Infectious diseases, such as those infections transmitted via respiratory, sexually and transplant, are becoming increasingly difficult to manage and represent an ever-growing medical need for millions of patients worldwide.
Access to accurate and reliable diagnostic tests help clinicians improve patient care, limit healthcare spending and contribute to protecting public health.
The cobas 5800 System is a compact new addition to the Roche Molecular portfolio, providing efficiency, simplicity and timely results to laboratories of all sizes so clinicians can quickly determine the best treatment strategies for their patients.
Basel, 18 November 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the launch of the cobas® 5800 System, a new molecular laboratory instrument, in countries accepting the CE mark. Testing is one of the first lines of defense to protect a patient’s general well-being and is vitally important in quickly guiding their treatment. The cobas 5800 System helps address challenges that laboratories are facing from an increase in patient testing, reimbursement complexities and the need for a more diverse testing menu while providing meaningful and timely results.
“Diagnostics continue to revolutionise how providers care for patients, and it is essential, now more than ever, that the healthcare community has access to reliable and accurate testing solutions,” said Thomas Schinecker, CEO Roche Diagnostics. ”We are pleased that we can now offer a compact, fully-automated molecular system such as the cobas 5800 System. This new system helps to address the high demands on labs through greater efficiency, optimised workflows and cost savings, enabling them to deliver quality patient care.”
The cobas 5800 system, the newest member of the Roche Molecular Work Area, is built upon the innovation of the cobas® 6800 and cobas® 8800 systems, making molecular testing accessible to more patients around the world.
Roche anticipates receiving U.S. Food and Drug Administration approval during Q3 2022.
The cobas 5800 System is a real-time PCR molecular testing solution that provides excellent performance and value-added utility from a compact footprint. The cobas 5800 System delivers automation, consolidation, integration and standardisation – making it a scalable, cost-efficient solution for smaller labs seeking big lab performance, or for bigger labs seeking small lab agility.
The system is built to offer a fully automated workflow that encompasses sample supply, transfer and preparation, amplification and detection, result calculation and delivery to the laboratory information system (LIS). The single module enables a walkaway time of up to eight hours*, resulting in less hands-on time, which can lead to improved productivity, reduced possibility for errors and more predictable turnaround times.
The cobas 5800 System will offer the same menu as the cobas 6800 and 8800 Systems, including the cobas® SARS-CoV-2 test. The system offers an expanded on board capacity allowing labs to test multiple assays simultaneously and delivers up to 144 results in an eight-hour shift. This streamlined molecular testing system will allow most labs to consolidate greater than 90 percent** of their routine molecular tests onto a single platform
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
EU marketing authorisation follows approvals in Japan, the United Kingdom and Australia
Approval based on data demonstrating Ronapreve reduced risk of hospitalisation in certain patients with mild to moderate disease and reduced risk of symptomatic COVID-19 infections in people exposed to the virus.
Basel, 12 November 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the European Commission has granted a marketing authorisation for Ronapreve™ (casirivimab and imdevimab), for treating COVID-19 in adults and adolescents (from 12 years of age and weighing at least 40 kilograms) who do not require supplemental oxygen and who are at increased risk of their disease becoming severe, and for preventing COVID-19 in people aged 12 years and older weighing at least 40 kilograms (pre- or post-exposure prophylaxis). This decision follows one day after the positive opinion by the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP), reflecting the EMA and the European Commission’s priority to shorten review timelines for safe, effective and high-quality therapeutics during the COVID-19 public health emergency.
“We welcome this quick approval from the European Commission, which adds to the growing number of health authorities that recognize Ronapreve as an important therapy for the treatment and prevention of COVID-19,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Although vaccinations are increasing globally, Europe is entering a fourth wave of rising cases and treatment options for the full range of disease severity and variants of concern are still needed. We look forward to working with additional global regulatory bodies as we continue to tackle COVID-19 together.”
Today’s decision from the European Commission is based on the EMA’s CHMP’s review of positive data from the REGN-COV 2067 treatment study in non-hospitalised patients and the REGN-COV 2069 prophylaxis study in people exposed to SARS-CoV-2 virus. Roche will continue to work with the EMA to potentially extend the marketing authorisation of Ronapreve to treat hospitalised patients with COVID-19.
Outside of the European Union, Ronapreve has been approved for use in Japan and conditionally in the United Kingdom and Australia, and is authorised for emergency or temporary pandemic use in additional territories, including the United States, India and Canada. Ronapreve, being jointly developed by Roche and Regeneron, is currently available in nearly 50 countries via bilateral purchase agreements across many geographies and economies, including lower middle-income countries. In addition, the World Health Organization recommended the use of Ronapreve for the treatment of patients with COVID-19.
COVID-19 remains prevalent across the world, partly due to the multiple SARS-CoV-2 variants in circulation. In the past two months, new cases in Europe have reached >180,000 per day consistently. Currently, the region reports the highest weekly case incidence rates worldwide and cases are on the rise as we enter the winter season.1 In these exceptional times, Roche stands together with society, governments, healthcare providers and all those working towards the common goal of overcoming the COVID-19 pandemic.
The efficacy and safety of Ronapreve™ (casirivimab and imdevimab, known as REGEN-COV®in the United States) have been studied across multiple phase III clinical trials in non-hospitalised and hospitalised COVID-19 patients, and in the preventive setting. In addition, data from preclinical studies showed that Ronapreve retained neutralisation activity against key emerging variants, as referenced in publications in Cell and Nature.
The decision from the European Commission is based on data from multiple studies, including:
the REGN-COV 2067 study, showing that Ronapreve reduced hospitalisation or death by 70% and symptom duration by four days.
the REGN-COV 2069 study, showing that the administration of Ronapreve reduced the risk of symptomatic infections by 81% in those who were not infected when they entered the trial.
There have been no new safety signals identified for Ronapreve in these studies.
Ronapreve is being jointly developed by Roche and Regeneron. It is a combination of two monoclonal antibodies, casirivimab and imdevimab, and was designed to block infectivity of SARS-CoV-2, the virus that causes COVID-19.
As a leading healthcare company, we are doing all we can to support countries in their fight against COVID-19 and minimising its impact. We have developed a growing number of diagnostic solutions that help to detect and diagnose the infection, as well as providing digital support to healthcare systems. We also continue to identify, develop and support potential therapies which can play a role in treating the disease.
The impact of COVID-19 goes beyond those who contract it. That is why we are working with healthcare providers, laboratories, authorities and organisations to help make sure patients continue to receive the tests, treatment and care they need during these challenging times. Building on a longstanding tradition of partnerships, we are working together with governments and others to make healthcare stronger and more sustainable in the future.
Reliable, high-quality testing is essential to help healthcare systems overcome this pandemic and Roche has so far launched 16 diagnostics solutions to help minimise the impact of COVID-19. As soon as the novel SARS-CoV-2 virus was sequenced in early 2020, we got to work. On 13 March 2020 we became the first company to receive U.S. Food and Drug Administration (FDA) Emergency Use Authorization (EUA) for a high-volume molecular test to detect the virus. Since then, we have continued to add a range of diagnostics solutions to our global portfolio to help in the fight against COVID-19. In addition to the gold standard PCR test, we have developed antigen tests to; help diagnose the virus in settings where there is limited molecular laboratory infrastructure, rapid antigen tests where the virus can be detected on the spot, tests that can test for both flu and COVID-19 at the same time, both high throughput and at the point of care, and tests that can detect virus antibodies that can help monitor the spread of the virus and can also support in vaccine development. In March 2021 the SARS-CoV-2 variant test was launched, designed to detect key spike mutations.
Aside from these tests we have also looked at how we can support care for patients who have COVID-19, receiving an FDA EUA for the Elecsys® IL-6 test to assist in identifying severe inflammatory response in patients with confirmed COVID-19, as well as launching Roche v-TAC, a digital algorithm that could help simplify the screening, diagnosis and monitoring of respiratory-compromised patients with COVID-19. Roche is working closely with governments and health authorities around the world, and has significantly increased production to support availability of tests globally.
Roche is actively involved in understanding the potential of the existing portfolio and is researching options for the future. In 2020, Roche entered into a number of new partnerships, including with Regeneron, Atea and Gilead to develop, manufacture and distribute molecules that can potentially both treat and prevent COVID-19.
In October 2020, Roche announced a partnership with Atea Pharmaceuticals to jointly develop the investigational compound AT-527. AT-527 is still being evaluated in clinical trials, across non-hospitalised and hospitalised COVID-19 patients and is not currently approved or authorised for the treatment of COVID-19 by any health authority.
