FL.
Additional combination studies investigating Gazyva/Gazyvaro with other approved or investigational medicines, including cancer immunotherapies and small molecule inhibitors, are underway across a range of blood cancers.
Polivy is a first-in-class anti-CD79b antibody-drug conjugate (ADC). The CD79b protein is expressed specifically in the majority of B-cells, an immune cell impacted in some types of non-Hodgkin lymphoma (NHL), making it a promising target for the development of new therapies.[12,13] Polivy is designed to bind to CD79b and destroys these B-cells through the delivery of an anti-cancer agent, which is thought to minimise the effects on normal cells.[14,15] Polivy is being developed by Roche using Seagen ADC technology and is currently being investigated for the treatment of several types of NHL.
Roche is currently developing two CD20xCD3 T-cell engaging bispecific antibodies, Lunsumio® (mosunetuzumab) and glofitamab, designed to target CD20 on the surface of B-cells and CD3 on the surface of T-cells. This dual targeting activates and redirects a patient’s existing T-cells to engage and eliminate target B-cells by releasing cytotoxic proteins into the B-cells. Lunsumio and glofitamab differ in their structures, and both are being developed by Roche as part of our ongoing strategy to explore multiple bispecific formats in order to identify those that maximise potential clinical benefits for patients. Lunsumio has a structure similar to that of a natural human antibody in that it has two ‘Fab’ regions but is different from naturally-occurring antibodies in that one ‘Fab’ region targets CD20 and the other ‘Fab’ region targets CD3. Glofitamab is based on a novel structural format that we call ‘2:1,’ which refers to the structure of the antibody. It is engineered to have two ‘Fab’ regions that bind to CD20 and one ‘Fab’ region that binds to CD3. The clinical development programmes for Lunsumio and glofitamab include ongoing investigations of these molecules as monotherapies and in combination with other medicines for the treatment of people with CD20-positive B cell non-Hodgkin lymphomas, including diffuse large B-cell lymphoma and follicular lymphoma.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
In the Phase III IMpower010 trial, adjuvant Tecentriq reduced the risk of disease recurrence or death by 57% in people with PD-L1 high resected Stage II-III NSCLC, compared with best supportive care
Tecentriq is now the first and only cancer immunotherapy available for certain people with early-stage NSCLC in Europe
Today’s approval marks Tecentriq’s sixth lung cancer indication in Europe
Basel, 09 June 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the European Commission has approved Tecentriq® (atezolizumab) as an adjuvant treatment, following complete resection and platinum-based chemotherapy, for adults with non-small cell lung cancer (NSCLC) with a high risk of recurrence* whose tumours express PD-L1≥50% and who do not have EGFR mutant or ALK-positive NSCLC.
“Today’s approval represents an important advance, as Tecentriq becomes the first cancer immunotherapy approved in Europe for the treatment of certain types of early-stage NSCLC,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Since approximately half of all people with early NSCLC develop recurrence after surgery, which in some cases is no longer curable, treating this cancer at an earlier stage offers the best chance to prevent recurrence.”
This approval is based on results from an interim analysis of the Phase III IMpower010 study. The results showed treatment with Tecentriq, following complete resection and platinum-based chemotherapy, reduced the risk of disease recurrence or death (DFS) by 57% (hazard ratio [HR]=0.43, 95% CI: 0.26-0.71)** in people with resected Stage II-IIIA NSCLC (UICC/AJCC 7th edition) whose tumours express PD-L1≥50%, who do not have EGFR mutant or ALK-positive NSCLC, compared with best supportive care (BSC).1 A DFS benefit was consistently seen across most subgroups including histology or stage of disease with adjuvant Tecentriq, compared with BSC. Overall survival (OS) data for patients with PD-L1 high resected Stage II-III NSCLC, and who do not have EGFR mutant or ALK-positive disease are immature and were not formally tested at the DFS interim analysis, however, a trend towards OS improvement with Tecentriq was seen, with a stratified HR of 0.39 (95% CI: 0.18-0.82).2
Follow-up will continue with planned analyses of more mature OS data later this year. Safety data for Tecentriq were consistent with its known safety profile and no new safety signals were identified.1
“Today’s approval now offers patients in Europe, whose tumours express high levels of PD-L1, the opportunity to reduce their risk of disease recurrence following surgery and chemotherapy,” said Professor Enriqueta Felip, Head of the Thoracic Cancer Unit at Vall d’Hebron Institute of Oncology, Barcelona, Spain. “This milestone reinforces the need for biomarker testing at diagnosis for all people with NSCLC, irrespective of disease stage, to ensure they receive optimal treatment.”
To date, Tecentriq has been approved in 19 countries, including the US and China, as adjuvant treatment, following complete resection and chemotherapy, for adults with Stage II-IIIA NSCLC (UICC/AJCC 7th edition) whose tumours express PD-L1≥1%. In three countries, including Canada and the UK, Tecentriq has been approved as adjuvant, treatment following complete resection and chemotherapy, for adult patients with Stage II-IIIA NSCLC (UICC/AJCC 7th edition) whose tumours have PD-L1 expression on ≥50% of tumour cells.
Tecentriq has shown clinically meaningful benefit in various types of lung cancer, with six currently approved indications in countries around the world. It was the first approved cancer immunotherapy for the first-line treatment of adults with extensive-stage small cell lung cancer (SCLC) in combination with carboplatin and etoposide (chemotherapy). Tecentriq also has four approved indications in advanced or metastatic NSCLC as either a single agent or in combination with targeted therapies and/or chemotherapies. Tecentriq is available in three dosing options, providing the flexibility to choose administration every two, three or four weeks.
Roche has an extensive development programme for Tecentriq including multiple ongoing and planned Phase III studies across lung, genitourinary, skin, breast, gastrointestinal, gynaecological, and head and neck cancers. This includes studies evaluating Tecentriq both alone and in combination with other medicines, as well as studies in metastatic, adjuvant and neoadjuvant settings across various tumour types.
