Columvi, in combination with chemotherapy, demonstrated a statistically significant improvement in overall survival for people with relapsed or refractory diffuse large B-cell lymphoma
Data from the STARGLO study will be submitted to health authorities and presented at an upcoming medical meeting
Basel, 15 April 2024 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today the Phase III STARGLO study met its primary endpoint of overall survival. The study demonstrated that people with relapsed or refractory (R/R) diffuse large B-cell lymphoma (DLBCL), who have received at least one prior line of therapy and are not candidates for autologous stem cell transplant, lived longer when treated with Columvi® (glofitamab) in combination with gemcitabine and oxaliplatin (GemOx) versus MabThera®/Rituxan® (rituximab) in combination with GemOx. Safety of the combination appeared consistent with the known safety profiles of the individual medicines. The data will be submitted to health authorities and shared at an upcoming medical meeting.
“People with this aggressive lymphoma facing relapse or progression after initial treatment have limited options – particularly those who are ineligible for stem cell transplant,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Building on Columvi’s established benefits, these data demonstrate the potential of this combination regimen to improve survival outcomes in earlier lines of treatment.”
Columvi was the first fixed-duration bispecific antibody to receive accelerated approval by the U.S. Food and Drug Administration and conditional marketing authorisation from the European Commission to treat people with R/R DLBCL after two or more lines of systemic therapy. These approvals were based on positive results of Columvi as a monotherapy from the pivotal Phase I/II NP30179 study in patients with R/R DLBCL who had previously received two or more prior treatments.
Columvi is a CD20xCD3 T-cell engaging bispecific antibody designed to be off-the-shelf and ready for infusion, so patients can start treatment soon after diagnosis. This is particularly important for patients with highly aggressive disease who are at risk of rapid disease progression. Columvi is given as a fixed-duration treatment, offering people with R/R DLBCL who have failed two or more lines of therapy a treatment end date and the possibility of a treatment-free period, unlike continuous treatments.
The STARGLO study [GO41944; NCT04408638] is a Phase III, multicentre, open-label, randomised study evaluating the efficacy and safety of Columvi® (glofitamab) in combination with gemcitabine plus oxaliplatin (GemOx) versus MabThera®/Rituxan® (rituximab) in combination with GemOx in patients with diffuse large B-cell lymphoma who have received at least one prior line of therapy and who are not candidates for autologous stem cell transplant.1 Outcome measures include overall survival (primary endpoint), progression-free survival, complete response rate, objective response rate, duration of objective response (secondary endpoints), and safety and tolerability.
Columvi is a CD20xCD3 T-cell engaging bispecific antibody designed to target CD3 on the surface of T-cells and CD20 on the surface of B-cells. Columvi was designed with a novel 2:1 structural format. This T-cell engaging bispecific antibody is engineered to have one region that binds to CD3, a protein on T-cells, a type of immune cell, and two regions that bind to CD20, a protein on B-cells, which can be healthy or malignant. This dual-targeting brings the T-cell in close proximity to the B-cell, activating the release of cancer cell-killing proteins from the T-cell. Columvi is part of Roche’s broad and industry-leading CD20xCD3 T-cell-engaging bispecific antibody clinical development programme that also includes Lunsumio® (mosunetuzumab), which aims to provide tailored treatment options that suit the diverse needs, preferences, and experiences of people with blood cancers and healthcare systems. Roche is investigating Columvi as a monotherapy and in combination with other medicines, including Polivy® (polatuzumab vedotin), in earlier lines of treatment for people with B-cell non-Hodgkin lymphomas, including diffuse large B-cell lymphoma and other blood cancers.
DLBCL is the most common form of non-Hodgkin lymphoma (NHL), accounting for about one in three cases of NHL.2 DLBCL is an aggressive (fast-growing) type of NHL.2 While it is generally responsive to treatment in the frontline, as many as 40% of people will relapse or have refractory disease, at which time salvage therapy options are limited and survival is short.3 Improving treatments earlier in the course of the disease and providing much needed alternative options could help to improve long-term outcomes. Approximately 160,000 people worldwide are diagnosed with DLBCL each year.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 25 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera®/Rituxan® (rituximab), Gazyva®/Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie, Hemlibra® (emicizumab), Lunsumio® (mosunetuzumab) and Columvi® (glofitamab). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibody cevostamab, targeting both FcRH5 and CD3, Tecentriq® (atezolizumab), and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The Elecsys® pTau217 plasma biomarker test is being developed as part of an ongoing partnership between Roche and Eli Lilly and Company
Once approved, the test will aid healthcare providers in identifying amyloid pathology, a key feature of Alzheimer’s disease
Roche and Lilly believe the test could play an important role in improving access to early and accurate Alzheimer’s diagnosis
Basel, 11 April 2024 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that its Elecsys pTau217 assay received Breakthrough Device Designation from the U.S. Food and Drug Administration (FDA). This blood test, which is being developed in collaboration with Eli Lilly and Company, will be used to help identify the presence or absence of amyloid pathology in individuals, which can help ensure they are able to receive appropriate care. This may include participation in clinical trials or access to approved disease-modifying therapies. If approved, the test could help rapidly broaden access to a more timely and accurate diagnosis and potentially mitigate the impact of Alzheimer’s disease on people and society.
“The incidence of dementia is growing worldwide, with 75 percent of cases remaining undiagnosed1. Consequently, there is a critical role for Diagnostics to play in addressing this global health challenge,” said Matt Sause, CEO of Roche Diagnostics. “We believe pTau217 is going to be crucial in the diagnosis of Alzheimer’s disease, a condition where Roche Diagnostics is committed to improving the lives of patients worldwide. We plan to leverage our installed base of diagnostic systems, which is the largest in the world, to ensure we are able to create access to this test for those who need it the most.”
“The development of the Elecsys pTau217 plasma assay is another milestone in our collaboration with Roche Diagnostics that will advance the Alzheimer’s diagnostic ecosystem,” said Anne White, executive vice president of Eli Lilly and Company, and president of Lilly Neuroscience. “We’re excited to help meet the growing need for additional diagnostic tools to enable a timely and accurate diagnosis for people with Alzheimer’s disease.”
pTau217, which is a phosphorylated fragment of the protein tau, is a biomarker that has shown the ability in research settings to distinguish Alzheimer’s disease from other neurodegenerative disorders and has shown strong performance relative to other biomarkers2.
As global leaders in Alzheimer’s innovation, Roche and Lilly hope that this collaboration can bring additional speed and scale to testing and diagnosis in this important area of unmet medical need.