In addition, we have explored the potential of our existing medicine Actemra/RoActemra in three global phase III clinical trials investigating its safety and efficacy in COVID-19 associated pneumonia (COVACTA, EMPACTA and REMDACTA). In June 2021, Actemra/RoActemra received an Emergency Use Authorization from the U.S. FDA for the intravenous treatment of COVID-19 in hospitalised adults and paediatric patients (2 years of age and older) who are receiving systemic corticosteroids and require supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation. In addition, the World Health Organization recommended the use of Actemra/RoActemra for the treatment of certain patients with COVID-19.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
EU marketing authorisation follows approvals in Japan, the United Kingdom and Australia
Approval based on data demonstrating Ronapreve reduced risk of hospitalisation in certain patients with mild to moderate disease and reduced risk of symptomatic COVID-19 infections in people exposed to the virus.
Basel, 12 November 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the European Commission has granted a marketing authorisation for Ronapreve™ (casirivimab and imdevimab), for treating COVID-19 in adults and adolescents (from 12 years of age and weighing at least 40 kilograms) who do not require supplemental oxygen and who are at increased risk of their disease becoming severe, and for preventing COVID-19 in people aged 12 years and older weighing at least 40 kilograms (pre- or post-exposure prophylaxis). This decision follows one day after the positive opinion by the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP), reflecting the EMA and the European Commission’s priority to shorten review timelines for safe, effective and high-quality therapeutics during the COVID-19 public health emergency.
“We welcome this quick approval from the European Commission, which adds to the growing number of health authorities that recognize Ronapreve as an important therapy for the treatment and prevention of COVID-19,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Although vaccinations are increasing globally, Europe is entering a fourth wave of rising cases and treatment options for the full range of disease severity and variants of concern are still needed. We look forward to working with additional global regulatory bodies as we continue to tackle COVID-19 together.”
Today’s decision from the European Commission is based on the EMA’s CHMP’s review of positive data from the REGN-COV 2067 treatment study in non-hospitalised patients and the REGN-COV 2069 prophylaxis study in people exposed to SARS-CoV-2 virus. Roche will continue to work with the EMA to potentially extend the marketing authorisation of Ronapreve to treat hospitalised patients with COVID-19.
Outside of the European Union, Ronapreve has been approved for use in Japan and conditionally in the United Kingdom and Australia, and is authorised for emergency or temporary pandemic use in additional territories, including the United States, India and Canada. Ronapreve, being jointly developed by Roche and Regeneron, is currently available in nearly 50 countries via bilateral purchase agreements across many geographies and economies, including lower middle-income countries. In addition, the World Health Organization recommended the use of Ronapreve for the treatment of patients with COVID-19.
COVID-19 remains prevalent across the world, partly due to the multiple SARS-CoV-2 variants in circulation. In the past two months, new cases in Europe have reached >180,000 per day consistently. Currently, the region reports the highest weekly case incidence rates worldwide and cases are on the rise as we enter the winter season.1 In these exceptional times, Roche stands together with society, governments, healthcare providers and all those working towards the common goal of overcoming the COVID-19 pandemic.
The efficacy and safety of Ronapreve™ (casirivimab and imdevimab, known as REGEN-COV®in the United States) have been studied across multiple phase III clinical trials in non-hospitalised and hospitalised COVID-19 patients, and in the preventive setting. In addition, data from preclinical studies showed that Ronapreve retained neutralisation activity against key emerging variants, as referenced in publications in Cell and Nature.
The decision from the European Commission is based on data from multiple studies, including:
the REGN-COV 2067 study, showing that Ronapreve reduced hospitalisation or death by 70% and symptom duration by four days.
the REGN-COV 2069 study, showing that the administration of Ronapreve reduced the risk of symptomatic infections by 81% in those who were not infected when they entered the trial.
There have been no new safety signals identified for Ronapreve in these studies.
Ronapreve is being jointly developed by Roche and Regeneron. It is a combination of two monoclonal antibodies, casirivimab and imdevimab, and was designed to block infectivity of SARS-CoV-2, the virus that causes COVID-19.
As a leading healthcare company, we are doing all we can to support countries in their fight against COVID-19 and minimising its impact. We have developed a growing number of diagnostic solutions that help to detect and diagnose the infection, as well as providing digital support to healthcare systems. We also continue to identify, develop and support potential therapies which can play a role in treating the disease.
The impact of COVID-19 goes beyond those who contract it. That is why we are working with healthcare providers, laboratories, authorities and organisations to help make sure patients continue to receive the tests, treatment and care they need during these challenging times. Building on a longstanding tradition of partnerships, we are working together with governments and others to make healthcare stronger and more sustainable in the future.
Reliable, high-quality testing is essential to help healthcare systems overcome this pandemic and Roche has so far launched 16 diagnostics solutions to help minimise the impact of COVID-19. As soon as the novel SARS-CoV-2 virus was sequenced in early 2020, we got to work. On 13 March 2020 we became the first company to receive U.S. Food and Drug Administration (FDA) Emergency Use Authorization (EUA) for a high-volume molecular test to detect the virus. Since then, we have continued to add a range of diagnostics solutions to our global portfolio to help in the fight against COVID-19. In addition to the gold standard PCR test, we have developed antigen tests to; help diagnose the virus in settings where there is limited molecular laboratory infrastructure, rapid antigen tests where the virus can be detected on the spot, tests that can test for both flu and COVID-19 at the same time, both high throughput and at the point of care, and tests that can detect virus antibodies that can help monitor the spread of the virus and can also support in vaccine development. In March 2021 the SARS-CoV-2 variant test was launched, designed to detect key spike mutations.
Aside from these tests we have also looked at how we can support care for patients who have COVID-19, receiving an FDA EUA for the Elecsys® IL-6 test to assist in identifying severe inflammatory response in patients with confirmed COVID-19, as well as launching Roche v-TAC, a digital algorithm that could help simplify the screening, diagnosis and monitoring of respiratory-compromised patients with COVID-19. Roche is working closely with governments and health authorities around the world, and has significantly increased production to support availability of tests globally.
Roche is actively involved in understanding the potential of the existing portfolio and is researching options for the future. In 2020, Roche entered into a number of new partnerships, including with Regeneron, Atea and Gilead to develop, manufacture and distribute molecules that can potentially both treat and prevent COVID-19.
In October 2020, Roche announced a partnership with Atea Pharmaceuticals to jointly develop the investigational compound AT-527. AT-527 is still being evaluated in clinical trials, across non-hospitalised and hospitalised COVID-19 patients and is not currently approved or authorised for the treatment of COVID-19 by any health authority.
In addition, we have explored the potential of our existing medicine Actemra/RoActemra in three global phase III clinical trials investigating its safety and efficacy in COVID-19 associated pneumonia (COVACTA, EMPACTA and REMDACTA). In June 2021, Actemra/RoActemra received an Emergency Use Authorization from the U.S. FDA for the intravenous treatment of COVID-19 in hospitalised adults and paediatric patients (2 years of age and older) who are receiving systemic corticosteroids and require supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation. In addition, the World Health Organization recommended the use of Actemra/RoActemra for the treatment of certain patients with COVID-19.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Every year, over 604,000 women worldwide are diagnosed with cervical cancer and approximately 342,000 die from this preventable disease, caused by Infection with high-risk human papillomavirus (HPV).
As recently published in the International Journal of Cancer, the IMPACT (IMproving Primary screening And Colposcopy Triage) trial demonstrated that Roche's CINtec PLUS Cytology, when used as a triage test for high-risk HPV, showed a significantly higher sensitivity in detecting cervical pre-cancers in HPV-positive women compared to Pap cytology.
It is the first FDA-approved dual-stain biomarker technology available for clinicians to guide decision-making based on individual patient risk, with the goal of helping to eliminate cervical cancer.
Basel, 29 October 2021 – Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that results from the IMPACT (IMproving Primary screening And Colposcopy Triage) trial demonstrate clear patient benefit in using Roche’s CINtec PLUS Cytology dual-stain biomarker technology as a triage test for women who test positive for high-risk human papillomavirus (HPV). The data from the trial, established from a study cohort of more than 35,000 women aged 25-65 years, was published recently in the International Journal of Cancer.
In the IMPACT trial, women who were positive for high-risk HPV received a follow-up triage test to help determine if their cervical cells were transforming to cervical pre-cancer. The biomarker-based CINtec PLUS Cytology test showed a significantly higher sensitivity in detecting cervical pre-cancers, compared to Pap cytology. The Roche test aids clinicians in more confidently determining which women are at increased risk for high-grade cervical pre-cancer and require immediate further diagnostic procedures, and which women may need repeat testing or routine screening.
Over 604,000 women are diagnosed with cervical cancer worldwide each year and approximately 342,000 die from the disease.2 Persistent infection with high-risk HPV is the principal cause of cervical cancer, implicated in more than 99 percent of cases worldwide.3 Cervical cancer is nearly 100 percent preventable with proper HPV vaccination, screening and treatment.