IMpower010 is a Phase III, global, multicentre, open-label, randomised study evaluating the efficacy and safety of Tecentriq compared with BSC, in participants with Stage IB-IIIA NSCLC (UICC/AJCC 7th edition), following surgical resection and up to 4 cycles of adjuvant cisplatin-based chemotherapy. The study randomised 1,005 people with a ratio of 1:1 to receive either Tecentriq (up to 16 cycles) or BSC. The primary endpoint is investigator-determined DFS in the PD-L1-positive Stage II-IIIA, all randomised Stage II-IIIA and intention-to-treat (ITT) Stage IB-IIIA populations. Key secondary endpoints include overall survival in the overall study population, ITT Stage IB-IIIA NSCLC.
Lung cancer is one of the leading causes of cancer death globally.3 Each year 1.8 million people die as a result of the disease; this translates into more than 4,900 deaths worldwide every day.3 Lung cancer can be broadly divided into two major types: NSCLC and SCLC. NSCLC is the most prevalent type, accounting for around 85% of all cases.4 Approximately 50% of patients with NSCLC are diagnosed with early-stage (Stages I and II) or locally advanced (Stage III) disease.4 Today, about half of all people with early lung cancer still experience a cancer recurrence following surgery.5 Treating lung cancer early, before it has spread, may help prevent the disease from returning and provide people with the best opportunity for a cure.
Tecentriq is a cancer immunotherapy approved for some of the most aggressive and difficult-to-treat forms of cancer. Tecentriq was the first cancer immunotherapy approved for the treatment of a certain type of early-stage non-small cell lung cancer (NSCLC), small cell lung cancer (SCLC) and hepatocellular carcinoma (HCC). Tecentriq is also approved in countries around the world, either alone or in combination with targeted therapies and/or chemotherapies, for various forms of metastatic NSCLC, certain types of metastatic urothelial cancer, PD-L1-positive metastatic triple-negative breast cancer and BRAF V600 mutation-positive advanced melanoma.
Tecentriq is a monoclonal antibody designed to bind with a protein called programmed death ligand-1 (PD-L1), which is expressed on tumour cells and tumour-infiltrating immune cells, blocking its interactions with both PD-1 and B7.1 receptors. By inhibiting PD-L1, Tecentriq may enable the activation of T-cells. Tecentriq is a cancer immunotherapy that has the potential to be used as a foundational combination partner with other immunotherapies, targeted medicines and various chemotherapies across a broad range of cancers. In addition to intravenous infusion, the formulation of Tecentriq is also being investigated as a subcutaneous injection to help address the growing burden of cancer treatment for patients and healthcare systems.
Lung cancer is a major area of focus and investment for Roche, and we are committed to developing new approaches, medicines and tests that can help people with this deadly disease. Our goal is to provide an effective treatment option for every person diagnosed with lung cancer. We currently have six approved medicines to treat certain kinds of lung cancer and more than ten medicines being developed to target the most common genetic drivers of lung cancer or to boost the immune system to combat the disease.
Roche’s rigorous pursuit of groundbreaking science has contributed to major therapeutic and diagnostic advances in oncology over the last 50 years, and today, realising the full potential of cancer immunotherapy is a major area of focus. With over 20 molecules in development, Roche is investigating the potential benefits of immunotherapy alone, and in combination with chemotherapy, targeted therapies or other immunotherapies with the goal of providing each person with a treatment tailored to harness their own unique immune system to attack their cancer. Our scientific expertise, coupled with an innovative pipeline and extensive partnerships, gives us the confidence to continue pursuing the vision of finding a cure for cancer by ensuring the right treatment for the right patient at the right time.
In addition to Roche’s approved PD-L1 checkpoint inhibitor, Tecentriq® (atezolizumab), Roche’s broad cancer immunotherapy pipeline includes other checkpoint inhibitors, such as tiragolumab, a novel cancer immunotherapy designed to bind to TIGIT, individualised neoantigen therapies and T-cell bispecific antibodies.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
FoundationOne CDx can now be used to identify patients with ROS1 fusion-positive non-small cell lung cancer or patients with NTRK fusion-positive solid tumours for whom treatment with Rozlytrek may be appropriate
This approval marks the first and only companion diagnostic indication for Rozlytrek, and another important milestone in tumour-agnostic approaches for people living with rare cancers
Roche is a leader in driving personalised healthcare around the world through validated diagnostic tools, genomic insights and a continued focus on drug development
Basel, 9 June 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the United States Food and Drug Administration (U.S. FDA) has approved Foundation Medicine’s FoundationOne®CDx as a companion diagnostic (CDx) for Roche’s Rozlytrek® (entrectinib). FoundationOne CDx is a comprehensive genomic profiling (CGP) pan-tumour tissue biopsy test that assesses an individual’s cancer to identify the unique molecular ‘fingerprint’ of the tumour. It is the first and only U.S. FDA-approved CDx to identify patients with ROS1 fusion-positive non-small cell lung cancer (NSCLC), or patients with NTRK fusion-positive cancers, for whom treatment with Rozlytrek may be appropriate.
“The ability to tailor cancer therapies based on specific genomic alterations using validated comprehensive genomic profiling (CGP) has transformed the traditional ‘one-size fits-all’ approach to cancer,” said Levi Garraway, M.D., Ph.D., Chief Medical Officer and Head of Global Product Development. “This approval marks a significant step forward in expanding treatment options and improving outcomes for patients, particularly those with rare tumours.”
Using CGP to identify the genomic alterations that are associated with driving an individual’s cancer can support physicians in making an informed treatment decision for the individual patient, potentially achieving better clinical outcomes.1
Rozlytrek is a targeted therapy approved to treat ROS1 fusion-positive metastatic NSCLC and a tumour-agnostic medicine for locally advanced or solid tumours that harbour NTRK fusions. ROS1 gene fusions account for 1-2% of NSCLC, the most common type of lung cancer that accounts for up to 85% of all diagnoses.2,3 NTRK gene fusions have been identified in a range of solid tumour types, and are present in up to 90% of some rare cancer types and less than 1% of other more common cancers, including lung and colorectal.4
The approval is based on data from the phase I ALKA-372-001 (EudraCT 2012-000148-88), phase I STARTRK-1 (NCT02097810) and phase II STARKTRK-2 (NCT02568267) trials. As a condition of this approval, Foundation Medicine will conduct a post-approval study powered by the Flatiron Health-Foundation Medicine’s Clinico-Genomic Database (CGDB) to further demonstrate FoundationOne CDx’s ability to identify patients with ROS1 fusion-mutated NSCLC for whom treatment with Rozlytrek may be appropriate. The CGDB is a de-identified, HIPAA-compliant database that links outcomes data from Flatiron’s network of oncology clinics and genomic data from Foundation Medicine’s CGP assays. The database currently contains more than 100,000 linked genomic profiles , and is continually growing.