Elecsys Phospho-Tau (217P) is intended to be an in-vitro diagnostic immunoassay for the quantitative determination of the protein Phospho-Tau (217P) (pTau217) in human plasma from individuals aged 60 years and older. The test is intended for use as an aid in identifying amyloid pathology, a pathological feature of Alzheimer’s disease.
A positive Elecsys pTau217 result indicates a high likelihood of having a positive amyloid PET/CSF result.
A negative Elecsys pTau217 result indicates a high likelihood of having a negative amyloid PET/ CSF result.
An indeterminate pTau217 result indicates uncertainty on the amyloid PET/CSF result.
The pTau217 result should be used in the diagnostic pathway in conjunction with other clinical information.
With more than two decades of scientific research in Alzheimer’s, Roche is working toward a day when we can detect the disease early and stop its progression to preserve what makes people who they are. Today, the company’s Alzheimer’s portfolio spans investigational medicines for different targets, types and stages of the disease. This includes trontinemab, an innovative Brainshuttle™ anti-amyloid treatment that is specifically engineered to cross the blood-brain barrier. On the diagnostics side, it also includes approved and investigational tools, including digital and blood-based tests and cerebrospinal fluid (CSF) assays, aiming to more effectively detect, diagnose and monitor the disease. Yet the global challenges of Alzheimer’s go well beyond the capabilities of science, and making a meaningful impact requires collaboration both within the Alzheimer’s community and outside of healthcare. We will continue to work together with numerous partners with the hope we can transform millions of lives.
The Breakthrough Devices Program is a voluntary program for certain medical devices that provide for more effective treatment or diagnosis of a life-threatening or irreversibly debilitating disease or condition. This program is designed to expedite the development and review of these medical devices.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The cobas Malaria test is the first FDA-approved molecular test to screen U.S. blood donors for malaria
Malaria is a serious and potentially fatal parasitic infection most commonly transmitted by mosquitoes that can also be spread through blood transfusion
Roche is dedicated to saving patients’ lives through diagnostic solutions that aid in the protection of the global blood supply from infectious diseases
Basel, 26 March 2024 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today the U.S. Food and Drug Administration (FDA) approval of the cobas® Malaria test for use on the cobas® 6800/8800 Systems. This approved test can aid healthcare professionals in reducing potential risks of patient infection from transfused blood products. The cobas Malaria test provides a highly sensitive and specific solution to help ensure that infected blood units are removed from the blood supply.
The cobas Malaria molecular test screens whole blood samples for the five main species of Plasmodium parasites that are known to cause human infection. The potential value of a molecular donor screening test for malaria is to improve both blood safety and availability. The test is intended for use in screening blood, organ and tissue donors.
“As the first FDA-approved blood screening test for malaria, this represents an important step forward in safeguarding the global supply of donated blood," said Matt Sause, CEO of Roche Diagnostics. “The approval of cobas Malaria represents a significant advancement in malaria detection, offering healthcare professionals a reliable tool for donor screening and improving the safety of patients worldwide.”
Globally, a large number of possible donors are excluded from giving blood because of travel to or from residences in malaria-endemic areas. Existing microscopy and serological tests are not sensitive enough to reliably mitigate malaria transfusion risk.
The Roche Blood Safety Solutions offering provides the most comprehensive molecular, serological testing and automation options for donor screening in the US market.
In 2022, nearly half of the world's population was at risk of malaria. While sub-Saharan Africa carries a disproportionately high share of the global malaria burden, the WHO regions of Southeast Asia, the Eastern Mediterranean, the Western Pacific, and the Americas also report significant numbers of cases and deaths. There were an estimated 249 million cases of malaria in 2022, and the estimated number of malaria deaths stood at 608,000. In 2022, the African Region was home to 94% and 95% of malaria cases and deaths, respectively. 2
The test will be available in the United States at the end of Q2 2024. Approval in CE-marked countries is anticipated later this year.
The cobas Malaria test, a qualitative in vitro nucleic acid screening test, allows for direct detection of Plasmodium RNA and DNA in whole blood samples from individual human donors. The test, which can be performed with other routine blood donor screening tests, is designed for use on the cobas 6800/8800 Systems in the U.S. The test is not intended for use to diagnose Plasmodium infection, for use on cord blood samples or for use on cadaveric blood specimens. The test utilises the Roche Whole Blood Collection Tube, which allows for direct draw from the donor and is loaded directly onto the cobas 6800/8800 Systems for increased workflow efficiency.
Since 2014, the cobas 6800 and cobas 8800 Systems have established the new standard for routine molecular testing by delivering fully integrated, automated solutions that serve the areas of donor screening, infectious disease, sexual health, transplant, respiratory and antimicrobial stewardship. The current molecular donor screening assay menu includes cobas® MPX, cobas® DPX, cobas® HEV, cobas® WNV, cobas® CHIKV/DENV, cobas® Zika and cobas® Babesia (not all tests are available in the U.S.).
Malaria is a serious and sometimes fatal disease caused by a parasite that commonly infects a certain type of mosquito which feeds on humans. People who get malaria typically experience high fever, shaking chills and flu-like symptoms. The disease can also be transmitted through blood transfusion, organ transplant or the shared use of needles or syringes contaminated with blood.1
Malaria is preventable and curable. There are 5 parasite species that cause malaria in humans, and 2 of these species – Plasmodium falciparum and Plasmodium vivax – pose the greatest threat.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Detailed results from the NIH-sponsored Phase III OUtMATCH study showed treatment with Xolair increased the amount of peanuts, tree nuts, egg, milk and wheat that people as young as 1 year consumed without an allergic reaction
The U.S. FDA recently approved Xolair as the first and only medicine for children and adults with one or more food allergies
Allergic reactions can be life-threatening and it is estimated that food-related anaphylaxis results in 30,000 medical events treated in emergency rooms in the U.S. each year
Basel, 25 February 2024 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today data from Stage 1 of the National Institutes of Health (NIH)-sponsored pivotal Phase III OUtMATCH study evaluating the efficacy and safety of Xolair® (omalizumab) in patients allergic to peanuts and at least two other common foods were published in the New England Journal of Medicine (NEJM) and featured in a late-breaking symposium at the 2024 American Academy of Allergy, Asthma & Immunology (AAAAI) Annual Meeting. The study showed treatment with Xolair increased the amount of peanuts, milk, egg, wheat and tree nuts (cashew, hazelnut and walnut) that it took to cause moderate to severe allergic reactions in multi-food allergic people as young as 1 year. Safety findings were consistent with the known safety profile of Xolair across its approved indications and in previous clinical trials.4 The U.S. Food and Drug Administration (FDA) recently approved the expanded use of Xolair in children and adults with IgE-mediated food allergies based on the OUtMATCH data.