“As we approach the one-year anniversary of the World Health Organization’s global strategy to accelerate the elimination of cervical cancer, Roche is committed to investing in and leading efforts such as the IMPACT trial to bring forth clinically validated solutions for women,” said Thomas Schinecker, CEO Roche Diagnostics. “The elimination of cervical cancer is within reach, and all countries must act now so that women, no matter where in the world they live, no longer die from this preventable disease. Our investment in HPV primary screening and next-generation biomarker technology gives clinicians even more powerful tools in the fight against cervical cancer.”
"These latest results from the IMPACT trial confirm data from previous studies that show incorporating the CINtec PLUS Cytology test in cervical cancer screening programs can provide real benefits to both clinicians and their patients," says Dr. Thomas Wright, Professor Emeritus in Pathology and Cell Biology at Columbia University Medical Center, New York. "As a triage test for HPV-positive cervical cancer screening results, the CINtec PLUS Cytology test can be very useful to differentiate women who will benefit most from immediate referral to colposcopy from those women who can be followed up with less invasive methods.
Cervical cancer is nearly 100 percent preventable with proper HPV vaccination, screening and treatment. More than 604,000 new cases of cervical cancer are diagnosed each year worldwide. In 2020, cervical cancer was responsible for 7.7 percent of all female cancer deaths.2,4
The landmark IMPACT trial was a prospective observational cervical cancer screening clinical study that enrolled approximately 35,000 women aged 25-65 years who were undergoing routine cervical cancer screening at 32 clinical sites in 16 states across the US. The study provides validation for the clinical utility of cobas HPV testing for primary screening in combination with CINtec PLUS Cytology as a follow-up test for patients with positive screening results.
The study showed that triaging with CINtec PLUS Cytology may lead to significantly improved detection of cervical disease when women are screened for cervical cancer. Cervical screening helps identify women at risk for disease before invasive cancer develops. While most HPV infections resolve on their own, some women who test positive for the virus may develop pre-cancerous cervical lesions that, if left untreated, may progress to cervical cancer. Early identification of women who are most at risk is vital.
In the study, HPV-positive women with CINtec PLUS Cytology negative triage test results showed a very low cumulative 1-year risk for disease, which was significantly lower than the risks associated with a negative Pap cytology triage test result in HPV-positive women.
Based on the results of the IMPACT trial, the FDA approved the CINtec PLUS Cytology test to be used as triage for positive HPV test results using cobas HPV on cobas 4800, 6800 and 8800 Systems in primary screening or co-testing programs.
Recommended clinical guidelines have also been evolving in favor of HPV tests for primary screening, supported by an interest to improve outcomes and the availability of technologies to help laboratories achieve the efficiency and scale they need to meet the demands of high-volume cervical screening programs.
The CINtec PLUS Cytology test detects the simultaneous presence within a single cell of the two biomarkers -- p16 and Ki-67. This abnormality is associated with HPV infections that are transforming and can, if left untreated, progress to pre-cancer or cancer. A positive result of these two biomarkers in a single cell signals that a woman is more significantly at risk for disease. The ability of CINtec PLUS Cytology to distinguish those women who are at a higher risk for cervical disease, in conjunction with the clinician's assessment of patient screening history and other risk factors, provides labs, physicians and women with the information needed to guide patient management. Women with negative dual-stain results are at significantly lower risk for cervical disease and their bodies can be given more time to clear the HPV infection on their own. This could reduce the number and frequency of follow-up visits, saving some patients worry and time.
The CINtec PLUS Cytology test, which runs on the BenchMark ULTRA IHC/ISH system, is performed using the same sample that is used for HPV or liquid-based Pap cytology tests. This eliminates the need for additional or repeat sample collection or time spent waiting to find out if an infection is clearing.
CINtec PLUS Cytology, now available globally, was FDA approved in March 2020.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The AVENIO Tumor Tissue Comprehensive Genomic Profiling (CGP) Kit is the first jointly-developed product that brings together the expertise and reach of Roche with Foundation Medicine’s pioneering leadership in genomic science.
This is the first time that research scientists can conduct oncology research with Foundation Medicine’s comprehensive genomic profiling in their own laboratories to gain meaningful genomic insights and deepen their understanding of cancer biology.
This kit is an important step forward in Roche and Foundation Medicine's vision of enabling personalised healthcare for more patients living with cancer by broadening access to CGP and making it essential in clinical decision making in the future.
Basel, 25 October 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that it has launched the AVENIO Tumor Tissue CGP Kit. The Kit complements the current CGP portfolio offered by Roche and Foundation Medicine and allows laboratories to expand their oncology research in-house. Ultimately, a future version of the kit may lead to additional resources for clinicians to use in the diagnosis and treatment of cancer.
Cancer is a disease of the genome and treatment no longer depends solely on the tissue of origin. Rather, cancer is a collection of dozens or even hundreds of diseases driven in part by specific genomic characteristics.
“To treat cancer effectively, we must understand what drives it at a molecular level. CGP helps inform decisions about available treatment options, including targeted therapies, immunotherapies, tumour-agnostic treatments and clinical trial participation, based on the unique genomic profile of a patient’s tumour,” said Thomas Schinecker, CEO of Roche Diagnostics. “We are bringing powerful insights from CGP to enable smarter, more efficient research and development. Our new kit provides the information researchers need and will ultimately provide insights physicians can use to develop personalised treatment strategies for individual patients.”
Unlike smaller panels such as hotspot or single gene tests, CGP tests deliver comprehensive information in a single test and can also provide information on complex genomic signatures such as Tumour Mutational Burden (TMB), Microsatellite Instability (MSI) and Loss of Heterozygosity (LOH).
The kit for research use is just the first step in bringing CGP closer to people living with cancer through enabling critical research and clinical trials in laboratories across the world. The kit will launch in regions throughout the world including Europe, North America, Asia and South America. Roche and Foundation Medicine also plan to develop additional solutions for its portfolio.
“The launch of this kit will significantly expand access to genomic profiling globally by providing an in-house solution for those who cannot otherwise access our portfolio of tests through our centralised laboratories,” said Foundation Medicine Chief Executive Officer Brian Alexander. “The launch of the kit strengthens our ability to act as an essential partner to researchers in navigating the complex landscape of cancer care.”
The AVENIO Tumor Tissue CGP Kit is a Research Use Only next-generation sequencing test that provides an end-to-end workflow from DNA extraction and library preparation to generation of variant results by the FoundationOne Analysis Platform via AVENIO Connect. The Kit uses a gene panel based on the FoundationOne platform that was designed to match the FoundationOne CDx panel content to analyse 324 cancer-related genes across four main classes of genomic alterations and genomic signatures known to cause and drive cancer. These genes provide valuable insights on biomarkers known today and emerging biomarkers of tomorrow.
Each of the kits is configured for running 24 samples, with the entire workflow from DNA isolation to variant results completed in 5 days. The kit runs on Illumina’s NextSeq 500/550 RUO and NextSeq 550 Dx System in research mode. In January 2020, Roche entered into an agreement with Illumina to develop, manufacture and commercialise AVENIO tests for both tissue and blood for use on Illumina’s IVD systems.
Foundation Medicine, a wholly owned subsidiary of Roche, is a molecular information company dedicated to a transformation in cancer care in which treatment is informed by a deep understanding of the genomic changes that contribute to each patient's unique cancer. Foundation Medicine offers a full suite of comprehensive genomic profiling assays to identify the molecular alterations in a patient’s cancer and match them with relevant targeted therapies, immunotherapies and clinical trials. Foundation Medicine`s CGP portfolio is available in more than 100 countries worldwide.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Lung cancer remains the leading cause of cancer-related deaths worldwide, with more than 2.2 million people diagnosed globally last year.1
The VENTANA PD-L1 (SP263) Assay helps determine which non-small cell lung cancer patients may benefit from treatment with Tecentriq immunotherapy based on the results of the Phase III IMpower010 study.
This new test expands Roche’s industry leading portfolio of companion diagnostics and builds on our commitment to improve personalized healthcare for better patient outcomes.
Basel, 22 October 2021 – Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced U.S. Food and Drug Administration (FDA) approval of the VENTANA PD-L1 (SP263) Assay in non- small cell lung cancer (NSCLC) as a companion diagnostic test for Tecentriq, advancing the company's commitment to guide clinical decision making through innovative, high quality assays that improve patient access to personalized healthcare.
The current standard of care for patients with early stage lung cancer is surgery to remove the tumor, which may be followed by chemotherapy. Unfortunately, about half of these patients will have their cancer return following surgery.2 Tecentriq received FDA approval on 15 October 2021 as adjuvant treatment following surgery and platinum-based chemotherapy for adults whose Stage II-IIIA NSCLC tumors have PD-L1 expression on ≥1% of tumor cells. The VENTANA PD-L1 (SP263) Assay identifies NSCLC patients who may be eligible for Tecentriq (atezolizumab) monotherapy in this indication.