By combining tumour-agnostic drug development with data collection, high-quality diagnostics and the implementation of CGP, Roche is leading the realisation of personalised healthcare around the world, enabling physicians to develop tailored treatment strategies for each individual, and enable smarter, more efficient research and development to ensure no person is left behind.
FoundationOne CDx is a next-generation sequencing-based in vitro diagnostic assay for detection of substitutions, insertion and deletion alterations (indels), and copy number alterations in 324 genes and select gene rearrangements, as well as genomic signatures, including microsatellite instability and tumour mutational burden, using DNA isolated from formalin-fixed paraffin embedded tumour tissue specimens. FoundationOne CDx is for prescription-use only and is intended as a companion diagnostic (CDx) to identify people who may benefit from treatment with certain targeted therapies in accordance with their approved therapeutic product labeling. Additionally, FoundationOne CDx is intended to provide tumour mutation profiling to be used by qualified healthcare professionals in accordance with professional guidelines in oncology for patients with solid malignant neoplasms. Use of the test does not guarantee a patient will be matched to a treatment. A negative result does not rule out the presence of an alteration. Some patients may require a biopsy.
FoundationOne CDx was approved by the United States Food and Drug Administration in November 2017 and is currently approved as a CDx for 25 CDx indications, and three group claims across 30 targeted therapies. For a full list of targeted therapies for which FoundationOne CDx is indicated as a CDx, please visit http://www.foundationmedicine.com/genomic-testing/foundation-one-cdx.
Rozlytrek is a tumour-agnostic once-daily oral medicine for the treatment of locally advanced or metastatic solid tumours that harbour NTRK1/2/3 or ROS1 gene fusions. It is a selective tyrosine kinase inhibitor designed to inhibit the kinase activity of the TRKA/B/C and ROS1 proteins, whose activating fusions drive proliferation in certain types of cancer. Rozlytrek can block NTRK and ROS1 kinase activity and may result in the death of cancer cells with NTRK or ROS1 gene fusions.
Rozlytrek is approved for the treatment of NTRK fusion-positive solid tumours and ROS1 fusion-positive non-small cell lung cancer by health authorities in more than 60 countries, including by the United States Food and Drug Administration (U.S. FDA), Japan’s Ministry of Health, Labour and Welfare (MHLW) and the European Medicines Agency (EMA). Prior to approval, Rozlytrek was granted Breakthrough Therapy Designation by the U.S. FDA, Sakigake designation for accelerated review by Japan’s MHLW, and Priority Medicines designation by the EMA.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Lunsumio® (mosunetuzumab) is the first CD20xCD3 T-cell engaging bispecific antibody available to treat the most common slow-growing form of non-Hodgkin lymphoma, follicular lymphoma (FL)
Lunsumio represents a new type of immunotherapy that is a chemotherapy-free, off-the-shelf, fixed-duration treatment option that could improve outcomes for people who have relapsed or are refractory to multiple previous treatments
Approval is based on the phase I/II GO29781 study, where Lunsumio induced high complete response rates, with the majority of complete responses lasting for at least 18 months in people with heavily pre-treated FL
Basel, 8 June 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the European Commission has granted conditional marketing authorisation for the CD20xCD3 T-cell engaging bispecific antibody Lunsumio® (mosunetuzumab), for the treatment of adult patients with relapsed or refractory (R/R) follicular lymphoma (FL) who have received at least two prior systemic therapies. Lunsumio is an off-the-shelf therapy that is readily available, so people do not have to wait to start treatment.
Each year, more than 28,000 people in Europe are diagnosed with FL, which accounts for approximately one in five non-Hodgkin lymphoma cases.1,2 Despite treatment advances, FL is considered an incurable disease and relapse is common, with outcomes worsening on each consecutive treatment.1
“We are delighted that Lunsumio is the first bispecific antibody approved in Europe for people with relapsed or refractory follicular lymphoma,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Lunsumio’s high response rates, off-the-shelf availability, and initial outpatient administration could transform how advanced follicular lymphoma is treated.”
“Having additional treatment options for people with follicular lymphoma, where multiple prior lines of therapy have failed, is critical to help them achieve better outcomes,” said Elizabeth Budde, M.D., Ph.D., Haematologic Oncologist and Associate Professor at City of Hope. “It is exciting to have a new class of immunotherapy like Lunsumio, offering a readily available, chemotherapy-free and fixed-duration treatment, with great potential to provide durable remissions without the need to stay on treatment continuously.”
The approval is based on positive results from the phase I/II GO29781 study where Lunsumio demonstrated high complete response rates, with the majority of complete responders maintaining responses for at least 18 months, and favourable tolerability in people with heavily pre-treated FL. After a median follow-up of 18.3 months, the median duration of response among responders was 22.8 months (95% CI: 9.7-not estimable), the complete response rate was 60% (n=54/90), the objective response rate was 80% (n=72/90). The most common adverse event was cytokine release syndrome (39%), which was generally low grade (grade 2: 14%), and resolved by the end of treatment. Other common (≥20%) AEs were neutropenia, pyrexia, hypophosphatemia and headache. The initial dose was administered without mandatory hospitalisation. Results were presented for the first time in December 2021 at the 63rd American Society of Hematology Annual Meeting & Exposition.3
Conditional approval is granted to a medicinal product that fulfils an unmet medical need where the benefit of immediate availability outweighs the risk of less comprehensive data than normally required.