“Over the past 35 years, I have seen how debilitating food allergies can be for patients and their loved ones, as they are consumed by the fear of accidental exposure,” said Robert Wood, M.D., director of the Eudowood Division of Allergy, Immunology and Rheumatology at Johns Hopkins Children's Center, and principal investigator of the OUtMATCH study. “While allergic reactions to exposures are common and often severe, there have been limited treatment advancements for food allergy. The results of the OUtMATCH study showed that anti-IgE therapy could significantly reduce the occurrence of allergic reactions across multiple foods in the event of an accidental exposure.”
“Living with food allergies has a profound impact on patients and their families, causing significant stress and requiring constant vigilance,” said R. Sharon Chinthrajah, M.D., associate professor of medicine, Stanford School of Medicine, Sean N. Parker Center for Allergy and Asthma Research and OUtMATCH co-lead study investigator. “The OUtMATCH study demonstrated that anti-IgE therapy increased most patients’ threshold for an allergic reaction. This presents an important new treatment option for patients and families in its potential to reduce the risk of allergic reactions from accidental exposures they may face in day-to-day life.”
One hundred eighty patients ages 1 to 55 years old entered Stage 1 of the OUtMATCH study unable to tolerate up to 100 mg of peanut protein (equivalent to about one third of a peanut), and up to 300 mg of at least two other food proteins among milk, egg, cashew, walnut, hazelnut and wheat. After 16 to 20 weeks of treatment with Xolair or placebo, each participant completed four separate blinded food challenges (including a placebo ingredient) to assess patients’ ability to consume a single dose of at least 600 mg of peanut protein (primary endpoint), and a single dose of at least 1,000 mg of milk, egg, wheat, cashew, hazelnut or walnut protein (secondary endpoints) without experiencing moderate to severe allergic reactions.
Results showed that, compared to placebo, a statistically significant (p<0.001) higher proportion of patients receiving Xolair were able to consume at least 600 mg of peanut protein and at least 1,000 mg of milk, egg and cashew protein without experiencing moderate to severe allergic reactions. Additionally, compared to placebo, a higher proportion of patients receiving Xolair were also able to consume at least 1,000 mg of walnut, hazelnut and wheat protein without experiencing moderate to severe allergic reactions. Detailed results are included in the table below.
Percentage of Patients Successfully Consuming Predefined Threshold Dose of Seven Foods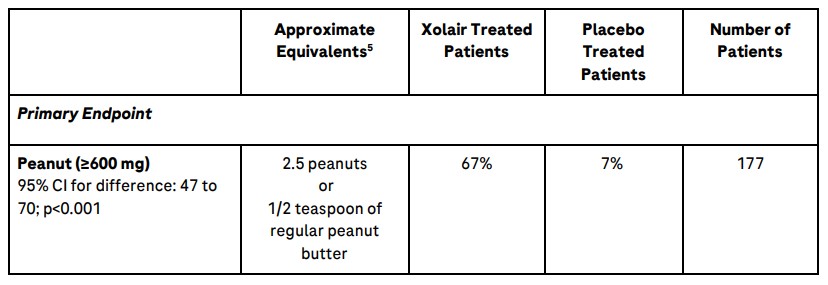
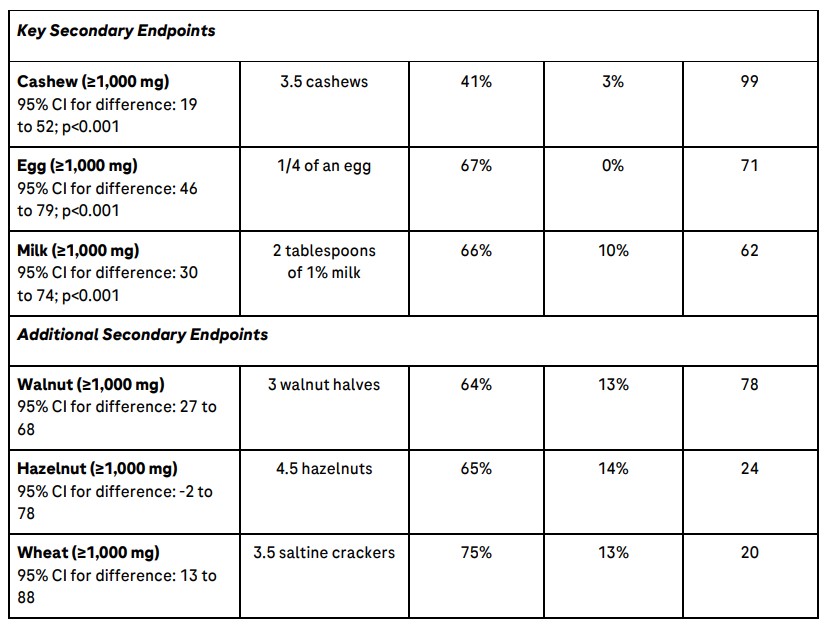
Additionally, out of three adult OUtMATCH participants, two completed Stage 1 of the study; the adult who received Xolair met the primary endpoint compared to the placebo-enrolled adult patient.
Adverse event rates in the study were similar between Xolair and placebo. The most common adverse event in Xolair-treated children and adolescents was injection site reaction (9%).
“The Phase III OUtMATCH results represent a major advance and reinforce our commitment to addressing critical gaps in care for conditions like food allergy,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Now recently FDA-approved, Xolair provides a new way to help manage food allergies for children and adults who live with the constant fear of life-threatening allergic reactions.”
While efficacy cannot be established from uncontrolled, open-label studies, for 38 children who continued Xolair for 24-28 weeks in an open-label extension, the percentage of patients who were able to consume 600 mg or more of peanut protein and 1,000 mg or more of egg, milk and/or cashew protein without moderate to severe dose-limiting symptoms was maintained.
About 3.4 million children and 13.6 million adults in the U.S. will have been diagnosed with IgE-mediated food allergies, based on estimates for 2024.1,2 Food allergy prevalence has been on the rise for the past 20 years.6 There are 160 different foods that cause IgE-mediated food allergy.7 Allergic reactions can range from mild to moderate, including hives and swelling, to severe and life-threatening, such as anaphylaxis.3 More than 40% of children and more than half of adults with food allergies have experienced a severe reaction at least once, and it is estimated that food-related anaphylaxis results in 30,000 medical events treated in emergency rooms in the U.S. each year.1-3
On 16 February, 2024, the FDA approved Xolair for the reduction of allergic reactions, including anaphylaxis, that may occur with accidental exposure to one or more foods in adult and paediatric patients aged 1 year and older with IgE-mediated food allergy. People taking Xolair for food allergies should continue to avoid all foods they are allergic to (commonly referred to as “food allergen avoidance”). Xolair should not be used for the emergency treatment of any allergic reactions, including anaphylaxis. Xolair is the first and only FDA-approved medicine to reduce allergic reactions in people with one or more food allergies.