“Early detection of lung cancer can change the treatment pathway for patients and give them more treatment options,” said Thomas Schinecker, CEO Roche Diagnostics. “We are proud to offer a companion diagnostic PD-L1 test that identifies lung cancer patients who may qualify for Tecentriq therapy. With the FDA approval of this companion diagnostic test, clinicians now have an effective tool for offering better patient care through targeted immunotherapy treatment.”
The VENTANA PD-L1 (SP263) Assay was used as part of the IMpower010 study sponsored by Genentech, a member of the Roche Group, to identify patients whose tumors expressed the PD-L1 protein. The IMpower010 clinical study began in 2015 with the goal of understanding how patients would respond to treatment with Tecentriq following traditional surgery and chemotherapy. In 2021, Genentech reported a 34% reduction in the risk of disease recurrence or death amongst Tecentriq patients whose tumors were shown to express PD-L1 protein
VENTANA PD-L1 (SP263) Assay is used to detect programmed death ligand-1 (PD-L1) protein in non-small cell lung carcinoma (NSCLC) patients. PD-L1 expression on tumor cells and immune cells has been shown in clinical studies to help predict the likelihood a patient may benefit from PD-L1/PD-1 immunotherapy drugs.3-6
VENTANA PD-L1 (SP263) Assay testing is performed on a BenchMark ULTRA instrument and is visualized using the OptiView DAB IHC Detection Kit.
Roche has developed a leading, comprehensive and differentiated lung cancer immunohistochemical portfolio, with biomarkers that support multiple guidelines for the diagnosis and stratification of lung cancers
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Susvimo, previously called Port Delivery System with ranibizumab, is the first nAMD treatment in 15 years to provide an alternative to standard-of-care eye injections needed as often as once a month
By continuously delivering medicine into the eye through a refillable implant, Susvimo may help people with nAMD maintain their vision with as few as two treatments per year
Neovascular AMD impacts approximately 20 million people worldwide and is a leading cause of blindness in people over the age of 60
Basel, 22 October 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the U.S. Food and Drug Administration (FDA) has approved Susvimo™ (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant for the treatment of people with neovascular or “wet” age-related macular degeneration (nAMD) who have previously responded to at least two anti-vascular endothelial growth factor (VEGF) injections. Neovascular AMD is a potentially blinding condition that requires treatment with eye injections as often as once a month.1,2,3,4 Susvimo, previously called Port Delivery System with ranibizumab, is the first and only FDA-approved treatment for nAMD that offers as few as two treatments per year.5,6
“Susvimo represents a major advancement in the treatment of retinal disease and is an important new option for patients with wet AMD,” said Carl Regillo, M.D., Chief of Retina Service at Wills Eye Hospital in Philadelphia and an Archway study investigator. “With Susvimo, my patients now have an option that can help them maintain their vision as well as anti-VEGF injections, but on a more manageable twice-yearly treatment schedule.”
Susvimo delivers ranibizumab continuously, offering people living with nAMD an alternative to anti-VEGF eye injections needed as often as once a month.3,4,5 The implant is surgically inserted into the eye during a one-time, outpatient procedure and refilled every six months.5,6 If necessary, supplemental ranibizumab treatment can be given to the affected eye while the Susvimo implant is in place.5
“We believe that Susvimo can help people with nAMD preserve their vision while potentially alleviating the treatment burden associated with current standards of care,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Susvimo’s approval builds on Roche’s long-standing commitment to people living with vision-threatening conditions.”
The approval is based on positive results from the phase III Archway study primary analysis, which showed nAMD patients treated with Susvimo achieved and maintained vision gains equivalent to monthly ranibizumab injections – +0.2 and +0.5 eye chart letters from baseline, respectively – at weeks 36 and 40 of treatment. In addition, only 1.6% of Susvimo patients received supplemental ranibizumab treatment before their first refill, and more than 98% could go six months before their first refill.5
In the Archway study, Susvimo was generally well-tolerated, with a favourable benefit-risk profile. However, the Susvimo implant has been associated with a three-fold higher rate of endophthalmitis than monthly intravitreal injections of ranibizumab. Many of these events were associated with conjunctival retractions or erosions. Appropriate conjunctiva management and early detection with surgical repair of conjunctival retractions or erosions may reduce the risk of endophthalmitis. In clinical trials, 2.0% of patients receiving a ranibizumab implant experienced at least one episode of endophthalmitis. The most common adverse events (AEs) were conjunctival haemorrhage, conjunctival hyperaemia, iritis and eye pain.6 The safety profile of Susvimo in the clinical trial setting is well understood and will continue to be monitored closely.5
Roche has a robust phase III clinical development programme for Susvimo, including the Portal, Pagoda, Pavilion and Velodrome studies. Portal is an extension study evaluating the long-term safety and efficacy of Susvimo in nAMD.7 Pagoda is evaluating Susvimo for the treatment of people with diabetic macular edema (DME), while Pavilion is a study of Susvimo in diabetic retinopathy without DME.8,9 Velodrome is evaluating Susvimo refilled every nine months in nAMD.10 Susvimo is also currently under review for the treatment of nAMD by the European Medicines Agency (EMA).
Susvimo will be available in the United States in the coming months.
Roche’s late-stage ophthalmology portfolio also includes faricimab, a bispecific antibody under FDA and EMA review for the treatment of nAMD and DME. The FDA is additionally reviewing faricimab for the treatment of diabetic retinopathy.
Archway (NCT03677934) was a randomised, multicentre, open-label phase III study evaluating the efficacy and safety of Susvimo™ (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant administered via the Susvimo eye implant, refilled every six months at fixed intervals, compared to monthly intravitreal injections of ranibizumab 0.5 mg in 415 people living with neovascular or “wet” age-related macular degeneration (nAMD). Patients enrolled in Archway were responders to prior treatment with anti-vascular endothelial growth factor (VEGF) therapy. In both study arms, patients were treated with at least three anti-VEGF injections within the six months prior to their Archway screening visit. The primary endpoint of the study was the change in best-corrected visual acuity (BCVA) score (the best distance vision a person can achieve – including with correction such as glasses – when reading letters on an eye chart) from baseline at the average of Week 36 and Week 40. Secondary endpoints include safety, overall change in vision (BCVA) from baseline and change from baseline in centre point thickness over time.
According to pre-specified study criteria, Susvimo was shown to be non-inferior and equivalent to monthly ranibizumab injections. On average, patients had received five prior ranibizumab injections before their first study treatment visit. In the Susvimo arm of the study, patients gained an average of 0.2 eye chart letters in visual acuity from baseline compared with 0.5 eye chart letters for the monthly ranibizumab arm. During the first treatment interval, before the first scheduled refill, 1.6% of Susvimo patients assessed (n=4/246) received supplemental ranibizumab treatment, and 98.4% of patients (n=242/246) did not receive supplemental treatment.
In the Archway study, Susvimo was generally well-tolerated, with a favourable benefit-risk profile. The safety profile of Susvimo in the clinical trial setting is well understood and will continue to be monitored closely.
Age-related macular degeneration (AMD) is a condition that affects the part of the eye that provides sharp, central vision needed for activities like reading.13 Neovascular or “wet” AMD (nAMD) is an advanced form of the disease that can cause rapid and severe vision loss.1,2 It develops when new and abnormal blood vessels grow uncontrolled under the macula, causing swelling, bleeding and/or fibrosis.2 Worldwide, around 20 million people are living with nAMD – the leading cause of vision loss in people over the age of 60 – and the condition will affect even more people around the world as the global population ages.13, 14, 15
About Susvimo (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant
Susvimo™ (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant is a refillable eye implant surgically inserted into the eye during a one-time, outpatient procedure. Susvimo continuously delivers a customised formulation of ranibizumab over time.5 Susvimo is indicated for intravitreal use via the Susvimo eye implant only.6 Ranibizumab is a vascular endothelial growth factor (VEGF) inhibitor designed to bind to and inhibit VEGF-A, a protein that has been shown to play a critical role in the formation of new blood vessels and the leakiness of the vessels.4
Susvimo is different from the ranibizumab intravitreal injection, a medicine marketed as Lucentis®* (ranibizumab injection), which is approved to treat neovascular or “wet” age-related macular degeneration (nAMD) and other retinal diseases. Lucentis®️* was first approved for nAMD by the FDA in 2006.
Roche is focused on saving people’s eyesight from the leading causes of vision loss through pioneering therapies. Through our innovation in the scientific discovery of new potential drug targets, personalised healthcare, molecular engineering, biomarkers and continuous drug delivery, we strive to design the right therapies for the right patients.