A robust development programme for Lunsumio is ongoing including two phase III studies: CELESTIMO, investigating Lunsumio plus lenalidomide in second line plus (2L+) FL, and SUNMO, investigating Lunsumio plus Polivy® (polatuzumab vedotin) in 2L+ diffuse large B-cell lymphoma (DLBCL).
This is Roche’s second EU approval in lymphoma in 2022, following the approval of Polivy in combination with MabThera® (rituximab) plus cyclophosphamide, doxorubicin and prednisone (R-CHP) in previously untreated DLBCL.4 With a broad portfolio and pipeline, Roche is committed to providing treatment solutions for different stages of blood disorders, that are tailored to the disease, patient, physician, and healthcare system, as monotherapies or in combination with established and/or novel agents.
Lunsumio is a first-in-class CD20xCD3 T-cell engaging bispecific antibody designed to target CD20 on the surface of B-cells and CD3 on the surface of T-cells. This dual targeting activates and redirects a patient’s existing T-cells to engage and eliminate target B-cells by releasing cytotoxic proteins into the B-cells. A robust clinical development programme for Lunsumio is ongoing, investigating the molecule as a monotherapy and in combination with other medicines, for the treatment of people with B-cell non-Hodgkin lymphomas, including follicular lymphoma, diffuse large B-cell lymphoma, and other blood cancers.
The GO29781 study [NCT02500407] is a phase I/II, multicentre, open-label, dose-escalation and expansion study evaluating the safety, efficacy and pharmacokinetics of Lunsumio® (mosunetuzumab) in people with relapsed or refractory B-cell non-Hodgkin lymphoma. Outcome measures include complete response rate (best response) by independent review facility (primary endpoint), objective response rate, duration of response, progression-free survival, safety, and tolerability (secondary endpoints).
Follicular lymphoma (FL) is the most common indolent (slow-growing) form of non-Hodgkin lymphoma (NHL), accounting for about one in five cases of NHL.1 It is considered incurable and relapse is common. It is estimated that more than 100,000 people are diagnosed with FL each year worldwide, including over 28,000 people in Europe
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera® (rituximab), Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclyxto® (venetoclax) in collaboration with AbbVie, and Hemlibra® (emicizumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies, glofitamab and Lunsumio® (mosunetuzumab), targeting both CD20 and CD3, and cevostamab, targeting both FcRH5 and CD3; Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1 and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Data to be presented for the first time at ASCO and EHA 2022 show glofitamab induces high and durable complete response rates in people with heavily pre-treated diffuse large B-cell lymphoma
Glofitamab has the potential to offer a readily available, fixed-duration CD20xCD3 bispecific antibody treatment approach for people with aggressive lymphoma
Glofitamab is part of Roche’s industry-leading CD20xCD3 bispecific antibody development programme, which aims to address the diverse needs and preferences of people with blood cancers
Basel, 27 May 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that new pivotal data on its investigational CD20xCD3 T-cell engaging bispecific antibody, glofitamab, will be presented for the first time at the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting from 3-7 June and the European Hematology Association (EHA) 2022 Congress from 9-12 June. Data from the phase II NP30179 expansion study demonstrated that, after a median follow-up of more than 12 months, fixed-duration glofitamab (given for a fixed amount of time, and not taken until disease progression) induces durable complete responses (CRs) in patients with relapsed or refractory (R/R) diffuse large B-cell lymphoma (DLBCL) who had received a median of three prior therapies.1
“These data bring us one step closer towards our goal of finding solutions for people with heavily pre-treated diffuse large B-cell lymphoma, which often relapses and becomes more aggressive each time it returns,” said Levi Garraway, M.D., Ph.D., Chief Medical Officer and Head of Global Product Development. “The potential of glofitamab as a new fixed-duration, readily available treatment could be instrumental to improving outcomes for people with this difficult-to-treat cancer who otherwise have limited options.”
The pivotal phase II NP30179 expansion study included patients with heavily pre-treated and highly refractory DLBCL, with 58.3% of patients refractory to their initial therapy and about one-third (33.1%) having received prior CAR T-cell therapy.1 After a median follow-up of 12.6 months, 39.4% of patients (n=61/155) achieved a CR (primary efficacy endpoint) and half of them (51.6%; n=80/155) achieved an overall response (the percentage of patients with a partial or complete response; secondary efficacy endpoint), as assessed by an independent review committee. The majority (77.6%) of complete responses were durable and ongoing at 12 months and the median duration of complete response had not yet been reached (not evaluable [16.8 months, not evaluable]). Cytokine release syndrome (CRS) was the most common adverse event occurring in 63.0% of patients. CRS events were predictable, generally low grade (mainly Grade 1 [47.4%] or 2 [11.7%]), occurred at initial doses, and only one patient discontinued glofitamab due to CRS. Incidence of Grade 3+ CRS was low (3.9%), with no Grade 5 events.
“I’m encouraged by these data as they signify new hope for these patients who otherwise have limited effective treatment options and have faced disappointment from not responding to multiple rounds of treatments,” said Associate Professor Michael Dickinson, Haematologist and Lead of the Aggressive Lymphoma Disease Group within Clinical Haematology at Peter MacCallum Cancer Centre and Royal Melbourne Hospital, Australia. “These glofitamab data suggest that patients may be able to achieve durable responses with a set course of treatment that they don’t have to take continuously until disease progression.”
Data from the NP30179 study have been submitted for approval to the European Medicines Agency (EMA), and submissions to additional health authorities worldwide, including to the U.S. Food and Drug Administration (FDA), are planned this year. Glofitamab is being investigated in several clinical trials and explored in earlier lines of lymphoma treatment.
Roche is committed to improving standards of care to enhance the treatment experience and outcomes for people with blood cancers and the scientific data we are sharing at ASCO and EHA from our portfolio propels us further towards this goal. Roche is investigating its CD20xCD3 T-cell engaging bispecific antibodies glofitamab and mosunetuzumab further as subcutaneous formulations and in additional phase III studies that will expand the understanding of their impact in earlier lines of treatment, with the aim of providing people with different types of lymphomas with robust and durable treatment outcomes. Additionally, the European Commission (EC) recently granted approval of Polivy® (polatuzumab vedotin) in combination with MabThera® (rituximab) plus cyclophosphamide, doxorubicin, and prednisone (R-CHP) for the treatment of adult patients with previously untreated diffuse large B-cell lymphoma (DLBCL), and the EMA’s Committee for Medicinal Products for Human Use recommended mosunetuzumab for approval for patients with R/R follicular lymphoma, who have received at least two prior systemic therapies.