In the U.S., Genentech, a member of the Roche Group, and Novartis Pharmaceuticals Corporation work together to develop and co-promote Xolair.
The Omalizumab as Monotherapy and as Adjunct Therapy to Multi-Allergen Oral Immunotherapy in Food Allergic Children and Adults (OUtMATCH; NCT03881696) study is an NIH-sponsored, three-stage, multicentre, randomised, double-blind, placebo-controlled study evaluating Xolair safety and efficacy in patients aged 1 to 55 years who are allergic to peanuts and at least two other common foods.
Stage 1 included 180 patients (177 children and adolescents; 3 adults) who were randomly assigned to receive placebo or Xolair injections either every two weeks or every four weeks for 16 to 20 weeks. The Xolair dose and dosing interval were determined by total serum IgE level and body weight at baseline.
After 16 to 20 weeks of treatment with Xolair or placebo, each participant completed four separate blinded food challenges where they were given gradually increasing amounts of peanut protein, two other food proteins they were allergic to, and a placebo ingredient. The food challenges were conducted in a carefully controlled setting with investigators looking for signs and symptoms of allergic reaction to assess patients’ ability to consume a single dose of at least 600 mg of peanut protein (primary endpoint), and a single dose of at least 1,000 mg of milk, egg, wheat, cashew, hazelnut or walnut protein (secondary endpoints) without experiencing dose-limiting symptoms, which were defined as moderate to severe allergic reactions, including skin, respiratory or gastrointestinal symptoms.
The OUtMATCH study is being sponsored and funded by the National Institute of Allergy and Infectious Diseases (NIAID), part of the NIH, and conducted by the NIAID-funded Consortium for Food Allergy Research (CoFAR) at 10 clinical sites across the U.S. led by Johns Hopkins Children's Center and co-led by Stanford School of Medicine. The study is also supported by Genentech and Novartis Pharmaceuticals Corporation.
Xolair is the only approved antibody designed to target and block immunoglobulin E (IgE). By reducing free IgE, down-regulating high-affinity IgE receptors and limiting mast cell degranulation, Xolair minimises the release of mediators throughout the allergic inflammatory cascade.
The Roche Group’s immunology medicines include: Actemra®/RoActemra® (tocilizumab) for rheumatoid arthritis, polyarticular juvenile idiopathic arthritis (pJIA), systemic juvenile idiopathic arthritis (sJIA) and giant cell arteritis (GCA) and for the treatment of severe or life-threatening chimeric antigen receptor (CAR) T cell-induced cytokine release syndrome (CRS); Rituxan®/MabThera® (rituximab) for rheumatoid arthritis granulomatosis with polyangiitis and microscopic polyangiitis and for pemphigus vulgaris (PV); Xolair® (omalizumab) for allergic asthma and chronic idiopathic urticaria (CIU); Pulmozyme® (dornase alfa) for cystic fibrosis; and Esbriet® (pirfenidone) for idiopathic pulmonary fibrosis (IPF). Roche has more than 15 investigational medicines in clinical development for immunological diseases that include asthma, autoimmune diseases, rheumatoid arthritis, ulcerative colitis and Crohn's disease.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Subcutaneous (SC) injection offers the potential for a faster, more convenient alternative to intravenous (IV) infusion and is preferred by cancer patients, nurses and physicians
Tecentriq SC reduces treatment time by approximately 80%, compared with standard IV infusion
Roche is working closely with national health systems in Europe to ensure patients can access Tecentriq SC as quickly as possible
Basel, 16 January 2024 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that the European Commission has granted marketing authorisation for Tecentriq® SC (atezolizumab), the European Union (EU)’s first PD-(L)1 cancer immunotherapy for subcutaneous (under the skin) injection. Last year, more than 38,000 people in the EU received Tecentriq to treat different types of lung, liver, bladder and breast cancer.7 Until now, Tecentriq has been given directly into patients’ veins by IV infusion which takes approximately 30-60 minutes.6 The new subcutaneous injection will cut treatment time to approximately seven minutes, with most injections taking between four and eight minutes.6 The marketing authorisation applies to all approved indications of Tecentriq IV.7
“We are pleased to introduce the first subcutaneous PD-L1 cancer immunotherapy in Europe,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Giving Tecentriq subcutaneously provides more flexibility to patients, while also helping to free up resources in constrained healthcare systems.”
“Ensuring the best possible quality of life is crucial for people living with cancer,” said Dr. Enriqueta Felip, Head of the Thoracic Cancer Unit of Vall d'Hebron Hospital, Spain. “The availability of a subcutaneous cancer immunotherapy option that can minimise the time receiving treatment and even allow for treatment outside of a hospital will undoubtedly make a significant difference to patients and their loved ones.”
The approval is based on pivotal data from the Phase IB/III IMscin001 study, which showed comparable levels of Tecentriq in the blood, when administered subcutaneously, and a safety and efficacy profile consistent with the IV formulation.6,8 90% of healthcare professionals who were surveyed as part of the study agreed that the SC formulation is easy to administer and 75% said it could save time for healthcare teams compared with the IV formulation.6
In addition to offering shorter treatment time, Tecentriq SC may be administered by a healthcare professional outside of the hospital, in a community care setting or at a patient's home, depending on national regulations and health systems. Roche is in discussion with several providers in Europe to include Tecentriq SC in cancer homecare initiatives where possible.
IMscin001 is a Phase IB/III, global, multicentre, randomised study evaluating the pharmacokinetics, safety and efficacy of Tecentriq SC, compared with Tecentriq IV, in patients with previously treated locally advanced or metastatic non-small cell lung cancer (NSCLC) for whom prior platinum therapy has failed. The study enrolled 371 patients.
The study met its primary endpoints, demonstrating comparable levels of Tecentriq in the blood during a given dosing interval on the basis of established pharmacokinetic measurements; observed serum Ctrough and model-predicted area under the curve. Efficacy, as measured by the overall response rate, progression-free survival, overall survival and duration of response, was similar between the SC and IV treatment arms and consistent with the known profile of Tecentriq IV. The safety profile of Tecentriq SC was also consistent with that of Tecentriq IV.