We have the broadest retina pipeline in Ophthalmology, covering early- and late-stage products, which is led by science and informed by insights from people with eye diseases. Our late-stage pipeline includes faricimab, a potential first-of-a-kind treatment being evaluated in a number of retinal conditions including neovascular age-related macular degeneration (nAMD), diabetic macular edema and diabetic retinopathy. Faricimab is the first investigational bispecific antibody designed for the eye.17 It targets two distinct pathways – via angiopoietin-2 (Ang-2) and vascular endothelial growth factor-A (VEGF-A) – that drive a number of retinal conditions, to stabilise blood vessels, potentially improving vision outcomes for longer.4,17 Our early-stage pipeline includes gene therapies and treatments for geographic atrophy and other vision-threatening diseases, including rare and inherited conditions.
Applying our extensive experience, we have already brought breakthrough ophthalmic treatments to people living with vision loss through Lucentis®️* (ranibizumab injection), the first treatment approved to improve
vision in people with certain retinal conditions, and SusvimoTM (ranibizumab injection) 100 mg/mL for intravitreal use via ocular implant, the first FDA-approved refillable eye implant for nAMD that continuously delivers a customised formulation of ranibizumab over a period of months.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
35% reduction in risk of needing a walking aid in relapsing multiple sclerosis (RMS) after 7.5 years vs. initiation 2 years later in Phase III open-label extension (OLE)
29% reduction in 48-week confirmed disability progression in primary progressive MS (PPMS) after 8 years vs. initiation after double-blind period in Phase III OLE
New 8-year safety data show consistent benefit-risk profile across all OCREVUS clinical trials
Shorter 2-hour infusion of OCREVUS was equally well-tolerated in Black, African-American, Hispanic and Latino populations compared with overall study populations across three studies Roche and research partners will be presenting late-breaking data on COVID-19 in treated patients
Basel, 13 October 2021 – Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced new long-term data that reinforce the benefit of early initiation and ongoing treatment of OCREVUS® (ocrelizumab) on disability progression in relapsing multiple sclerosis (RMS) and primary progressive MS (PPMS), as well as safety outcomes for an analysis of shorter 2-hour infusion in minority populations. OCREVUS data from all clinical trials consistently show a favourable benefit-risk profile over eight years. Roche and research partners will also present four late-breaking abstracts to share the latest data regarding COVID-19 and vaccine response in patients treated with OCREVUS. These data are being presented virtually at the 37th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS).
“Many neurologists have had first-hand experience with OCREVUS over eight years in clinical trials and witnessed the consistently favourable efficacy and safety outcomes in RMS and PPMS, especially the reductions in progression to disability when given early in the disease,” said Levi Garraway, M.D., Ph.D. Roche's Chief Medical Officer and Head of Global Product Development. “Additionally, the new safety analysis of the shorter two-hour OCREVUS infusion is encouraging particularly for groups that are often underrepresented in clinical trials. We continue our commitment to diversity and health equity in clinical trial participation and access to treatment.”
Long-term OCREVUS treatment continues to demonstrate sustained reduction in disability progression and suppression of disease activity in people with RMS. Earlier intervention with OCREVUS resulted in a 35% reduction in the risk of patients with RMS needing a walking aid over seven and a half years compared with patients who switched from interferon beta-1a to OCREVUS after the 96-week double-blind period (5.2% vs 7.0%, respectively; 95% CI: 0.65 [0.44–0.97]; p=0.034). The risk was measured by the length of time until a person reached a score on the Expanded Disability Status Scale of 6 or greater (EDSS≥6) that was sustained for at least 48 weeks in a post-hoc analysis. Data also showed that switching from interferon beta-1a to OCREVUS at the start of the OLE period was associated with a rapid and robust reduction in annualised relapse rate (ARR) that was maintained through the 5.5-year OLE period. ARR was 0.2 pre-switch, 0.1 after 1 year of OCREVUS treatment and 0.03 after 5.5 years of OCREVUS treatment in the OLE. OCREVUS continuers maintained a low ARR of 0.03 after 7.5 years of OCREVUS treatment.
For decades, advocacy groups, academia, industry and the broader healthcare community have collectively progressed our understanding of Alzheimer’s disease (AD) – with the hope that every step would help lead to advancements in treating this devastating disease. Given the complexity of Alzheimer’s on a disease and societal level, we anticipate the need for several treatment options to meet the multiple and diverse needs of all people living with Alzheimer’s and their caregivers. As both a pharmaceutical and diagnostics company, we’re in the unique position to contribute to this progress through our ongoing research and studies of investigational medicines for different targets, types and stages of Alzheimer’s and delivering tests to detect, diagnose and monitor the disease. We are hopeful that our ongoing research will lead to scientific progress that could impact millions of people. We are also working in over 70 partnerships with organizations from across the healthcare sector to accelerate progress and improve care for people with AD.
After eight years, outcomes continue to favour early and ongoing treatment with OCREVUS to slow disability progression in people with PPMS. Earlier intervention with OCREVUS resulted in a 29% reduction in 48-week confirmed disability progression (CDP) in patients with PPMS over eight years compared with patients who switched to OCREVUS from placebo after the double-blind period of at least 120 weeks (95% CI: 0.71 [0.57–0.87]; p=0.001). A 24% (95% CI: 0.76 [0.62–0.92]; p=0.005) reduced risk of recurrent 48-week CDP (re-baselining EDSS after onset of CDP event) was seen in patients who were continuously treated with OCREVUS compared with those who switched from placebo. Many people with PPMS eventually transition into a wheelchair; therefore, maintaining the ability to use their hands and arms is important for these patients. Upper limb disability progression, measured by the nine-hole peg test (9-HPT), was also reduced in patients who were continuously treated with OCREVUS compared with those who switched from placebo (95% CI: 0.66 [0.50–0.86] respectively; p=0.002).
New safety data as of November 2020 will be presented, representing 5,688 patients with RMS and PPMS and 21,675 patient-years of exposure to OCREVUS, across all OCREVUS clinical trials. These findings further demonstrate the consistently favourable benefit-risk profile of OCREVUS over eight years.
When treated with a shorter two-hour OCREVUS infusion, the rate and severity of infusion-related reactions in Black, African-American, Hispanic and Latino populations were similar to those reported in the overall patient population in a subgroup analysis of three studies (SaROD, CHORDS and ENSEMBLE PLUS). These patient populations may experience greater disease severity and faster progression, yet are vastly underrepresented in most clinical trials. A shorter infusion time may help reduce the burden on these patient populations and increase their access to treatment.
Patient safety is Roche’s highest priority and we are closely monitoring the evolving COVID-19 situation. We are committed to working closely with the community to better understand the impact of COVID-19 on people who are treated with OCREVUS, and will continue to share new insights with the MS community as they emerge.
Four late-breaking abstracts on COVID-19 in patients treated with OCREVUS, including vaccination response, will be presented by Roche and research partners.
With rapidly growing real-world experience and more than 200,000 people treated globally, OCREVUS is the first and only therapy approved for relapsing MS (RMS; including RRMS and active, or relapsing, secondary progressive MS [SPMS], in addition to clinically isolated syndrome [CIS] in the U.S.) and PPMS. At Roche, we are constantly striving to optimise the care for people with MS and a shorter two-hour OCREVUS infusion time, dosed twice yearly (six-monthly), is now approved for eligible people with RMS or PPMS in the U.S. and European Union (EU).
OCREVUS is approved in 97 countries across North America, South America, the Middle East, Eastern Europe, as well as in Australia, Switzerland, the United Kingdom and the EU.
Multiple sclerosis (MS) is a chronic disease that affects more than 2.8 million people worldwide. MS occurs when the immune system abnormally attacks the insulation and support around nerve cells (myelin sheath) in the central nervous system (brain, spinal cord and optic nerves), causing inflammation and consequent damage. This damage can cause a wide range of symptoms, including muscle weakness, fatigue and difficulty seeing, and may eventually lead to disability. Most people with MS experience their first symptom between 20 and 40 years of age, making the disease the leading cause of non-traumatic disability in younger adults.
People with all forms of MS experience disease progression – permanent loss of nerve cells in the central nervous system and gradual worsening of disability – at the beginning of their disease even if their clinical symptoms aren’t apparent or don’t appear to be getting worse. Delays in diagnosis and treatment can negatively impact people with MS, both in terms of their physical, mental and financial health. An important goal of treating MS is to slow the progression of disability as early as possible.
Relapsing-remitting MS (RRMS) is the most common form of the disease and is characterised by episodes of new or worsening signs or symptoms (relapses) followed by periods of recovery. Approximately 85% of people with MS are initially diagnosed with RRMS. The majority of people who are diagnosed with RRMS will eventually transition to secondary progressive MS (SPMS), in which they experience steadily worsening disability over time. Relapsing forms of MS (RMS) include people with RRMS and people with SPMS who continue to experience relapses. Primary progressive MS (PPMS) is a debilitating form of the disease marked by steadily worsening symptoms but typically without distinct relapses or periods of remission. Approximately 15% of people with MS are diagnosed with the primary progressive form of the disease. Until the FDA approval of OCREVUS, there had been no FDA-approved treatments for PPMS.