Follow Roche on Twitter via @Roche and keep up to date with ASCO 2022 news and updates by using the hashtag #ASCO22. For exclusive materials sharing insights into Roche’s vision and strategy, and providing context behind the data being presented, visit Roche’s Oncology Newsroom here.
Glofitamab is an investigational CD20xCD3 T-cell engaging bispecific antibody designed to target CD20 on the surface of B-cells and CD3 on the surface of T-cells. Glofitamab is based on a novel structural format called ‘2:1’. It is engineered to have two ‘Fab’ regions which bind to CD20, and one ‘Fab’ region which binds to CD3. This dual targeting activates and redirects a patient’s existing T-cells to engage and eliminate target B-cells by releasing cytotoxic proteins into the B-cells. A robust clinical development programme for glofitamab is ongoing, investigating the molecule as a monotherapy and in combination with other medicines, for the treatment of people with B-cell non-Hodgkin lymphomas, including diffuse large B-cell lymphoma and mantle cell lymphoma, and other blood cancers.
The NP30179 study [NCT03075696] is a phase I/II, multicentre, open-label, dose-escalation and expansion study evaluating the safety, efficacy and pharmacokinetics of glofitamab in people with relapsed or refractory diffuse large B-cell lymphoma. Outcome measures include complete response rate by independent review committee (primary endpoint), overall response rate, duration of response, progression-free survival, safety and tolerability (secondary endpoints).
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera®/Rituxan® (rituximab), Gazyva®/Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie, and Hemlibra® (emicizumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies, glofitamab and mosunetuzumab, targeting both CD20 and CD3, and cevostamab, targeting both FcRH5 and CD3; Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1 and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Roche and its subsidiary TIB Molbiol have developed three different LightMix® Modular Virus kits for the detection of the monkeypox virus
Multiple clusters of monkeypox virus have been reported in non-endemic countries, and the World Health Organization (WHO) expects more cases will be identified as surveillance expands
Use of these new research test kits assess the spread of the virus and can help monitor the potential impact of therapeutics, vaccines and public health interventions
Basel, 25 May 2022 – Roche (SIX: RO, ROG; OTCQX: RHHBY) and its subsidiary TIB Molbiol have developed three unique LightMix® Modular Virus kits in response to recent monkeypox virus concerns. Multiple clusters of the monkeypox virus have been reported within the past two weeks in several European countries and North America, which are regions where the virus is not normally found.
“Roche has very quickly developed a new suite of tests that detect the monkeypox virus and aid in following its epidemiologic spread,” said Thomas Schinecker, CEO Roche Diagnostics. “Diagnostic tools are crucial for responding to and ultimately controlling emerging public health challenges as they advance response measures such as tracing efforts and treatment strategies.”
The first LightMix Modular Virus kit detects orthopoxviruses, including all monkeypox viruses from both the West African and Central African forms of the virus (referred to as clades).The second kit is a specific test detecting monkeypox viruses only (West African and Central African clade). For researchers interested in obtaining both of these results, a third kit is available that simultaneously detects orthopoxviruses plus provides information on whether a monkeypox virus is present or not (West African and Central African clade).
The test kits are available for research use in the majority of countries worldwide.
The LightMix® Modular Orthopox /Monkeypox Virus Kits are assays that detect Orthopoxviruses, including the monkeypox virus, using a technology called quantitative PCR (qPCR). To do this, first sample using an established NA extraction method. The assay is then performed on either a LightCycler® 480 II Instrument or cobas z 480 Analyzer. The kit, reagents and instruments are all available to purchase from the majority of Roche Diagnostics affiliates.
Monkeypox was first detected in laboratory monkeys in 1958. The virus is, however, assumed to transmit from wild animals such as rodents to people — or from human to human. In an average year, a few thousand cases occur in Western and Central Africa. But cases outside Africa have been limited to just a handful that are associated with travel to Africa or with the importation of infected animals. Treatments and vaccines that were developed for smallpox are available and are thought to be useful to limit spreading of Monkeypox.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
First new treatment option in more than 20 years to show a clinically meaningful improvement in progression-free survival is approved for people with previously untreated diffuse large B-cell lymphoma (DLBCL)
Approval is based on pivotal data from the phase III POLARIX study, where Polivy plus R-CHP significantly improved progression-free survival with comparable safety versus the standard of care, R-CHOP
First-line treatment with Polivy plus R-CHP has the potential to reduce the burden on patients and healthcare systems, associated with disease progression
Basel, 25 May 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the European Commission (EC) has granted approval of Polivy® (polatuzumab vedotin) in combination with MabThera®(rituximab) plus cyclophosphamide, doxorubicin, and prednisone (R-CHP) for the treatment of adult patients with previously untreated diffuse large B-cell lymphoma (DLBCL).
DLBCL is an aggressive blood cancer and the most common form of non-Hodgkin lymphoma.2 Each year in Europe, it is estimated that 40,000 people are diagnosed with DLBCL.3 While many patients are responsive to initial treatment, four out of ten are not cured with the current standard of care, and the majority of people who require subsequent lines of therapy have poor outcomes.4,5 Most DLBCL relapses occur within 24 months of starting treatment and patients who remain progression-free 24 months following initiation of first-line therapy have favourable survival outcomes.4
“After more than 20 years with very limited treatment advances, the approval of Polivy plus R-CHP marks a new era for people battling this aggressive disease,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We are delighted that the European Commission has approved this Polivy regimen and believe it has the potential to make a significant impact on the lives of people with DLBCL.”
“The approval of Polivy plus R-CHP in the first-line setting is great news for people in the EU diagnosed with this aggressive lymphoma, giving them a greater opportunity for positive outcomes,” said Professor Hervé Tilly, POLARIX Principal Investigator and Professor of Haematology at the University of Rouen. “Treatment with this regimen has been shown to reduce the chance of relapse and therefore need for subsequent treatments, limiting the burden of DLBCL.”