Tecentriq® (atezolizumab) in Tecentriq SC is the same monoclonal antibody as in Tecentriq IV. It has been designed to bind with a protein called programmed death ligand-1 (PD-L1), which is expressed on tumour cells and tumour-infiltrating immune cells, blocking its interactions with both PD-1 and B7.1 receptors. By inhibiting PD-L1, Tecentriq may enable the activation of T-cells. Tecentriq is a cancer immunotherapy that has the potential to be used across a broad range of cancers.
Tecentriq SC combines Tecentriq with Halozyme Therapeutics’ Enhanze® drug delivery technology. The Enhanze drug delivery technology is based on a proprietary recombinant human hyaluronidase PH20 (rHuPH20), an enzyme that locally and temporarily degrades hyaluronan – a glycosaminoglycan or chain of natural sugars in the body – in the subcutaneous space. This increases the permeability of the tissue under the skin, allowing space for Tecentriq to enter, enabling it to be rapidly dispersed and absorbed into the bloodstream.
Tecentriq SC was first approved in Great Britain in August 2023. The approved indications for Tecentriq SC mirror those of Tecentriq IV.
The standard IV formulation of Tecentriq is approved for some of the most aggressive and difficult-to-treat forms of cancer. Tecentriq IV was the first cancer immunotherapy approved for the treatment of a certain type of early-stage (adjuvant) NSCLC, small cell lung cancer (SCLC) and hepatocellular carcinoma (HCC). Tecentriq IV is also approved in countries around the world, either alone or in combination with targeted therapies and/or chemotherapies, for various forms of metastatic NSCLC, certain types of metastatic urothelial cancer (mUC), PD-L1-positive metastatic triple-negative breast cancer (TNBC), BRAF V600 mutation-positive advanced melanoma and alveolar soft part sarcoma (ASPS).
To learn more about Roche’s scientific-led approach to cancer immunotherapy, please follow this link: https://www.roche.com/solutions/focus-areas/oncology/cancer-immunotherapy
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
With Lunsumio, people with heavily pre-treated follicular lymphoma may experience remission with a chemotherapy-free, fixed-duration treatment that can be accessed in an outpatient setting
Results from the pivotal phase II GO29781 study demonstrated that 80% of patients who received at least two prior therapies achieved durable response rates, with 60% experiencing complete remission
Lunsumio is now the first CD20xCD3 T-cell engaging bispecific antibody approved by the FDA to treat the most common slow-growing form of non-Hodgkin lymphoma, follicular lymphoma
Basel, 23 December 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the U.S. Food and Drug Administration (FDA) has approved Lunsumio® (mosunetuzumab-axgb) for the treatment of adult patients with relapsed or refractory (R/R) follicular lymphoma (FL) after two or more lines of systemic therapy. This indication is approved under accelerated approval based on response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. Lunsumio, a CD20xCD3 T-cell engaging bispecific antibody, represents a new class of fixed-duration cancer immunotherapy, which is off-the-shelf and readily available, so that patients do not have to wait to start treatment. Lunsumio will be available in the United States in the coming weeks.
“This approval is a significant milestone for people with relapsed or refractory follicular lymphoma, who have had limited treatment options until now,” said Elizabeth Budde, M.D., Ph.D., Haematologic Oncologist and Associate Professor, City of Hope Division of Lymphoma, Department of Hematology & Hematopoietic Cell Transplantation, and Lunsumio clinical trial investigator. “As a first-in-class T-cell engaging bispecific antibody that can be initiated in an outpatient setting, Lunsumio’s high response rates and fixed-duration could change the way advanced follicular lymphoma is treated.”
“Despite treatment advances, follicular lymphoma remains incurable and relapse is common, with outcomes worsening following each consecutive treatment,” said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. “Lunsumio represents our first approved T-cell engaging bispecific antibody and builds on our legacy of more than 20 years of innovation in blood cancer.”
The FDA approval is based on positive results from the phase II GO29781 study of Lunsumio in people with heavily pre-treated FL, including those who were at high risk of disease progression or whose disease was refractory to prior therapies. Results from the study showed high and durable response rates. An objective response was seen in 80% (72/90 [95% confidence interval (CI): 70-88]) of patients treated with Lunsumio, with a majority maintaining responses for at least 18 months (57% [95% CI: 44-70]). The objective response rate is the combination of complete response (CR) rate (a disappearance of all signs and symptoms of cancer) and partial response rate (a decrease in the amount of cancer in the body). The median duration of response among those who responded was almost two years (22.8 months [95% CI: 10-not reached]). A CR was achieved in 60% of patients (54/90 [95% CI: 49-70]). Among 218 patients with haematologic malignancies who received Lunsumio at the recommended dose, the most common adverse event (AE) was cytokine release syndrome (CRS; 39%), which can be severe and life-threatening. The median duration of CRS events was three days (range: 1-29). Other common AEs (≥20%) included fatigue, rash, pyrexia and headache.
Lunsumio is administered as an intravenous infusion for a fixed-duration, which allows for time off therapy, and can be infused in an outpatient setting. Hospitalisation may be needed to manage select AEs, should be considered for subsequent infusions following a Grade 2 CRS event, and is recommended for subsequent infusions following a Grade 3 CRS event.
Lunsumio was developed based on the Roche Group's broad expertise in creating bispecific antibodies. Lunsumio is designed to address the diverse needs of people with blood cancer, physicians, and practice settings, and is part of the company’s robust bispecific antibody clinical programme in lymphoma. Lunsumio is being further investigated as a subcutaneous formulation (i.e., administered under the skin) and in phase III studies that will expand the understanding of its impact in earlier lines of treatment in people with non-Hodgkin lymphoma.
The GO29781 study [NCT02500407] is a phase II, multicentre, open-label, dose-escalation and expansion study evaluating the safety, efficacy and pharmacokinetics of Lunsumio® (mosunetuzumab-axgb) in people with relapsed or refractory B-cell non-Hodgkin lymphoma. Outcome measures include complete response rate (best response) by independent review facility (primary endpoint), objective response rate, duration of response, progression-free survival, safety, and tolerability (secondary endpoints).
Follicular lymphoma (FL) is the most common slow-growing (indolent) form of non-Hodgkin lymphoma, accounting for about one in five cases.1 It typically responds well to treatment but is often characterised by periods of remission and relapse. The disease typically becomes harder to treat each time a patient relapses, and early progression can be associated with poor long-term prognosis. It is estimated that, in the United States, approximately 13,000 new cases of FL will be diagnosed in 2022 and more than 100,000 people are diagnosed with FL each year worldwide.