OCREVUS is the first and only therapy approved for both RMS (including RRMS and active, or relapsing, SPMS and CIS in the U.S.) and PPMS, with six-month dosing. OCREVUS is a humanised monoclonal antibody designed to target CD20-positive B cells, a specific type of immune cell thought to be a key contributor to myelin (nerve cell insulation and support) and axonal (nerve cell) damage. This nerve cell damage can lead to disability in people with MS. Based on preclinical studies, OCREVUS binds to CD20 cell surface proteins expressed on certain B cells, but not on stem cells or plasma cells, suggesting that important functions of the immune system may be preserved. OCREVUS is administered by intravenous infusion every six months. The initial dose is given as two 300 mg infusions given two weeks apart. Subsequent doses are given as single 600 mg infusions.
Neuroscience is a major focus of research and development at Roche. Our goal is to pursue groundbreaking science to develop new treatments that help improve the lives of people with chronic and potentially devastating diseases.
Roche is investigating more than a dozen medicines for neurological disorders, including multiple sclerosis, Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, Duchenne muscular dystrophy and autism spectrum disorder. Together with our partners, we are committed to pushing the boundaries of scientific understanding to solve some of the most difficult challenges in neuroscience today.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Gantenerumab is an investigational antibody in Phase III development for early Alzheimer's disease (AD)
Gantenerumab is the first and only anti-amyloid antibody being investigated for subcutaneous administration in late-stage trials for the treatment of AD
Ongoing Phase III GRADUATE programme with gantenerumab is anticipated to deliver a comprehensive data set with expected readout in the second half of 2022
Basel, 8 October 2021 – Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that gantenerumab, an anti-amyloid beta antibody developed for subcutaneous administration, has been granted Breakthrough Therapy Designation by the U.S. Food and Drug Administration (FDA) for the treatment of people living with Alzheimer’s disease (AD). This designation is based on data showing that gantenerumab significantly reduced brain amyloid plaque, a pathological hallmark of AD, in the ongoing SCarlet RoAD and Marguerite RoAD open-label extension trials, as well as other studies. Learnings from these studies have been incorporated into the optimised design of two ongoing parallel, global, placebo-controlled and randomised Phase III trials, GRADUATE 1 and 2. The pivotal trials are evaluating gantenerumab in more than 2,000 participants for more than two years and are expected to be completed in the second half of 2022.
“For more than a decade, we’ve been committed to advancing the science of Alzheimer’s as well as our investigational medicine gantenerumab, and we look forward to delivering a comprehensive and robust data set that furthers our collective understanding of this devastating disease,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “This Breakthrough Therapy Designation reinforces our confidence in gantenerumab, which would be the first subcutaneous medicine for the treatment of Alzheimer’s disease with the potential for at-home administration.”
Breakthrough Therapy Designation is designed to accelerate the development and review of medicines intended to treat serious or life-threatening conditions with preliminary evidence that indicates they may demonstrate a substantial improvement over available therapies that have received full FDA approval. This designation for gantenerumab marks the 39th Breakthrough Therapy Designation for Roche’s portfolio of medicines.
AD is a progressive, fatal disease of the brain characterised by a decline in memory, language and other thinking skills, as well as changes in mood and behavior. Biological changes in the brain are believed to start decades before clinical symptoms of AD become evident. Alzheimer's is the most common form of dementia, which currently affects more than 55 million people worldwide, and is projected to reach 78 million by 2030. An enormous and growing public health challenge, it is predicted to cost the global economy a cumulative $20 trillion over the next decade, or US $2.8 trillion per year by 2030. Approximately 10 million people globally are diagnosed with AD each year. Given the medical and societal complexities of AD, several tools and treatment options will likely be required to meet the multiple and diverse needs of people living with the disease.
Roche and Genentech are continuing to explore multiple approaches and molecules that may address key pathways of AD, including beta amyloid and tau, as well as innovative tools designed to more effectively diagnose AD and support clinicians in monitoring disease progression.
Gantenerumab is an investigational IgG1 antibody designed to bind to aggregated forms of beta-amyloid and remove brain amyloid plaques, a pathological hallmark of Alzheimer’s disease (AD). Gantenerumab significantly lowered amyloid plaque in patients with sporadic AD in the SCarlet RoAD and Marguerite RoAD open-label extension studies, and with dominantly inherited AD in the DIAN-TU-001 study. Learnings from these studies have been incorporated into the optimised design of two ongoing parallel, global, placebo-controlled and randomised Phase III trials, GRADUATE 1 and 2.
The pivotal GRADUATE trials are investigating the effect of gantenerumab on amyloid load and downstream biomarkers of disease progression as well as the safety and efficacy of gantenerumab in people with early (prodromal-to-mild) AD. The studies include more than 2,000 patients treated for more than two years in up to 350 study centers in more than 30 countries worldwide. It is evaluating a monthly target dose of 1,020 mg with an optimised titration, aimed at maximizing exposure and minimising dose interruption throughout the study period for better detection of a potential clinical benefit. Both trials are expected to be completed in the second half of 2022.
Other studies evaluating gantenerumab in AD include:
Open RoAD, a rollover open-label study for the former SCarlet RoAD and Marguerite RoAD OLEs to continue to evaluate the safety and tolerability of long-term administration of gantenerumab in participants with AD.
GRADUATION, an open-label study to evaluate the pharmacodynamic (PD) effects of once weekly administration of gantenerumab in participants with early AD. The study design is similar to the GRADUATE studies and includes the option of home administration by a caregiver.
An exploratory OLE of DIAN-TU-001, aiming to build on learnings from DIAN-TU-001 and further investigating the relationship of biomarker changes with cognitive and clinical findings in people with autosomal dominant AD (ADAD).
For decades, advocacy groups, academia, industry and the broader healthcare community have collectively progressed our understanding of Alzheimer’s disease (AD) – with the hope that every step would help lead to advancements in treating this devastating disease. Given the complexity of Alzheimer’s on a disease and societal level, we anticipate the need for several treatment options to meet the multiple and diverse needs of all people living with Alzheimer’s and their caregivers. As both a pharmaceutical and diagnostics company, we’re in the unique position to contribute to this progress through our ongoing research and studies of investigational medicines for different targets, types and stages of Alzheimer’s and delivering tests to detect, diagnose and monitor the disease. We are hopeful that our ongoing research will lead to scientific progress that could impact millions of people. We are also working in over 70 partnerships with organizations from across the healthcare sector to accelerate progress and improve care for people with AD.
Neuroscience is a major focus of research and development at Roche. Our goal is to pursue groundbreaking science to develop new treatments that help improve the lives of people with chronic and potentially devastating diseases.
Roche is investigating more than a dozen medicines for neurological disorders, including multiple sclerosis, Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, Duchenne muscular dystrophy and autism spectrum disorder. Together with our partners, we are committed to pushing the boundaries of scientific understanding to solve some of the most difficult challenges in neuroscience today.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
OCREVUS (ocrelizumab) data to show sustained reduction in disability progression through 8 years for primary progressive multiple sclerosis (PPMS) and 7.5 years for relapsing MS (RMS)
Long-term safety analysis of all clinical trials will reinforce the consistently favourable benefit-risk profile of OCREVUS
ENSPRYNG (satralizumab) data to show efficacy and safety sustained over four years of treatment for people living with neuromyelitis optica spectrum disorder (NMOSD)
Study design for SAkuraBONSAI, a new study on disease activity and progression in ENSPRYNG patients, who are treatment naïve or where prior rituximab (or biosimilar) treatment has failed, will be presented
Basel, 5 October 2021 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that new OCREVUS® (ocrelizumab) and ENSPRYNG® (satralizumab) data will be presented at the 37th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) from 13 - 15 October 2021. These data include 38 abstracts highlighting new longer-term efficacy and safety for both OCREVUS and ENSPRYNG, as well as our ongoing efforts to evaluate the impact of the COVID-19 pandemic for people living with MS. Additional data will show how a deeper scientific understanding of MS and NMOSD in diverse patient populations could help ensure access to treatment.
“The longer-term efficacy and safety data for both OCREVUS and ENSPRYNG reinforce the impact of these treatments - by significantly slowing disease progression in MS and by preventing debilitating relapses in NMOSD, respectively,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “We continue to see that early and ongoing treatment markedly improves outcomes, and we’ll continue to use scientific and real-world insights to improve our understanding and ways to support people living with these neurological disorders.”