Today’s EC approval was based on results from the phase III POLARIX study (GO39942), the first trial to show a clinically meaningful improvement in progression free survival (PFS), compared to standard of care rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP), in people with previously untreated DLBCL. All patients were followed for at least 24 months and at a median follow-up of 28.2 months, results of the study showed a 27% reduction in the risk of disease worsening or death with Polivy plus R-CHP compared to R-CHOP in first-line DLBCL (hazard ratio [HR] 0.73; 95% confidence interval [CI]: 0.57–0.95; P<0.0177).6 The safety profile was comparable for Polivy plus R-CHP versus R-CHOP. The most frequently-reported (≥ 30%) adverse events with Polivy plus R-CHP were peripheral neuropathy (52.9%), nausea (41.6%), neutropenia (38.4%), and diarrhoea (30.8%).6 Results were presented for the first time in December 2021 at the 63rd American Society of Hematology Annual Meeting & Exposition and simultaneously published in the New England Journal of Medicine. The POLARIX study is being conducted in collaboration with The Lymphoma Study Association (LYSA) and The Lymphoma Academic Research Organisation (LYSARC).
In addition to today’s approval, the EC also converted Polivy's initial conditional marketing authorisation in the EU for the treatment of adult patients with relapsed or refractory DLBCL, who are not candidates for a haematopoietic stem cell transplant, to a full approval.
Roche continues to explore areas where Polivy has the potential to deliver additional benefit, including ongoing studies investigating combinations of Polivy with CD20xCD3 T-cell engaging bispecific antibodies in previously treated/untreated DLBCL.
Polivy is a first-in-class anti-CD79b antibody-drug conjugate (ADC). The CD79b protein is expressed specifically in the majority of B-cells, an immune cell impacted in some types of non-Hodgkin lymphoma (NHL), making it a promising target for the development of new therapies.7,8 Polivy binds to cancer cells such as CD79b and destroys these B-cells through the delivery of an anti-cancer agent, which is thought to minimise the effects on normal cells.9,10 Polivy is being developed by Roche using Seagen ADC technology and is currently being investigated for the treatment of several types of NHL. Polivy is currently marketed in the EU for the treatment of relapsed or refractory diffuse large B-cell lymphoma.
POLARIX [NCT03274492] is an international phase III, randomised, double-blind, placebo-controlled study evaluating the efficacy, safety and pharmacokinetics of Polivy® (polatuzumab vedotin) plus MabThera® (rituximab), cyclophosphamide, doxorubicin, and prednisone (R-CHP) versus rituximab, cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP) in people with previously untreated diffuse large B-cell lymphoma. Eight-hundred and seventy-nine patients were randomised 1:1 to receive either Polivy plus R-CHP plus a vincristine placebo for six cycles, followed by rituximab for two cycles; or R-CHOP plus a Polivy placebo for six cycles, followed by two cycles of rituximab. The primary outcome measure is progression-free survival (PFS) as assessed by the investigator using the Lugano Response Criteria for malignant lymphoma. PFS is a clinically meaningful disease-related outcome for patients with previously untreated DLBCL as it represents the goal of first-line therapy: decreasing the risk of disease worsening.
The Lymphoma Study Association, or LYSA, is the internationally leading cooperative group for lymphoma research in Europe, conducting clinical studies ranging from the first tests of new medicines in humans to the establishment of reference therapeutic strategies. LYSA includes in its network more than 90 care centres distributed throughout three countries (France, Belgium, Portugal), and collaborates with many scientific teams at the international level.
The Lymphoma Academic Research Organisation, or LYSARC, is the LYSA operational structure that conducts clinical research projects on lymphomas at the international level.
DLBCL is the most common form of non-Hodgkin lymphoma (NHL), accounting for about one in three cases of NHL. 2 DLBCL is an aggressive (fast-growing) type of NHL.2 While it is generally responsive to treatment in the frontline, as many as 40% of people will relapse or have refractory disease, at which time salvage therapy options are limited and survival is short. Approximately 150,000 people worldwide are estimated to be diagnosed with DLBCL each year.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera® (rituximab), Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclyxto® (venetoclax) in collaboration with AbbVie, and Hemlibra® (emicizumab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies, glofitamab and mosunetuzumab, targeting both CD20 and CD3, and cevostamab, targeting both FcRH5 and CD3; Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1 and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Pivotal data on glofitamab, a potential first-in-class CD20xCD3 T-cell engaging bispecific antibody, in heavily pre-treated patients with aggressive lymphoma, will be presented as part of our industry-leading haematology portfolio
Further studies exploring broad genomic testing to support informed treatment decisions for patients and advance cancer care approaches will be presented
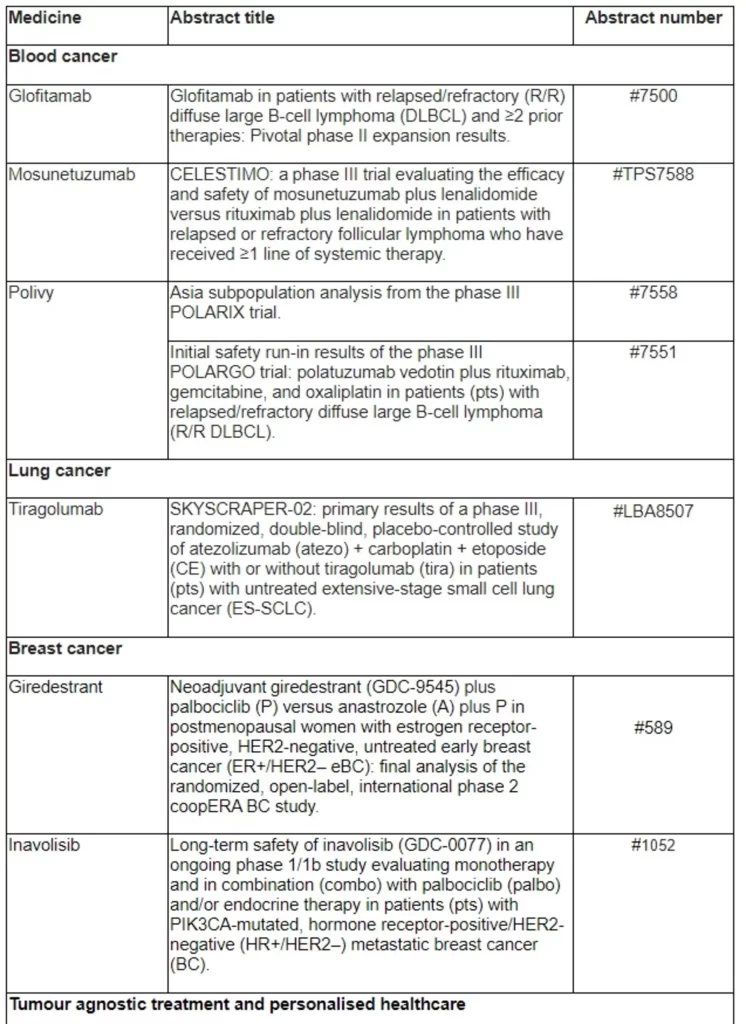
Basel, 24 May 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that new data from clinical trials of 18 approved and investigational medicines across more than 20 cancer types will be presented at the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting, which will be held 3-7 June, 2022. Roche and its partners will present clinical studies across medicines, comprehensive genomic tests, and real-world data at this year’s meeting.