Lunsumio is a first-in-class CD20xCD3 T-cell engaging bispecific antibody designed to target CD20 on the surface of B-cells and CD3 on the surface of T-cells. This dual targeting activates and redirects a patient’s existing T-cells to engage and eliminate target B-cells by releasing cytotoxic proteins into the B-cells. A robust clinical development programme for Lunsumio is ongoing, investigating the molecule as a monotherapy and in combination with other medicines, for the treatment of people with B-cell non-Hodgkin lymphomas, including follicular lymphoma and diffuse large B-cell lymphoma, and other blood cancers.
Roche has been developing medicines for people with malignant and non-malignant blood diseases for more than 20 years; our experience and knowledge in this therapeutic area runs deep. Today, we are investing more than ever in our effort to bring innovative treatment options to patients across a wide range of haematologic diseases. Our approved medicines include MabThera®/Rituxan® (rituximab), Gazyva®/Gazyvaro® (obinutuzumab), Polivy® (polatuzumab vedotin), Venclexta®/Venclyxto® (venetoclax) in collaboration with AbbVie, Hemlibra® (emicizumab) and Lunsumio® (mosunetuzumab-axgb). Our pipeline of investigational haematology medicines includes T-cell engaging bispecific antibodies, glofitamab, targeting both CD20 and CD3, and cevostamab, targeting both FcRH5 and CD3; Tecentriq® (atezolizumab), a monoclonal antibody designed to bind with PD-L1 and crovalimab, an anti-C5 antibody engineered to optimise complement inhibition. Our scientific expertise, combined with the breadth of our portfolio and pipeline, also provides a unique opportunity to develop combination regimens that aim to improve the lives of patients even further.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Actemra is the first FDA-approved monoclonal antibody to treat COVID-19
Since the beginning of the pandemic, more than one million people hospitalised with COVID-19 have been treated with Actemra worldwide
Actemra is approved for this use in more than 30 countries for the treatment of COVID-19
Basel, 21 December 2022 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that the U.S. Food and Drug Administration (FDA) has approved Actemra® (tocilizumab) intravenous (IV) for the treatment of COVID-19 in hospitalised adult patients who are receiving systemic corticosteroids and require supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO). Actemra is the first FDA-approved monoclonal antibody to treat COVID-19 and is recommended for use as a single 60-minute IV infusion.
“With new variants emerging, FDA-approved treatments including Actemra remain essential to the continued fight against COVID-19,” said Levi Garraway, M.D., Ph.D., Chief Medical Officer and Head of Global Product Development. “Actemra is the first FDA-approved monoclonal antibody for treating patients with severe COVID-19, providing an important option for hospitalised patients and their healthcare providers who continue to be on the frontlines treating COVID-19.”
Four randomised, controlled studies evaluated Actemra for the treatment of COVID-19 in more than 5,500 hospitalised patients. Altogether, the results of these four studies (the University of Oxford-led RECOVERY trial, along with the Roche-sponsored global trials, EMPACTA, COVACTA and REMDACTA) showed that Actemra may improve outcomes in patients receiving corticosteroids and requiring supplemental oxygen or breathing support. The FDA approval is based on the results from the RECOVERY trial, as well as the EMPACTA trial, the first global, Phase III study in COVID-19 to focus on patients from underrepresented racial and ethnic groups. No new warnings and precautions related to Actemra in COVID-19 studies have been identified.
The FDA approval follows the FDA’s Emergency Use Authorization (EUA) for Actemra in hospitalised adults and children (ages 2 and older) with COVID-19, which was granted in June 2021. The use of Actemra to treat hospitalised people ages 2 to less than 18 years old is not FDA approved, however the EUA for this age group currently remains in place after the FDA approval for hospitalised adult patients.
More than one million people hospitalised with COVID-19 have been treated with Actemra worldwide, since the beginning of the pandemic. Around the world, Actemra is approved for use in more than 30 countries for patients hospitalised with severe COVID-19. In the United States, this is the seventh FDA approved indication for Actemra since the medicine was launched in 2010.
Roche stands together with society, governments, healthcare providers and all those working towards the common goal of overcoming the COVID-19 pandemic.
Actemra®/RoActemra® is approved for use in multiple territories including the United States, European Union, Japan, the United Kingdom, New Zealand, Russia and Brazil, provisionally approved in Australia, and authorised for emergency use in Ghana, Mexico and Korea for defined patients hospitalised with severe or critical COVID-19. It has also been recommended and prequalified by the World Health Organization.
Actemra®/RoActemra® was the first humanised interleukin-6 (IL-6) receptor antagonist approved for the treatment of adult patients with moderately to severely active rheumatoid arthritis (RA) who have used one or more disease-modifying antirheumatic drugs (DMARDs), such as methotrexate (MTX), that did not provide enough relief. The extensive Actemra RA IV clinical development program included five Phase III clinical studies and enrolled more than 4,000 people with RA in 41 countries. The Actemra RA subcutaneous clinical development program included two Phase III clinical studies and enrolled more than 1,800 people with RA in 33 countries. Actemra subcutaneous injection is also approved for the treatment of adult patients with giant cell arteritis (GCA), for the treatment of patients two years of age and older with active polyarticular juvenile idiopathic arthritis (PJIA) or active systemic juvenile idiopathic arthritis (SJIA), and for slowing the rate of decline in pulmonary function in adult patients with systemic sclerosis-associated interstitial lung disease (SSc-ILD). In addition, Actemra is also approved in the IV formulation for patients two years of age and older with active PJIA, SJIA, GCA or CAR T cell-induced cytokine release syndrome (CRS). Actemra is not approved for subcutaneous use in people with CRS. Actemra IV is approved for the treatment of COVID-19 in hospitalised adult patients who are receiving systemic corticosteroids and require supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO). It is not known if Actemra is safe and effective in children with PJIA, SJIA or CRS under two years of age or in children with conditions other than PJIA, SJIA or CRS.
As a leading healthcare company, we are doing all we can to support countries in their fight against COVID-19 and minimising its impact. That is why we are working with governments, policy makers, healthcare professionals and others to help contain the COVID-19 pandemic and make sure patients continue to receive the tests, treatment and care they need.
The pandemic has profoundly raised awareness of the role diagnostics play in COVID-19 diagnosis, treatment development and disease management. Roche has developed and launched more than 20 COVID-19 diagnostics solutions, including polymerase chain reaction (PCR) and rapid antigen and antibody tests. Our solutions serve the entire diagnostic continuum, from high-throughput laboratories to point-of-care and home self-testing, and cover all currently known variants. To help meet global demand, we have supplied more than 1.5 billion tests for COVID-19 since March 2020.