Roche will present 27 MS studies, including long-term data that show earlier treatment with OCREVUS continues to impact disability progression up to 8 years in people with primary progressive multiple sclerosis (PPMS) and up to 7.5 years in people with relapsing multiple sclerosis (RMS) in the Phase III open label extension studies. Additionally, updated long-term safety analysis of all clinical trials in patients with RMS and PPMS will reinforce the consistently favourable benefit-risk profile of OCREVUS.
Roche remains committed to addressing health disparities and we believe inclusive research can improve outcomes and derive insights that may address treatment barriers. A subgroup analysis of three studies (SaROD, CHORDS ENSEMBLE PLUS) in Black, African-American, Hispanic and Latino populations treated with a 2-hour OCREVUS infusion will be presented.
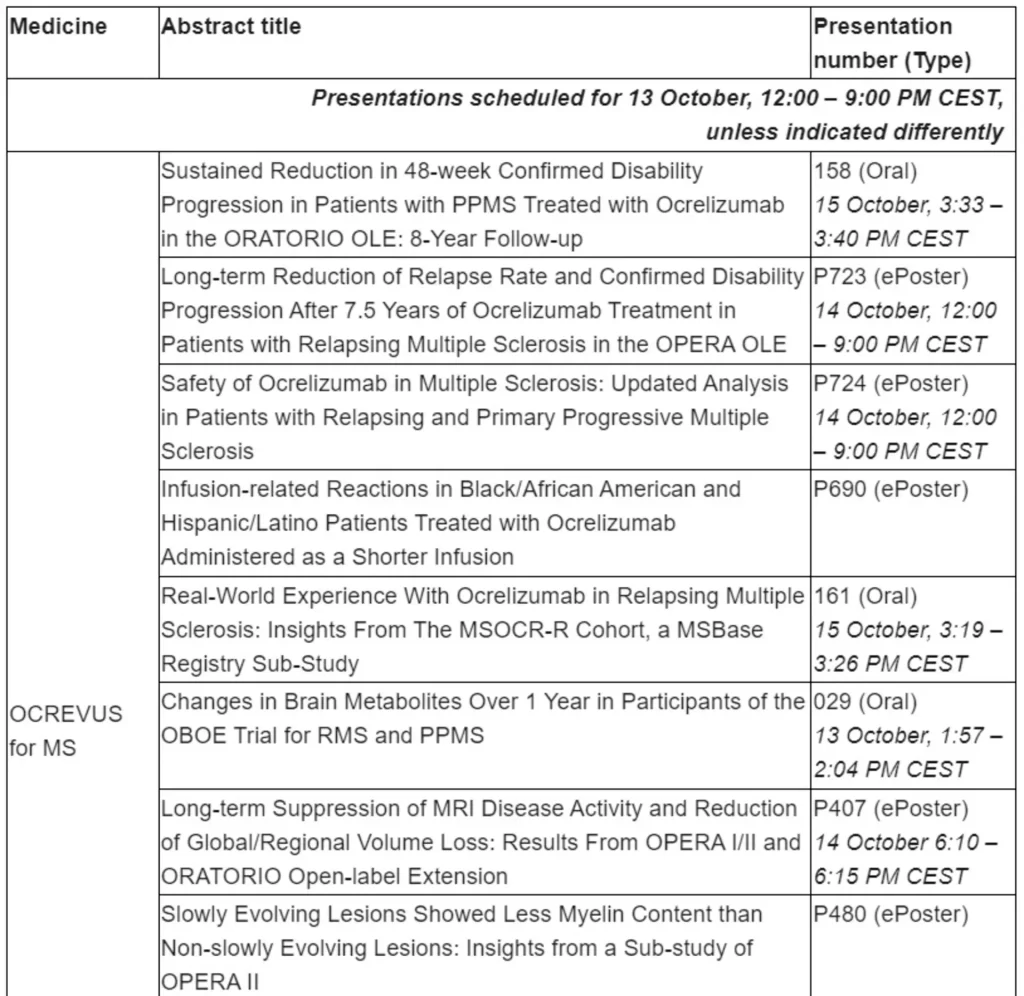
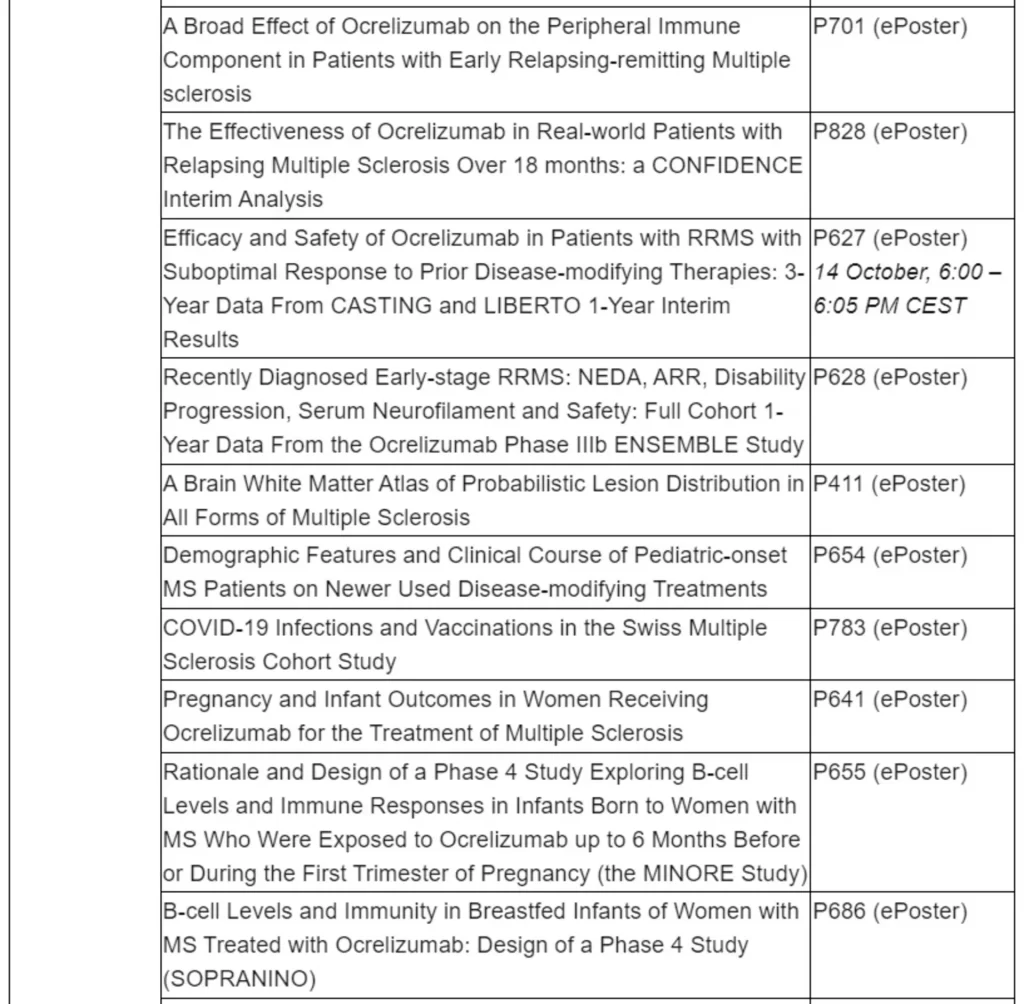
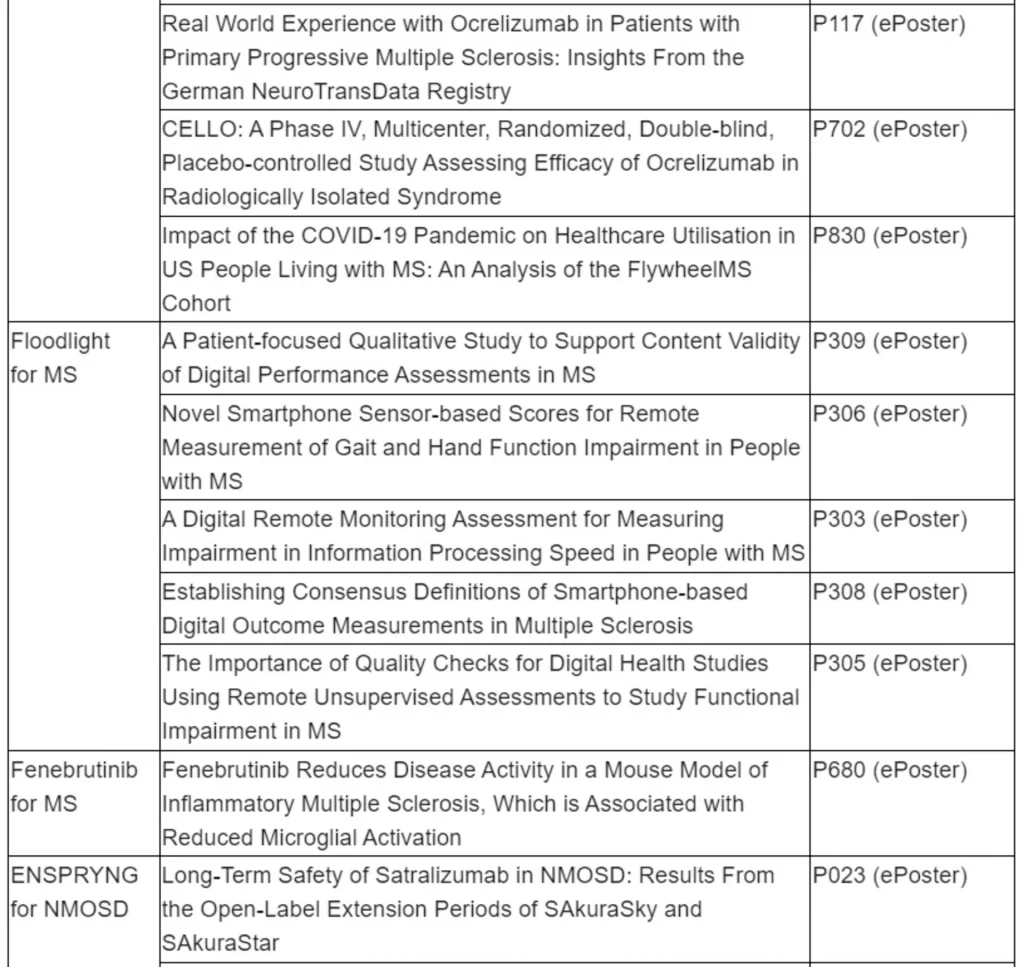
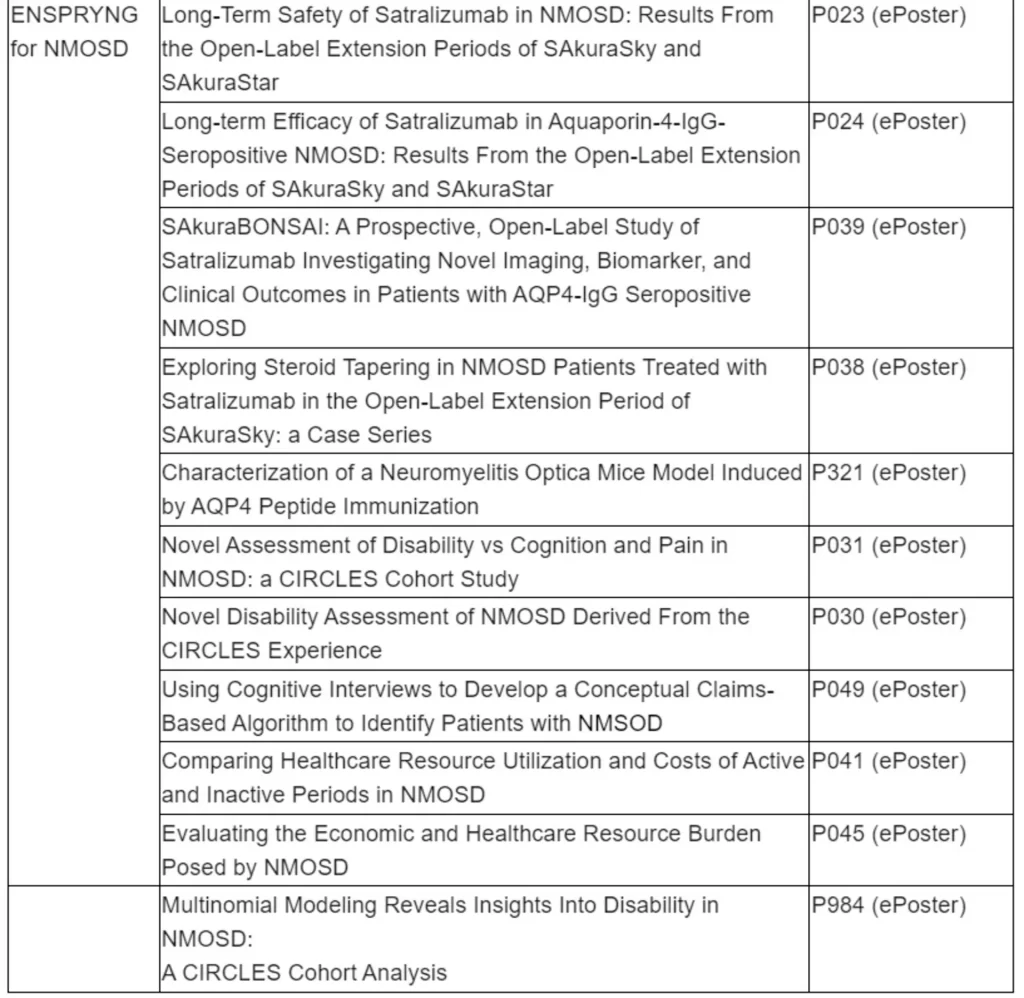
New longer-term results from the SAkuraStar and SAkuraSky OLE studies for ENSPRYNG will show efficacy observed in the pivotal trials is sustained with high proportions of patients remaining free from relapse over four years of treatment. Similarly, safety data from the SAkuraStar and SAkuraSky OLE studies will show the favourable safety profile of ENSPRYNG is sustained with longer-term treatment. ENSPRYNG has been approved in 58 countries globally, including in the U.S. as the first and only subcutaneous treatment for adults with anti-aquaporin-4 antibody (AQP4-IgG) seropositive NMOSD. ENSPRYNG has also been approved for both adults and adolescents in the European Union, Japan, Canada and Switzerland.
Roche is dedicated to increasing scientific understanding of NMOSD and improving care for all people living with the condition. The study design will be presented for SAkuraBONSAI, a prospective, open-label study of ENSPRYNG to generate data to further the understanding of the disease activity and mechanism of action of ENSPRYNG in patients living with AQP4-IgG seropositive NMOSD who are treatment naïve or where prior rituximab (or biosimilar) treatment has failed. Other presentations will examine the development of new tools and techniques to identify patients with NMOSD and assess disability better.
Follow Roche on Twitter via @Roche and keep up to date with ECTRIMS 2021 news and updates by using the hashtag #ECTRIMS2021.