“At ASCO this year, progress from our portfolio, partnerships and collaborations showcase our commitment to advance innovation in cancer care,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We’re especially pleased to present data from our broad haematology portfolio, including pivotal data for glofitamab, a potential first-in-class bispecific antibody that may improve the lives of people with heavily pre-treated aggressive lymphoma.”
New and updated data in non-Hodgkin lymphoma will be presented at ASCO. This includes pivotal data from the phase II NP30179 study evaluating glofitamab, an investigational CD20xCD3 T-cell engaging bispecific antibody, in heavily pre-treated patients with diffuse large B-cell lymphoma (DLBCL). DLBCL is an aggressive form of lymphoma, where as many as 40% of patients will relapse, at which point treatment options are limited and survival is shortened.1,2 Glofitamab is part of Roche’s broad bispecific antibody development programme, which may offer a new immunotherapy-based approach to tackle a range of blood cancers. It is being investigated in several clinical trials including the STARGLO phase III study, evaluating glofitamab in combination with gemcitabine and oxaliplatin (GemOx) versus MabThera®/Rituxan® (rituximab) in combination with GemOx in autologous stem-cell transplant ineligible relapsed or refractory DLBCL. In addition, key findings from an analysis of the Asia subpopulation from the pivotal phase III POLARIX study investigating Polivy® (polatuzumab vedotin) in combination with MabThera/Rituxan plus cyclophosphamide, doxorubicin and prednisone (R-CHP) in people with newly diagnosed DLBCL will be featured. Polivy plus R-CHP is the first treatment regimen to significantly improve outcomes in previously untreated DLBCL in more than 20 years, potentially transforming treatment for people with this disease.
More than 20 new pieces of research from partnerships with Foundation Medicine will be presented, which continue to support innovation as well as progress in personalised cancer care. This includes new data from the phase II Profiler02 study,* which investigates the use of a comprehensive genomic profiling testing panel from Foundation Medicine, with the aim of informing possible treatment decisions for patients based on their tumour’s unique genomic information.
Additionally, three abstracts from the Immunotherapy Centers Of Research Excellence (imCORE) Network will be presented at ASCO: a phase I study investigating autogene cevumeran (an mRNA-based individualised neoantigen-specific immunotherapy [iNeST]***) in the adjuvant setting of pancreatic ductal adenocarcinoma;** a data mining study evaluating intermediate endpoints for survival in metastatic breast cancer in the real-world setting;** and a study identifying mechanisms of acquired resistance to immune checkpoint blockade.**
imCORE is an academic-industry network for scientific collaboration. Established by Roche and connecting experts from 26 leading institutions around the globe, imCORE is committed to advancing and accelerating cancer immunotherapy research. imCORE is an example of Roche’s dedication to collaborating with the global cancer community to further understand cancer biology and immunology, help inform the development of potential future treatment, and transform patients’ lives.
Roche’s data presented at ASCO will feature its efforts to drive innovation and commitment to health equity through delivery of pioneering medicines and personalised cancer care that together improve outcomes for every patient while reducing the cost to society, inclusive clinical trials that remove barriers to participation, partnerships that multiply our ability to address challenges in cancer care, and bringing innovation into earlier stages of disease to maximise a chance of cure.
Roche’s Oncology Newsroom will be available to journalists from 24 May and feature exclusive materials providing insights into Roche’s vision, the latest data and perspectives on health inequities in cancer care. To access the Newsroom, please register here.