Roche continues to evaluate its existing therapeutic portfolio and is researching future options to help benefit patients with COVID-19. Our IL-6 inhibitor Actemra®/RoActemra® (tocilizumab) has been approved for patients hospitalised with severe COVID-19 in more than 30 countries including the European Union and the United States. The World Health Organization has prequalified Actemra for use in patients with severe COVID-19, facilitating its availability in low- and middle-income countries. In addition, we have been improving access to Actemra by introducing an international differentiated pricing strategy, providing the medicine at cost for use in low- and middle-income countries and non asserting patents in these regions during the pandemic.
We have also been partnering with Regeneron to jointly develop the antibody combination Ronapreve™ (casirivimab and imdevimab, known as REGEN-COV™ in the US). It has been approved in multiple territories including the European Union, Japan, and Switzerland and authorised for emergency or temporary pandemic use in many countries including the US. We are constantly monitoring and assessing Ronapreve’s neutralising activity against emerging variants of concern and entered a dialogue with regulators and industry representatives to discuss the efficacy of monoclonal antibodies in the context of rapidly evolving SARS-CoV-2 variants.
Our utmost goal remains to be a trusted partner who acts with urgency to save and improve the lives of patients with COVID-19 and to reduce its burden on society. For more information please visit our COVID-19 response page.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
The ranking acknowledges Roche’s commitment to sustainability as an integral part of its business strategy
Roche performed particularly well in innovation management, access to healthcare, greenhouse gas emissions, water management, resource efficiency and circularity, labour practice indicators and human rights
This marks the 15th consecutive year that Roche has maintained its leading ranking
Basel, 15 December 2023 - Roche (SIX: RO, ROG; OTCQX: RHHBY) has again been recognised as one of the most sustainable companies in the Pharmaceuticals index of the Dow Jones Sustainability Indices (DJSI). Roche ranked third, Chugai Pharmaceuticals, a member of the Roche Group, ranked second.
“We are extremely proud that our deep commitment to sustainability in all our business practices has been recognised again,” said Roche CEO Thomas Schinecker. “It is a fantastic achievement that Chugai and Roche came in second and third, respectively. This double recognition reflects the innovation the Roche Group brings to society, and our commitment to continually embed sustainable practices in our strategy and culture.”
For the past 15 consecutive years, Roche has maintained its leading ranking, which is based on an in-depth analysis of economic, social and environmental performance. The DJSI family of indices serves as a benchmark for investors who integrate sustainability considerations into their portfolios.
For over 125 years, sustainability has been an integral part of Roche’s business. Roche follows a holistic approach to sustainability and integrates the three dimensions of sustainability:
Society: how we contribute to a better tomorrow for all
Environment: how we minimise our impact on nature
Economy: how we invest in medical advances, create jobs and ensure livelihoods
Roche makes its biggest contribution to society by improving healthcare provision. It does so by developing innovative treatments across major disease areas and medical assays, digital solutions and services helping people live healthier lives.
Roche is aware that this cannot be done alone. The key to creating sustainable value and growth lies in partnering with stakeholders and engaging in open, constructive dialogue. By doing so, Roche generates value for society and achieves sustainable economic growth for the company. We apply this approach to reducing our overall environmental impacts across our business, well beyond decarbonisation, setting long-term goals, measuring our environmental impact and having an end-to-end focus on our products.
Further information on our activities across sustainability areas can be found here: www.roche.com/sustainability
This global ranking is published annually by the S&P Dow Jones Indices (S&P DJI) and measures the performance of the world’s sustainability leaders. S&P DJI invites publicly traded companies to participate. Companies are selected for the indices according to a systematic Corporate Sustainability Assessment. Only firms that lead their industries based on this assessment are included in the indices. This is determined by a comprehensive assessment of long-term economic, environmental and social criteria that take account of general as well as industry-specific sustainability trends.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Phase III (INAVO120) results show that inavolisib in combination with palbociclib and fulvestrant significantly improved progression-free survival in the first-line setting
PIK3CA mutations, found in approximately 40% of HR-positive breast cancers, are linked to tumour growth, disease progression, and treatment resistance
Data will be shared with health authorities and presented at an upcoming medical meeting
Basel, 5 December 2023 - Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today positive results from the Phase III INAVO120 study of the investigational therapy, inavolisib, in combination with palbociclib (Ibrance®) and fulvestrant as a potential first-line treatment option for people with PIK3CA-mutated, hormone receptor (HR)-positive, HER2-negative, endocrine-resistant, locally advanced or metastatic breast cancer. The study met its primary endpoint of progression-free survival (PFS), demonstrating a statistically significant and clinically meaningful improvement compared to palbociclib and fulvestrant alone. Overall survival data were immature at this time, but a clear positive trend has been observed. Follow-up will continue to the next analysis.
"These pivotal study results for this inavolisib combination could represent a transformative medical advance for people with PIK3CA-mutated HR-positive breast cancer," said Levi Garraway, M.D., Ph.D., Roche’s Chief Medical Officer and Head of Global Product Development. "We look forward to expanding our portfolio of breast cancer medicines into the HR-positive space and bringing this potentially best-in-class new treatment option to patients as quickly as possible."
The inavolisib combination was well tolerated and adverse events were consistent with the known safety profiles of the individual study treatments, with no new safety signals observed.
Inavolisib is an oral therapy with high in vitro potency and selectivity for PI3Kα inhibition and the ability to specifically trigger the breakdown of mutant PI3Kα protein. With this unique dual mechanism of action, inavolisib may provide well-tolerated, durable disease control and potentially improved outcomes for people with HR-positive/HER2-negative, PIK3CA-mutated advanced breast cancer. PIK3CA mutations can lead to mutated PI3Kα protein which contributes to uncontrolled tumour growth, disease progression and resistance to endocrine-based treatment.
Inavolisib is currently being investigated in three Phase III clinical studies in people with PIK3CA-mutated metastatic breast cancer (INAVO120, INAVO121, INAVO122) in various combinations.
The INAVO120 study [NCT04191499] is a Phase III, randomised, double-blind, placebo-controlled study evaluating the efficacy and safety of inavolisib in combination with palbociclib and fulvestrant versus placebo plus palbociclib and fulvestrant in people with PIK3CA-mutated, hormone receptor (HR)-positive, HER2-negative, locally advanced or metastatic breast cancer whose disease progressed during treatment or within 12 months of completing adjuvant endocrine therapy and who have not received prior systemic therapy for metastatic disease.