OCREVUS is the first and only therapy approved for both RMS (including RRMS and active, or relapsing, secondary progressive MS [SPMS], in addition to clinically isolated syndrome [CIS] in the U.S.) and PPMS. OCREVUS is a humanised monoclonal antibody designed to target CD20-positive B cells, a specific type of immune cell thought to be a key contributor to myelin (nerve cell insulation and support) and axonal (nerve cell) damage. This nerve cell damage can lead to disability in people with MS. Based on preclinical studies, OCREVUS binds to CD20 cell surface proteins expressed on certain B cells, but not on stem cells or plasma cells, suggesting that important functions of the immune system may be preserved. OCREVUS is administered by intravenous infusion every six months. The initial dose is given as two 300 mg infusions given two weeks apart. Subsequent doses are given as single 600 mg infusions.
ENSPRYNG, which was designed by Chugai, a member of the Roche Group, is a humanised monoclonal antibody that targets interleukin-6 (IL-6) receptor activity. The cytokine IL-6 is believed to be a key driver in NMOSD disease processes, triggering the inflammation cascade and leading to damage and disability. ENSPRYNG was designed using novel recycling antibody technology. When compared to conventional antibodies, ENSPRYNG’s recycling antibody technology enables the medicine to remain in the bloodstream for a longer period of time and bind repeatedly to its target (the IL-6 receptor) - maximally sustaining IL-6 suppression in a chronic disease like NMOSD and enabling subcutaneous dosing every four weeks.
Positive Phase III results for ENSPRYNG, as both monotherapy and in combination with baseline immunosuppressive therapy, suggest that IL-6 inhibition is an effective therapeutic approach for NMOSD. The Phase III clinical development programme for ENSPRYNG includes two studies: SAkuraStar and SAkuraSky.
ENSPRYNG is currently approved in 58 countries, including the United States, Canada, Japan, South Korea and the European Union.
ENSPRYNG has been designated as an orphan drug in the United States, Europe, Japan and Russia. In addition, it was granted Breakthrough Therapy Designation for the treatment of NMOSD by the FDA in December 2018, which is given to treatments that may demonstrate substantial improvement over other available options.
Neuroscience is a major focus of research and development at Roche. Our goal is to pursue groundbreaking science to develop new treatments that help improve the lives of people with chronic and potentially devastating diseases.
Roche is investigating more than a dozen medicines for neurological disorders, including multiple sclerosis, Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, Duchenne muscular dystrophy and autism spectrum disorder. Together with our partners, we are committed to pushing the boundaries of scientific understanding to solve some of the most difficult challenges in neuroscience today.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