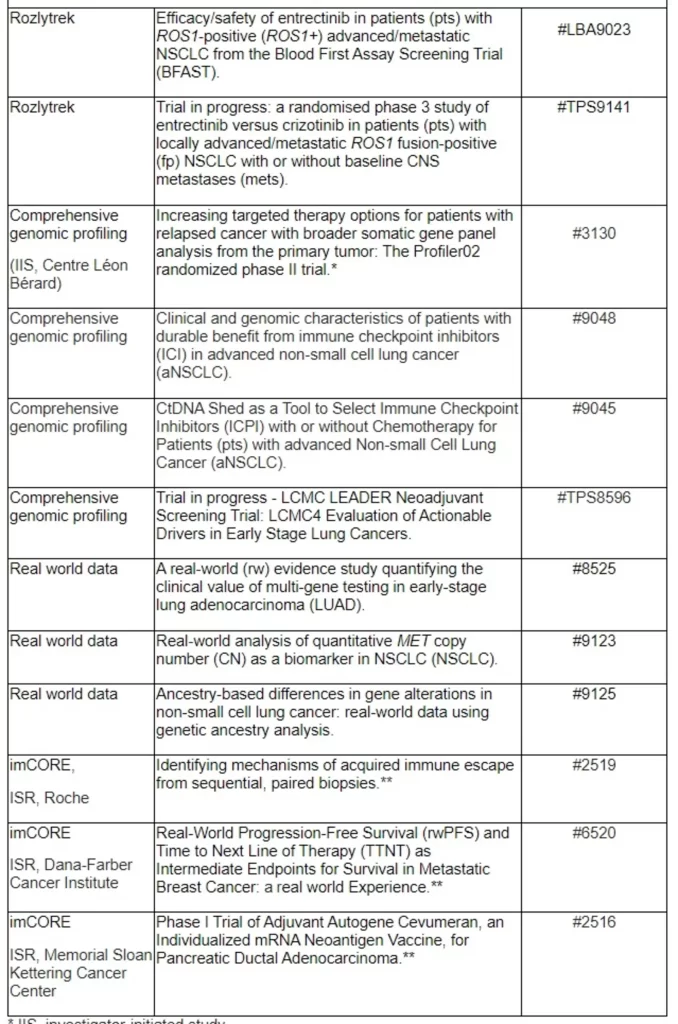
* IIS, investigator-initiated study
** ISR, institution-sponsored research
*** jointly developed by Roche and BioNTech
Roche has been working to transform cancer care for more than 50 years, bringing the first specifically designed anti-cancer chemotherapy drug, fluorouracil, to patients in 1962. Roche’s commitment to developing innovative medicines and diagnostics for cancers remains steadfast. The Roche Group’s portfolio of innovative cancer medicines includes: Alecensa® (alectinib); Avastin® (bevacizumab); Cotellic® (cobimetinib); Erivedge® (vismodegib); Gavreto® (pralsetinib); Gazyva®/Gazyvaro® (obinutuzumab); Herceptin® (trastuzumab); Kadcyla® (trastuzumab emtansine); MabThera®/Rituxan® (rituximab); Perjeta® (pertuzumab); Polivy® (polatuzumab vedotin); Tarceva® (erlotinib); Rozlytrek® (entrectinib); Tecentriq® (atezolizumab); Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie; Xeloda® (capecitabine); Zelboraf® (vemurafenib). Furthermore, the Roche Group has a robust investigational oncology pipeline focusing on new therapeutic targets and novel combination strategies. For more information on Roche’s approach to cancer, visit www.roche.com.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The SKYSCRAPER-01 study evaluating tiragolumab plus Tecentriq did not meet its co-primary endpoint of progression-free survival
The other co-primary endpoint of overall survival was immature and the study will continue until the next planned analysis
The tiragolumab development programme continues as planned in non-small cell lung cancer and other cancer types
Basel, 11 May 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced results from its phase III SKYSCRAPER-01 study, evaluating the investigational anti-TIGIT immunotherapy tiragolumab plus Tecentriq® (atezolizumab) versus Tecentriq alone as an initial (first-line) treatment for people with PD-L1-high locally advanced or metastatic non-small cell lung cancer (NSCLC). The study did not meet its co-primary endpoint of progression-free survival. At this first analysis, the other co-primary endpoint of overall survival (OS) was immature, and the study will continue until the next planned analysis. A numerical improvement was observed in both co-primary endpoints. Data suggest that tiragolumab plus Tecentriq was well-tolerated and no new safety signals were identified when adding tiragolumab. Further analyses of these results are ongoing and data will be presented at an upcoming medical meeting.
“While these results are not what we hoped for in our first analysis, we look forward to seeing mature overall survival for this study to determine next steps,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We continue to believe that TIGIT may have a role in cancer treatment and we will share additional results from our tiragolumab programme as they emerge.”
The tiragolumab programme continues to explore advances in multiple clinical trials to build on Tecentriq, expand into earlier stages of disease, and seeks to provide new treatment options in advanced and difficult-to-treat cancers with high unmet medical need.
SKYSCRAPER-01 is a global phase III, randomised double-blinded study evaluating tiragolumab plus Tecentriq® (atezolizumab) versus Tecentriq alone in 534 patients with first-line PD-L1-high locally advanced, unresectable or metastatic non-small cell lung cancer. Patients were randomized 1:1 to receive either tiragolumab plus Tecentriq or placebo plus Tecentriq, until disease progression, loss of clinical benefit or unacceptable toxicity. Co-primary endpoints are overall survival and progression-free survival.
Tiragolumab is an investigational novel immune checkpoint inhibitor with an intact Fc region. Tiragolumab selectively binds to TIGIT, a novel inhibitory immune checkpoint which suppresses the immune response to cancer.1 Based on preclinical research, tiragolumab is thought to work as an immune amplifier with other cancer immunotherapies such as Tecentriq® (atezolizumab).2 The TIGIT pathway is distinct but complementary to the PD-L1/PD-1 pathway. Dual blockade with tiragolumab and Tecentriq may help overcome immune suppression and restore the immune response.
Tecentriq is a cancer immunotherapy approved for some of the most aggressive and difficult-to-treat forms of cancer. Tecentriq was the first cancer immunotherapy approved for the treatment of a certain type of early-stage (adjuvant) non-small cell lung cancer (NSCLC), small cell lung cancer (SCLC) and hepatocellular carcinoma. Tecentriq is also approved in the US, EU and countries around the world, either alone or in combination with targeted therapies and/or chemotherapies, for various forms of metastatic NSCLC, certain types of metastatic urothelial cancer, PD-L1-positive metastatic triple-negative breast cancer and BRAF V600 mutation-positive advanced melanoma. Tecentriq is a monoclonal antibody designed to bind with a protein called programmed death ligand-1 (PD-L1), which is expressed on tumour cells and tumour-infiltrating immune cells, blocking its interactions with both PD-1 and B7.1 receptors. By inhibiting PD-L1, Tecentriq may enable the activation of T-cells. Tecentriq is a cancer immunotherapy that has the potential to be used as a foundational combination partner with other immunotherapies, targeted therapies and various chemotherapies across a broad range of cancers. In addition to intravenous infusion, the formulation of Tecentriq is also being investigated as subcutaneous injection to hopefully provide a faster and more convenient option for cancer patients.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