The study included 325 patients, who were randomly assigned to either the investigational or control treatment arm. The primary endpoint is progression-free survival, as assessed by investigators, defined as the time from randomisation in the clinical trial to the time when the disease progresses, or a patient dies from any cause. Secondary endpoints include overall survival, objective response rate, and clinical benefit rate.
Inavolisib is an investigational, oral targeted treatment with best-in-class potential that could provide well-tolerated, durable disease control and potentially improved outcomes for people with hormone receptor (HR)-positive, PIK3CA-mutated breast cancer, a common yet often overlooked mutation found in approximately 40% of this population.1 Inavolisib has been designed to help minimise the overall toxicity of treatment and is differentiated from other PI3K inhibitors due to its high in vitro potency and specificity for the PI3K alpha (PI3Kα) isoform inhibition, together with its unique mechanism of action, that leads to specific degradation of mutant PI3K alpha.3,4
Inavolisib is currently being investigated in three Roche-sponsored Phase III clinical studies in PIK3CA-mutated locally advanced or metastatic breast cancer:
in combination with palbociclib and fulvestrant vs. palbociclib and fulvestrant in first-line HR-positive HER2-negative breast cancer (INAVO120),
in combination with fulvestrant vs. alpelisib plus fulvestrant in HR-positive HER2-negative breast cancer post-CDK4/6 inhibitor and endocrine combination therapy (INAVO121),6 and
in combination with pertuzumab plus trastuzumab for subcutaneous injection (Phesgo®) vs. Phesgo as maintenance therapy in 1L HER2-positive breast cancer (INAVO122)
Hormone receptor (HR)-positive breast cancer is the most prevalent type of all breast cancers.8 A defining feature of HR-positive breast cancer is that its tumour cells have receptors that attach to one or both hormones – estrogen or progesterone – which can contribute to tumour growth.9 People diagnosed with HR-positive metastatic breast cancer often face the risk of disease progression and treatment side effects, creating a need for additional treatment options.10-12 The PI3K signalling pathway is commonly dysregulated in HR-positive breast cancer, often due to activating PIK3CA mutations, which have been identified as a potential mechanism for resistance to endocrine therapy and CDK4/6 inhibitors.
Roche has been advancing breast cancer research for more than 30 years with the goal of helping as many people with the disease as possible. Our medicines, along with companion diagnostic tests, have contributed to bringing breakthrough outcomes in HER2-positive and triple-negative breast cancers. As our understanding of breast cancer biology rapidly improves, we are working to identify new biomarkers and approaches to treatment for other subtypes of the disease, including estrogen receptor-positive breast cancer, which is a form of hormone receptor-positive breast cancer, the most prevalent type of all breast cancers.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
Elecsys® HBeAg quant is an immunoassay that can be used as an early marker of acute hepatitis B infection, as well as an indicator of chronic active hepatitis in combination with other laboratory results and clinical information.
The single test will inform clinicians if treatment is required and regimens are working in conjunction with other diagnostic assays.
Almost 300 million people globally have chronic hepatitis B, causing a significant burden to health systems as it puts patients at high risk of death from cirrhosis and liver cancer.
Basel, 27 November 2023 - Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the launch of Elecsys® HBeAg quant, an immunoassay that is able to determine both the presence and quantity (qualitative and quantitative) of the hepatitis B e antigen (HBeAg) in human serum and plasma. This launch means that, when combined with other laboratory results and clinical information, patients will now get confirmation on whether they have hepatitis B (HBV), and if so, to what extent, as well as treatment monitoring through one single assay. It is for use on the cobas® e analysers in countries accepting the CE Mark. HBV is the most common type of viral hepatitis affecting people of all ages, posing a significant burden on people and healthcare systems globally.1
The new Elecsys HBeAg quant test will complement existing HBV testing markers. When a patient with suspected HBV returns a positive result of HBeAg, clinicians can determine the patient’s disease phase,2,3 assess the activity of the virus in the liver4 and risk of progressive liver disease and Hepatocellular carcinoma (HCC).5,6
“Hepatitis B affects millions of people each year and is a major global health burden. An accurate diagnosis is critical to ensuring timely treatment options. If hepatitis B is left untreated, it can cause chronic infections, putting people at high risk of death from cirrhosis and liver cancer," said Matt Sause, CEO of Roche Diagnostics. "The addition of the Elecsys® HBeAg quant immunoassay to our viral hepatitis testing portfolio underlines Roche’s commitment to tackle healthcare’s biggest challenges to support clinicians and their patients.”
The new diagnostic tool reduces complexity and improves operational efficiency thanks to a leaner workflow for lab personnel. It can also simplify the experience for patients, as they only have to undergo one single test to indicate both the presence and quantity of HBeAg. The qualitative result provided by the test can help diagnose patients with HBeAg as an early marker of acute HBV, as well as indicate chronic or active HBV. The additional quantitative answer will enhance patient management enabling the most appropriate treatment plan for patients.
The Elecsys HBeAg quant can also be used as a tool in perinatal screening7 and to monitor antiviral treatment response within infected patients, potentially supporting the implementation of a therapeutic plan tailored to individual patient’s needs.
Elecsys® HBeAg quant is an immunoassay for the in vitro qualitative and quantitative determination of hepatitis B e antigen (HBeAg) in human serum and plasma. In conjunction with other laboratory results and clinical information, HBeAg quantification may be used as an aid for the diagnosis and monitoring of patients with hepatitis B viral infection. Elecsys® HBeAg quant reports both a qualitative and a quantitative readout, providing greater value to clinicians and patients. The test can be used for samples from patients with unknown HBeAg status (first line testing) as well as those who have previously tested positive (second line testing). The immunoassay is intended for use on all available cobas e analysers.
HBV is a viral infection that attacks the liver and can cause both acute and chronic disease.1 It is the most common type of viral hepatitis affecting people of all ages, posing a significant burden on people and healthcare systems globally.1 In 2019, WHO estimated that 296 million people were living with chronic hepatitis B infection, with 1.5 million new infections each year. In this same year, hepatitis B resulted in an estimated 820,000 deaths, mostly from cirrhosis and hepatocellular carcinoma (primary liver cancer). The virus is most commonly transmitted from mother to child during birth and delivery, as well as through contact with blood or other body fluids during sex with an infected partner, unsafe injections or exposures to sharp instruments.1 While a vaccine exists to prevent HBV, there is currently no cure for patients who have been diagnosed with the infection.
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management.
Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. More than thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the twelfth consecutive year, Roche has been recognised as one of the most sustainable companies in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2020 employed more than 100,000 people worldwide. In 2020, Roche invested CHF 12.2 billion in R&D and posted sales of CHF 58.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan
